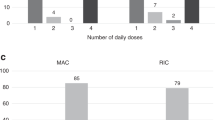
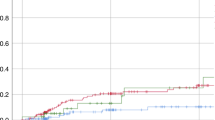
Summary:
We examined the effects of busulphan (BU) dose and patient age on toxicity and outcome in 63 children with acute leukaemia given BUCY prior to allogeneic or autologous BMT. BU was administered as four single daily oral doses, based either on weight (4 × 4 mg/kg) or surface area (4 × 150 mg/m2). BU pharmacokinetic analysis was not used to dose adjust. The average daily (mg/kg) BU dose was 43% higher for the group given 150 mg/m2 compared to the 4 mg/kg dose group. This produced a median BU AUC 61% higher than with the 4 mg/kg dose. Only one child did not achieve full allogeneic donor engraftment. Regimen-related toxicity was low. Although younger children had faster BU clearance, the 4 × 150 mg/m2 dose ensured equivalent systemic exposure to BU, and resulted in a high frequency of engraftment without a significant increase in serious toxicity. BU, given as four single daily doses of 150 mg/m2, is appropriate and safe in all age groups of children. Given the reliable pharmacokinetics, low toxicity and high rate of allogeneic engraftment, there is no need for routine pharmacokinetic monitoring or dose modifications. This dosage regimen may be applicable for use with i.v. BU.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Santos GW, Tutschka PJ, Brookmeyer R et al. Marrow transplantation for acute nonlymphocytic leukaemia after treatment with busulfan and cyclophosphamide. N Engl J Med 1983; 309: 1347–1353.
Tutschka PJ, Copelan EA, Kapoor N et al. Allogeneic bone marrow transplantation for leukaemia using chemotherapy as conditioning: 6-year results of a single institution trial. Transplant Proc 1991; 23: 1709–1710.
Lucarelli G, Polchi P, Galimberti M et al. Marrow transplantation for thalassemia following busulphan and cyclophosphamide. Lancet 1985; 1: 1355–1357.
Shaw PJ, Hugh-Jones K, Hobbs JR et al. Busulphan and cyclophosphamide cause little early toxicity during displacement bone marrow transplantation in fifty children. Bone Marrow Transplant 1986; 1: 193–200.
Hobbs JR, Hugh–Jones K, Shaw PJ et al. Engraftment rates related to busulphan and cyclophosphamide dosages for displacement bone marrow transplants in fifty children. Bone Marrow Transplant 1986; 1: 201–208.
Grochow LB, Krivit W, Whitley CB, Blazar B . Busulfan disposition in children. Blood 1990; 75: 1723–1727.
Hassan M, Oberg G, Bekassy AN et al. Pharmacokinetics of high-dose busulphan in relation to age and chronopharmaco-logy. Cancer Chemother Pharmacol 1991; 28: 130–134.
Regazzi MB, Locatelli F, Buggia I et al. Disposition of high-dose busulphan in pediatric patients undergoing bone marrow transplantation. Clin Pharmacol Ther 1993; 54: 45–52.
Shaw PJ, Scharping CE, Brian RJ, Earl JW . Busulphan pharmacokinetics using a single daily high dose regimen in children with acute leukemia. Blood 1994; 84: 2357–2362.
Vassal G, Deroussent A, Challine D et al. Is 600 mg/m2 the appropriate dosage of busulphan in children undergoing bone marrow transplantation? Blood 1992; 79: 2475–2479.
Yeager AM, Wagner Jr JE, Graham ML et al. Optimization of busulphan dosage in children undergoing bone marrow transplantation: a pharmacokinetic study of dose escalation. Blood 1992; 80: 2425–2428.
Poonkuzhali B, Srivastava A, Quernin MH et al. Pharmacokinetics of oral busulphan in children with beta thalassaemia major undergoing allogeneic bone marrow transplantation. Bone Marrow Transplant 1999; 24: 5–11.
Shaw PJ, Bergin ME, Burgess MA et al. Childhood acute myeloid leukaemia; outcome in a single centre using chemotherapy and consolidation with busulfan/cyclophosphamide for bone marrow transplantation. J Clin Oncol 1994; 12: 2138–2145.
O’Brien TA, Russell SJ, Vowels MR et al. Results of consecutive trials for children newly diagnosed with acute myeloid leukemia from the Australian and New Zealand Children's Cancer Study Group. Blood 2002; 100: 2708–2716.
