Summary:
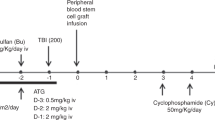
In T-cell-depleted allogeneic bone marrow transplantation (TCD-BMT) using unrelated donors, the role of donor lymphocyte infusion (DLI) for survival and disease control has not been defined. In a study of 116 patients (92 matched, 24 mismatched) who received CD3+ T-cell-depleted marrow graft, sequential infusions of escalated doses of donor T lymphocytes up to 1×106 CD3+ cells/kg were prospectively investigated. T cells were administered while patients were on cyclosporine, provided ≥grade II acute graft-versus-host-disease (GVHD) had not occurred. Acute GVHD of ≥grade II occurred in 27 of 110 (25%) patients before DLI and in 39 of 79 (49%) patients after DLI. In total, 12 of 27 (44%) patients without DLI and 44 of 72 (61%) patients who received DLI developed chronic GVHD. A total of 19 patients died of GVHD, with 17 of acute and two of chronic GVHD. Overall survival (OS) and event-free survival (EFS) at 5 years were 27 and 21%, respectively. The 2-year incidence of relapse was 14%. In multivariate analysis, only chronic GVHD was a good prognostic factor for both OS: hazard ratio (HR) 1.4, P=0.04, and EFS: HR 1.6, P=0.01. Both acute and chronic GVHD were favorable prognostic factors for relapse probability: HR 1.9 for both, P=0.02, 0.01, respectively. The 1-year cumulative incidence of transplant-related mortality (TRM), excluding cases of GVHD, was 42%. The two most common causes of 1-year non-GVHD death were viral infection (9%) and idiopathic pneumonia syndrome (12%). Although the incidence of relapse was low, the study suggests that the current scheme of DLI in unrelated TCD-BMT would not improve survival unless TRM decreases significantly.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hansen JA, Gooley TA, Martin PJ et al. Bone marrow transplants from unrelated donors for patients with chronic myeloid leukemia. N Engl J Med 1998; 338: 962–968.
Sierra J, Storer B, Hansen JA et al. Transplantation of marrow cells from unrelated donors for treatment of high-risk acute leukemia: the effect of leukemic burden, donor HLA-matching, and marrow cell dose. Blood 1997; 89: 4226–4235.
Davies SM, Kollman C, Anasetti C et al. Engraftment and survival after unrelated-donor bone marrow transplantation: a report from the national marrow donor program. Blood 2000; 96: 4096–4102.
Ash RC, Casper JT, Chitambar CR et al. Successful allogeneic transplantation of T-cell-depleted bone marrow from closely HLA-matched unrelated donors. N Engl J Med 1990; 322: 485–494.
Drobyski WR, Ash RC, Casper JT et al. Effect of T-cell depletion as graft-versus-host disease prophylaxis on engraftment, relapse, and disease-free survival in unrelated marrow transplantation for chronic myelogenous leukemia. Blood 1994; 83: 1980–1987.
Marks DI, Bird JM, Vettenranta K et al. T-cell-depleted unrelated donor bone marrow transplantation for acute myeloid leukemia. Biol Blood Marrow Transplant 2000; 6: 646–653.
Soiffer RJ, Weller E, Alyea EP et al. CD6+ donor marrow T-cell depletion as the sole form of graft-versus-host disease prophylaxis in patients undergoing allogeneic bone marrow transplant from unrelated donors. J Clin Oncol 2001; 19: 1152–1159.
Kernan NA, Collins NH, Juliano L et al. Clonable T lymphocytes in T cell-depleted bone marrow transplants correlate with development of graft-v-host disease. Blood 1986; 68: 770–773.
Marciniak E, Romond EH, Thompson JS et al. Laboratory control in predicting clinical efficacy of T cell-depletion procedures used for prevention of graft-versus-host disease: importance of limiting dilution analysis. Bone Marrow Transplant 1988; 3: 589–598.
Potter MN, Pamphilon DH, Cornish JM et al. Graft-versus-host disease in children receiving HLA-identical allogeneic bone marrow transplants with a low adjusted T lymphocyte dose. Bone Marrow Transplant 1991; 8: 357–361.
Verdonck LF, Dekker AW, de Gast GC et al. Allogeneic bone marrow transplantation with a fixed low number of T cells in the marrow graft. Blood 1994; 83: 3090–3096.
Hale G, Waldmann H . Control of graft-versus-host disease and graft rejection by T-cell depletion of donor and recipient with Campath-1 antibodies. Results of matched sibling transplants for malignant diseases. Bone Marrow Transplant 1994; 13: 597–611.
