Summary:
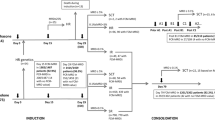
This study was designed to evaluate the results of high-dose therapy followed by purged autologous stem cell transplantation (ASCT) for patients with low-grade follicular non Hodgkin's lymphoma (LGFL), and the prognostic significance of PCR detection of residual Bcl-2/IgH-positive cells after ASCT. Between 1992 and 1998, 49 patients with LGFL received total body irradiation and high-dose cyclophosphamide followed by purged ASCT. PCR amplification of the Bcl-2/IgH rearrangement was performed at diagnosis, on stem cell collections before and after purging and on bone marrow and blood samples after ASCT. With a median follow-up of 76 months (37–103) 34 patients remain alive and event-free. A total of 20 patients had disease recurrence, three patients developed secondary myelodysplastic syndrome (MDS). In all, 11 patients died; 10 deaths were because of recurrent disease, one because of MDS. Kaplan–Meier estimates of event-free survival (EFS) and overall survival (OS) at 5 years were 65% (±7%) and 77% (±6%), respectively. Patients who achieved a sustained molecular complete response (CR) had a lower risk of disease recurrence and experienced significantly longer EFS (93% (±6%) vs 11% (±7%) P=0.0008) and OS (100 vs 55% (±12%) P=0.0057). In conclusion, myeloablative therapy followed by purged ASCT may induce long EFS in patients with LGFL. The achievement of sustained molecular CR after ASCT improves EFS and OS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Longo DL . What's the deal with follicular lymphomas? J Clin Oncol 1993; 11: 202–208.
Horning SJ . Treatment approaches to the low-grade lymphomas. Blood 1994; 83: 881–884.
Dana BW, Dahlberg S, Nathwani N et al. Long-term follow-up patients with low-grade malignant lymphomas treated with doxorubicin-based chemotherapy or chemoimmunotherapy. J Clin Oncol 1993; 11: 644–651.
Solal-Celigny P, Lepage E, Brousse N et al. Recombinant interferon alfa-2b combined with a regimen containing doxorubicin in patients with advanced follicular lymphoma. N Engl J Med 1993; 329: 1608–1614.
Schouten C, Bierman J, Vaughan P et al. Autologous bone marrow transplantation in follicular non-Hodgkin's lymphoma before and after histologic transformation. Blood 1989; 74: 2579–2584.
Colombat P, Donadio D, Fouillard L et al. Value of autologous bone marrow transplantation in follicular lymphoma: a France autogreffe retrospective study of 42 patients. Bone Marrow Transplant 1994; 13: 157–162.
Haas R, Moos M, Karcher A et al. Sequential high-dose therapy with peripheral blood progenitor-cell support in low-grade non-Hodgkin's lymphoma. J Clin Oncol 1994; 13: 1685–1692.
Bastion Y, Price P, Haioun C et al. Intensive therapy with peripheral blood progenitor cell transplantation in 60 patients with poor-prognosis follicular lymphoma. Blood 1995; 8: 3257–3262.
Haas R, Moos M, Mohle R et al. High-dose therapy with peripheral blood progenitor cell transplantation in low-grade non-Hodgkin's lymphoma. Bone Marrow Transplant 1996; 17: 149–155.
Bierman J, Vose M, Anderson R et al. High-dose therapy with autologous hematopoietic rescue for follicular low-grade non-Hodgkin's lymphoma. J Clin Oncol 1997; 15: 445–450.
Freedman S, Ritz J, Neuberg D et al. Autologous bone marrow transplantation in 69 patients with a history of low-grade B-cell non-Hodgkin's lymphoma. Blood 1991; 77: 2524–2529.
Rohatiner S, Johnson M, Price A et al. Myeloablative therapy with autologous bone marrow transplantation as consolidation therapy for recurrent follicular lymphoma. J Clin Oncol 1994; 12: 1177–1184.
Johnson PWM, Price CGA, Smith T et al. Detection of cells bearing the t(14;18) translocation following myeloablative treatment and autologous bone marrow transplantation for follicular lymphoma. J Clin Oncol 1994; 12: 798–805.
Hardingham E, Kotasek D, Sage E et al. Significance of molecular marker-positive cells after autologous peripheral-blood stem-cell transplantation for non-Hodgkin's lymphoma. J Clin Oncol 1995; 13: 1073–1076.
Freedman S, Gribben JG, Neuberg D et al. High-dose therapy and autologous bone marrow transplantation in patients with follicular lymphoma during first remission. Blood 1996; 7: 2780–2786.
Moos M, Schulz R, Martin S et al. The remission status before and the PCR status after high-dose therapy with peripheral blood stem cell support are prognostic factors for relapse-free survival in patients with follicular non-Hodgkin's lymphoma. Leukemia 1998; 12: 1971–1976.
Freedman AS, Neuberg D, Mauch P et al. Long-term follow-up of autologous bone marrow transplantation in patients with relapsed follicular lymphoma. Blood 1999; 10: 3325–3333.
