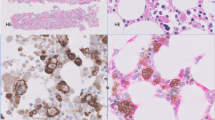
Abstract
We describe a 10-month-old boy diagnosed with X-linked hyper-IgM syndrome (XHIM) after suffering from life-threatening acute respiratory distress syndrome (ARDS) caused by Pneumocystis carinii pneumonia (PCP), although his previous clinical history and first level laboratory tests investigating immunological function did not indicate immunodeficiency. When the patient's overall condition was good, elective bone marrow transplantation from an HLA-matched older brother was performed successfully. We describe how correct diagnosis and successful treatment were made possible thanks to the involvement of a network of specialists.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
van Kooten C, Banchereau J . CD40–CD40 ligand J Leuk Biol 2000 67: 2 17
Levy J, Espanol-Boren T, Thomas C et al. Clinical spectrum of X-linked hyper-IgM syndrome J Pediatr 1997 131: 47 54
Berrington JE, Flood TJ, Abinun M et al. Unsuspected Pneumocystis carinii pneumonia at presentation of severe primary immunodeficiency Arch Dis Chid 2000 82: 144 147
Scholl PR, O'Gorman MRG, Pachman LM et al. Correction of neutropenia and hypogammaglobulinemia in X-linked hyper-IgM syndrome by allogenic bone marrow transplantation Bone Marrow Transplant 1998 22: 1215 1218
Thomas C, De Saint Basile G, Le Deist F et al. Brief report: correction of X-linked hyper-IgM syndrome by allogeneic bone marrow transplantation New Engl J Med 1995 333: 426 429
Kawai S, Sasahara Y, Minegishi M et al. Immunological reconstitution by allogeneic bone marrow transplantation in a child with the X-linked hyper-IgM syndrome Eur J Pediatr 1999 158: 394 397
Kato T, Tsuge I, Inaba J et al. Successful bone marrow transplantation in a chid with X-linked hyper-IgM syndrome Bone Marrow Transplant 1999 23: 1081 1083
Hayward AR, Levy J, Facchetti F et al. Cholangiopathy and tumours of the pancreas, liver, and biliary tree in boys with X-linked immunodeficiency with hyper-IgM J Immunol 1997 158: 977 983
Hadzic N, Pagliuca A, Rela M et al. Correction of the Hyper-IgM sindrome after liver and bone marrow transplantation New Engl J Med 2000 342: 320 323
Bernard GR, Artigas A, Brigham KL et al. Report of the American–European consensus conference on ARDS: definition, mechanism, relevant outcomes and clinical trials coordination Intens Care Med 1994 20: 225 232
Deerojanawong J, Chang AB, Eng PA et al. Pulmonary diseases in children with severe combined immune deficiency and Di George syndrome Pediatr Pulmonol 1997 130: 345 346
Lopez-Herce J, De Lucas N, Carillo A et al. Surfactant treatment for acute respiratory distress syndrome Arch Dis Child 1999 80: 248 252
Amrolia P, Gasper HB, Hassan A et al. Nonmyeloablative stem cell transplantation for congenital immunodeficiencies Blood 2000 96: 1239 1246
Alghasham AA, Nahata MC . Clinical use of fluoroquinolones in children Ann Pharmacother 2000 34: 347 359
Ball P, Mandell L, Niki Y, Tillotson G . Comparative tolerability of the newer fluoroquinolone antibacterials Drug Saf 1999 21: 407 421
Acknowledgements
This work was funded by the Italian Ministry of Health, grant No. ICS 2001/136.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leone, V., Tommasini, A., Andolina, M. et al. Elective bone marrow transplantation in a child with X-linked hyper-IgM syndrome presenting with acute respiratory distress syndrome. Bone Marrow Transplant 30, 49–52 (2002). https://doi.org/10.1038/sj.bmt.1703581
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703581