Lowenberg B, Wagemaker G, van Bekkum DW et al. Graft-versus-host disease following transplantation of ‘one log’ versus ‘two log’ T-lymphocyte-depleted bone marrow from HLA-identical donors. Bone Marrow Transplant 1986; 1: 133–140.
Atkinson K, Farrelly H, Cooley M et al. Human marrow T-cell dose correlates with severity of subsequent acute graft-versus-host disease. Bone Marrow Transplant 1987; 2: 51–57.
Verdonck LF, van Blokland WT, Bosboom-Kalsbeek EK et al. Complete donor T cell chimerism is accomplished in patients transplanted with bone marrow grafts containing a fixed low number of T-cells. Bone Marrow Transplant 1996; 18: 389–395.
Lee CK, Hayashi M, Harman GS et al. The Anti-CD2 T cell depletion with two level T cell addback in allogeneic bone marrow transplantation. Blood 1995; 86 (Suppl. 1): 570a (abstract).
Gingrich RD, Lee CK, Harman GS et al. Graded incremental donor T cell addback following allogeneic bone marrow transplantation. Blood 1995; 86 (Suppl. 1): 625a (abstract).
Lee CK, Gingrich RD, Harman GS et al. Risk factor dependent modulation of graft-versus-host disease by graded incremental T cell add-back in T cell depletion allogeneic bone marrow transplantation. Blood 1996; 88 (Suppl. 1): 418a (abstract).
Lee CK, Gingrich RD, deMagalhaes-Silverman M et al. Prophylactic reinfusion of T cells for T cell-depleted allogeneic bone marrow transplantation. Biol Blood Marrow Transplant 1999; 5: 15–27.
Deeg HJ, Cottler-Fox M . Clinical spectrum and pathophysiology of acute graft-vs-host disease. In: SJ Burakoff, HJ Deeg, Ferrara J, Atkinson K (eds). Graft-vs-Host Disease. Marcel Dekker Inc.: New York, NY, 1990, pp 311–338.
Glucksberg H, Storb R, Fefer A et al. Clinical manifestations of graft versus host disease in human recipients of marrow from HLA matched sibling donors. Transplantation 1974; 18: 295–304.
Cheson BD, Horning SJ, Coiffier B et al. Report of an international workshop to standardize response criteria for non-Hodgkin's lymphomas. NCI Sponsored International Working Group. J Clin Oncol 1999; 17: 1244–1253.
Barlogie B, Jagannath S, Desikan KR et al. Total therapy with tandem transplants for newly diagnosed multiple myeloma. Blood 1999; 93: 55–65.
Gooley TA, Leisenring W, Crowley J et al. Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999; 18: 695–706.
Ash RC, Casper JT, Chitambar CR et al. Successful allogeneic transplantation of T-cell-depleted bone marrow from closely HLA-matched unrelated donors. N Engl J Med. 1990; 322: 485–494.
Spencer A, Szydlo RM, Brookes PA et al. Bone marrow transplantation for chronic myeloid leukemia with volunteer unrelated donors using ex vivo or in vivo T-cell depletion: major prognostic impact of HLA class I identity between donor and recipient. Blood. 1995; 86: 3590–3597.
McGlave PB, Shu XO, Wen W et al. Unrelated donor marrow transplantation for chronic myelogenous leukemia: 9 years' experience of the National Marrow Donor Program. Blood 2000; 95: 2219–2225.
Champlin RE, Passweg JR, Zhang MJ et al. T-cell depletion of bone marrow transplants for leukemia from donors other than HLA-identical siblings: advantage of T-cell antibodies with narrow specificities. Blood 2000; 95: 3996–4003.
Keever-Taylor CA, Passweg J, Kawanishi Y et al. Association of donor-derived host-reactive cytolytic and helper T cells with outcome following alternative donor T cell-depleted bone marrow transplantation. Bone Marrow Transplant 1997; 19: 1001–1009.
Kaminski E, Hows J, Man S et al. Prediction of graft versus host disease by frequency analysis of cytotoxic T-cells after unrelated donor bone marrow transplantation. Transplantation 1989; 48: 608–613.
Irschick EU, Hladik F, Niederwieser D et al. Studies on the mechanism of tolerance or graft-versus-host disease in allogeneic bone marrow recipients at the level of cytotoxic T-cell precursor frequencies. Blood 1992; 79: 1622–1628.