Apostolodis J, Gupta R, Grenzelias D et al. High-dose therapy with autologous bone marrow support as consolidation of remission in follicular lymphoma: long-term clinical and molecular follow-up. J Clin Oncol 2000; 3: 527–536.
Horing SJ, Negrin RS, Hoppe RT et al. High dose therapy and autologous bone marrow transplantation for follicular lymphoma in first complete or partial remission: results of a phase II clinical trial. Blood 2001; 97: 404–411.
Hardingham JE, Kotasek D, Sage RE et al. Molecular detection of residual lymphoma cells in peripheral blood stem cell harvests and following autologous transplantation. Bone Marrow Transplant 1993; 11: 15–20.
Brenner MK, Rill DR, Moen RC et al. Gene-marking to trace origin of relapse after autologous bone-marrow transplantation. Lancet 1993; 341: 85–86.
Deisseroth AB, Zu Z, Claxton D et al. Genetic marding shows that Ph+ cells present in autologous transplants of chronic myelogenous leukemia (CML) contribute to relapse after autologous bone marrow in CML. Blood 1994; 83: 3068–3076.
Weiss LM, Warnhe RA, Sklar J, Cleary ML . Molecular analysis of the t(14;18) chromosomal translocation in malignant lymphomas. N Engl J Med 1987; 317: 1185–1188.
Gribben JG, Freedman AS, Woo SD et al. All advanced stage non-Hodgkin's lymphomas with a polymerase chain reaction amplifiable breakpoint of bcl-2 have residual cells containing the bcl-2 rearrangement at evaluation and after treatment. Blood 1991; 12: 3275–3280.
Mahe B, Milpied N, Hermouet S et al. G-CSF alone mobilized sufficient peripheral blood CD 34+ cells for positive selection in newly diagnosed patients with myeloma and lymphoma. Br J Haematol 1996; 92: 263–268.
Kaplan E, Maier P . Non parametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–462.
Mantel N . Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 1966; 50: 163–168.
Gribben JG, Freedman AS, Neuberg D et al. Immunologic purging of marrow assessed by PCR before autologous bone marrow transplantation for B-cell lymphoma. N Engl J Med 1991; 325: 1525–1533.
Corradini P, Astolfi M, Cherasco C et al. Molecular monitoring of minimal residual disease in follicular and mantle cell Non-Hodgkin's lymphomas treated with high-dose chemotherapy and peripheral blood progenitor cell autografting. Blood 1997; 89: 724–731.
Gribben JG, Neuberg D, Barber M et al. Detection of residual lymphoma cells by polymerase chain reaction in peripheral blood is significantly less predictive for relapse than detection in bone marrow. Blood 1994; 83: 3800–3807.
Gribben JG, Neuberg D, Freedman AS et al. Detection by polymerase chain reaction of residual cells with the bcl-2 translocation is associated with increased risk of relapse after autologous bone marrow transplantation for B-cell lymphoma. Blood 1993; 81: 3449–3457.
Paryani SB, Hoppe RT, Cox RS et al. The role of radiation therapy in the management of stage III follicular lymphomas. J Clin Oncol 1984; 2: 841–848.
Nagler A, Acherstein A, Or R . Immunotherapy with recombinant human interleukin-2 and recombinant interferon alpha in lymphoma patients postautologous marrow on stem cell transplantation. Blood 1997; 89: 3951–3959.
Maloney DG, Grillo-Lopez AJ, White CA et al. IDEC-C2B8 (Rituximab) anti CD20 antibody in patients with relapsed low-grade non-Hodgkin's lymphoma. Blood 1997; 90: 2188–2195.
Carella AM, Cavaliere M, Lerma E et al. Autografting followed by nonmyeloablative immunosuppressive chemotherapy and allogeneic peripheral blood hematopoietic stem-cell transplantation as treatment of resistant Hodgkin's disease and non-Hodgkin's lymphoma. J Clin Oncol 2000; 18: 3918–3924.
Price CGA, Meerabux J, Murtagn S et al. The significance of circulating cells carrying t(14;18) in long remission from follicular lymphoma. J Clin Oncol 1991; 9: 1527–1532.
Acknowledgements
We gratefully acknowledge the excellent technical assistance of A Cassidanius, S Bercegeay, and C Even.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mahé, B., Milpied, N., Mellerin, M. et al. PCR detection of residual Bcl-2/IgH-positive cells after high-dose therapy with autologous stem cell transplantation is a prognostic factor for event-free survival in patients with low-grade follicular non-Hodgkin's lymphoma. Bone Marrow Transplant 31, 467–473 (2003). https://doi.org/10.1038/sj.bmt.1703829
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703829
Keywords
This article is cited by
-
Current Perspectives in Genetics of “Double-Hit” Lymphoma with Possible Clinical Implications
Cell Biochemistry and Biophysics (2014)
-
Long-term event-free survivors after high-dose therapy and autologous stem-cell transplantation for low-grade follicular lymphoma
Bone Marrow Transplantation (2005)