Bearman SI, Appelbaum FR, Buckner CD et al. Regimen-related toxicity in patients undergoing bone marrow transplantation. J Clin Oncol 1988; 6: 1562–1568.
MacDonald GB, Cole DE, Balis F et al. Veno-occlusive disease of the liver after bone marrow transplantation: diagnosis, incidence, and predisposing factors. Hepatology 1984; 4: 116–122.
Smith A, Robson LG, Sharma P, Shaw PJ . Application of interphase FISH on direct bone marrow smears for evidence of chimerism in paediatric sex-mismatched bone marrow transplantation. Pathology 1999; 31: 25–28.
Slattery JT, Sanders JE, Buckner CD et al. Graft-rejection and toxicity following bone marrow transplantation in relation to busulphan pharmacokinetics. Bone Marrow Transplant 1995; 16: 31–42.
Slattery JT, Clift RA, Buckner CD et al. Marrow transplantation for chronic myeloid leukemia: the influence of plasma busulphan levels on the outcome of transplantation. Blood 1997; 89: 3055–3060.
Bolinger AM, Zangwill AB, Slattery JT et al. An evaluation of engraftment, toxicity and busulfan concentration in children receiving bone marrow transplantation for leukemia or genetic disease. Bone Marrow Transplant 2000; 25: 925–930.
Ljungman P, Hassan M, Bekassy AN et al. High busulphan concentrations are associated with increased transplant related mortality in allogeneic bone marrow transplant patients. Bone Marrow Transplant 1997; 20: 909–913.
Dix SP, Wingard JR, Mullins RE et al. Association of busulfan area under the curve with veno-occlusive disease following BMT. Bone Marrow Transplant 1996; 17: 225–230.
Grochow LB . Busulfan disposition: the role of therapeutic monitoring in bone marrow transplantation induction regimens. Semin Onc 1993; 20: 18–25.
Dupuis LL, Najdova M, Saunders EF . Retrospective appraisal of busulfan dose adjustment in children. Bone Marrow Transplant 2000; 26: 1143–1147.
Baker KS, Bostrom B, DeFor T et al. Busulfan pharmacokinetics do not predict relapse in acute myeloid leukemia. Bone Marrow Transplant 2000; 26: 607–614.
Ljungman P, Hassan M, Bekassy AN et al. Busulphan concentration in relation to permanent alopecia in recipients of bone marrow transplants. Bone Marrow Transplant 1995; 15: 869–871.
Grochow LB, Jones RJ, Brundrett RB et al. Pharmacokinetics of busulphan: correlation with venoocclusive disease in patients undergoing bone marrow transplantation. Cancer Chemother Pharmacol 1989; 25: 55–61.
Meresse V, Hartmann O, Vassal G et al. Risk factors for hepatic veno-occlusive disease after high dose busulphan containing regimens followed by autologous bone marrow transplantation: a study in 136 children. Bone Marrow Transplant 1992; 10: 135–141.
Vassal G, Koscielny S, Challine D et al. Busulphan disposition and hepatic veno-occlusive disease in children undergoing bone marrow transplantation. Cancer Chemother Pharmacol 1996; 37: 247–253.
Hugh-Jones K, Shaw PJ . No convulsions in children on high-dose busulphan. Lancet 1985; i: 220.
Hassan M, Oberg G, Bjorkholm M et al. Influence of prophylactic anticonvulsant therapy on high-dose busulfan kinetics. Cancer Chemother Pharmacol 1993; 33: 181–186.
Russell JA, Tran HT, Quinlan D et al. Once-daily intravenous busulphan given with fludarabine as conditioning for allogeneic stem cell transplantation: study of pharmacokinetics and early clinical outcomes. Biol Blood Marrow Transplant 2002; 8: 468–476.
Acknowledgements
CE Nath is supported by the Leukaemia Research and Support Fund. We would like to thank the Oncologists whose patients were included in the study, the nurses in the Oncology Unit for collecting blood samples and for excellent patient care.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information accompanies the paper on Bone Marrow Transplantation website (http://www.nature.com/bmt)
Supplementary information
Rights and permissions
About this article
Cite this article
Shaw, P., Nath, C., Berry, A. et al. Busulphan given as four single daily doses of 150 mg/m2 is safe and effective in children of all ages. Bone Marrow Transplant 34, 197–205 (2004). https://doi.org/10.1038/sj.bmt.1704560
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704560
Keywords
This article is cited by
-
Appropriate busulfan dosing
Bone Marrow Transplantation (2005)