Johnson BD, Truitt RL . Delayed infusion of immunocompetent donor cells after bone marrow transplantation breaks graft versus host tolerance and allows for persistent anti-leukemic reactivity without severe graft vs host disease. Blood 1995; 85: 3302–3312.
Weiden PL, Storb R, Tsoi MS et al. Infusion of donor lymphocytes into stable canine radiation chimeras: implications for mechanism of transplantation tolerance. J Immunol 1976; 116: 1212–1219.
Antin JH, Ferrara JL . Cytokine dysregulation and acute graft-versus-host disease. Blood 1992; 80: 2964–2968.
Szydlo R, Goldman JM, Klein JP et al. Results of allogeneic bone marrow transplants for leukemia using donors other than HLA-identical siblings. J Clin Oncol 1997; 15: 1767–1777.
Horowitz MM, Rowlings PA . An update from the International Bone Marrow Transplant Registry and the Autologous Blood and Marrow Transplant Registry on current activity in hematopoietic stem cell transplantation. Curr Opin Hematol 1997; 4: 395–400.
Kernan NA, Bartsch G, Ash RC et al. Analysis of 462 transplantations from unrelated donors facilitated by the National Marrow Donor Program. N Engl J Med 1993; 328: 593–602.
Shankar G, Scott Bryson J, Darrell Jennings C et al. Idiopathic pneumonia syndrome after allogeneic bone marrow transplantation in mice. Role of pretransplant radiation conditioning. Am J Respir Cell Mol Biol 1999; 20: 1116–1124.
Cooke KR, Krenger W, Hill G et al. Host reactive donor T-cells are associated with lung injury after experimental allogeneic bone marrow transplantation. Blood 1998; 92: 2571–2580.
McBride WH, Vegesna V . The role of T-cells in radiation pneumonitis after bone marrow transplantation. Int J Radiat Biol 2000; 76: 517–521.
Morishima Y, Sasazuki T, Inoko H et al. The clinical significance of human leukocyte antigen (HLA) allele compatibility in patients receiving a marrow transplant from serologically HLA-A, HLA-B, and HLA-DR matched unrelated donors. Blood 2002; 99: 4200–4206.
Martin PJ . How much benefit can be expected from matching for minor antigens in allogeneic marrow transplantation? Bone Marrow Transplant 1997; 20: 97–100.
Koh MB, Prentice HG, Corbo M et al. Alloantigen-specific T-cell depletion in a major histocompatibility complex fully mismatched murine model provides effective graft-versus-host disease prophylaxis in the presence of lymphoid engraftment. Br J Haematol 2002; 118: 108–116.
Soiffer RJ, Weller E, Alyea EP et al. CD6+ donor marrow T-cell depletion as the sole form of graft-versus-host disease prophylaxis in patients undergoing allogeneic bone marrow transplant from unrelated donors. J Clin Oncol 2001; 19: 1152–1159.
Lucas KG, Sun Q, Burton RL et al. A phase I–II trial to examine the toxicity of CMV- and EBV-specific cytotoxic T lymphocytes when used for prophylaxis against EBV and CMV disease in recipients of CD34-selected/T-cell-depleted stem cell transplants. Hum Gene Ther 2000; 11: 1453–1463.
Hebart H, Bollinger C, Fisch P et al. Analysis of T-cell responses to Aspergillus fumigatus antigens in healthy individuals and patients with hematological malignancies. Blood 2002; 100: 4521–4528.
Acknowledgements
We thank Colleen Chapleau for coordination for the Iowa Marrow Donor Program, Mary Dachtler for data management and all referring physicians for their dedication for the care of the patients.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, CK., deMagalhaes-Silverman, M., Hohl, R. et al. Donor T-lymphocyte infusion for unrelated allogeneic bone marrow transplantation with CD3+ T-cell-depleted graft. Bone Marrow Transplant 31, 121–128 (2003). https://doi.org/10.1038/sj.bmt.1703803
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703803
Keywords
This article is cited by
-
Cotransplantation of haploidentical hematopoietic and umbilical cord mesenchymal stem cells with a myeloablative regimen for refractory/relapsed hematologic malignancy
Annals of Hematology (2013)
-
Cytolytic activity of NK cell clones against acute childhood precursor-B-cell leukaemia is influenced by HLA class I expression on blasts and the differential KIR phenotype of NK clones
Bone Marrow Transplantation (2009)
-
Interleukin 2 regulation following semi-allogeneic bone marrow transplantation in mice
Cancer Immunology, Immunotherapy (2006)