Abstract
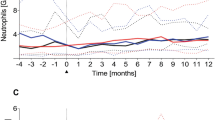
We prospectively studied the reconstitution of lymphocyte subpopulations in a group of 22 children, who survived disease-free at least 6 months after allogeneic BMT for a haematological malignancy. Absolute counts of total lymphocytes, B lymphocytes, T lymphocytes, and CD4+ helper T lymphocytes reached the 5th percentile (p5) of age-matched reference values within 6 months after BMT in 15, 17, 7 and 2 patients, respectively. In particular, CD4+ helper T lymphocyte reconstitution was very slow. Unexpectedly, CMV reactivation had a profound positive influence upon the number of CD4+ helper T lymphocytes in the children. In five patients, absolute B lymphocyte counts above the 95th percentile were reached from 6 months after BMT onwards, mimicking normal ontogeny. Unlike normal ontogeny, the percentages of helper T lymphocytes expressing the ‘naive’ CD45RA isoform were low and those expressing the ‘memory’ CD45RO isoform were high in the first 3 months after BMT, as described before. Thereafter, the CD45RA:CD45RO ratio slowly normalised. Also, CD7 expression was absent on up to 90% of T lymphocytes in the first months after BMT, and on a steadily decreasing percentage thereafter, as recently described in adults. However, the absolute counts of CD45RO+/CD4+ and CD7−/CD4+ helper T lymphocytes did not change significantly. So, we found no evidence of peripheral expansion of previously primed donor-derived ‘memory’ T lymphocytes during the follow-up period which spanned 1–18 months after BMT. The absolute counts of ‘naive’ CD45RA+ helper T lymphocytes did not show a faster increase after BMT than in adults, despite the presumed presence of a non-involuted thymus in children. Bone Marrow Transplantation (2000) 25, 267–275.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Vossen JMJJ . Bone marrow transplantation in children Baillière's Clin Pediatr 1994 1: 1029–1059
Sanders JE . Bone marrow transplantation for pediatric malignancies Pediatr Clin N Am 1997 44: 1005–1020
Serody JS, Shea TC . Prevention of infections in bone marrow transplant recipients Inf Dis Clin N Am 1997 11: 459–477
Storek J, Witherspoon RP, Storb R . T cell reconstitution after bone marrow transplantation into adult patients does not resemble T cell development in early life Bone Marrow Transplant 1995 16: 413–425
Lum LG . The kinetics of immune reconstitution after human marrow transplantation Blood 1987 69: 369–380
Atkinson K . Reconstitution of haematopoietic and immune systems after marrow transplantation Bone Marrow Transplant 1990 5: 209–226
Leino L, Lilius EM, Nikoskelainen J et al. The reappearance of 10 differentiation antigens on peripheral blood lymphocytes after allogeneic bone marrow transplantation Bone Marrow Transplant 1991 8: 339–344
Storek J, Ferrara S, Ku N et al . B cell reconstitution after human bone marrow transplantation: recapitulation of ontogeny? Bone Marrow Transplant 1993 12: 387–398
Verma UN, Mazumder A . Immune reconstitution following bone marrow transplantation Cancer Immunol Immunother 1993 37: 351–360
Storek J, Gooley T, Witherspoon RP, et al. Infectious morbidity in long-term survivors of allogeneic marrow transplantation is associated with low CD4 T cell counts Am J Hematol 1997 54: 131–138
Heitger A, Neu N, Kern H et al. Essential role of the thymus to reconstitute naive (CD45RA+) T-helper cells after human allogeneic BMT Blood 1997 90: 850–857
Pilarski LM, Gillitzer R, Zola H et al. Definition of the thymic generative lineage by selective expression of high molecular weight isoforms of CD45 (T200) Eur J Immunol 1991 19: 589–597
Mackall CL, Fleisher TA, Brown MR et al. Age, thymopoiesis, and CD4+ T lymphocyte regeneration after intensive chemotherapy New Engl J Med 1995 332: 143–149
Mackall CL, Fleisher TA, Brown MR et al. Distinctions between CD8+ and CD4+ T-cell regenerative pathways result in prolonged T-cell subset imbalance after intensive chemotherapy Blood 1997 89: 3700–3707
Foot ABM, Potter MN, Donaldson C et al. Immune reconstitution after BMT in children Bone Marrow Transplant 1993 11: 7–13
Kook H, Goldman F, Padley D et al. Reconstitution of the immune system after unrelated or partially matched T cell-depleted bone marrow transplantation in children: immunophenotyic analysis and factors affecting the speed of recovery Blood 1996 88: 1089–1097
Comans-Bitter WM, de Groot R, van den Beemd R et al. Immunophenotyping of blood lymphocytes in childhood. Reference values for lymphocyte subpopulations J Pediatr 1997 130: 388–393
Wagemaker G, Heidt PJ, Merchav S, Bekkum DW van . Abrogation of histo-incompatibility barriers in Rhesus monkeys. In: Baum SJ, Ledney GD, Thierfelder S (eds) Experimental Haematology Today Karger: Basel 1982 pp 11–118
Hebart H, Muller C, Loffler J et al. Monitoring of CMV infection: a comparison of PCR from whole blood, plasma-PCR, pp65-antigenemia and virus culture in patients after bone marrow transplantation Bone Marrow Transplant 1996 17: 861–868
Waldrop SL, Pitcher CJ, Peterson DM et al. Determination of antigen-specific memory effector CD4+ T cell frequencies by flow cytometry J Clin Invest 1997 99: 1739–1750
Klein JP, Moeschberger ML . Survival Analysis. Techniques for Censored and Truncated Data Springer: New York 1997
Turnbull BW . Nonparametric estimation of a survivorship function with double censored data J Am Stat Assoc 1974 69: 169–173
Gorla R, Airo P, Ferremi-Leali P et al. Predominance of ‘memory’ phenotype within CD4+ and CD8+ lymphocyte subsets after allogeneic BMT Bone Marrow Transplant 1993 11: 346–347
Herzenberg LA, Kantor AB, Herzenberg LA . Layered evolution in the immune system. A model for the ontogeny and develoment of multiple lymphocyte lineages. In: CD5 B cells in development and disease. Proceedings of a conference Ann NY Acad Sci 1991 651: 1–9
Small TN, Keever CA, Weiner-Fedus S et al. B-cell differentiation following autologous, conventional, or T-cell depleted bone marrow transplantation: a recapitulation of normal B-cell ontogeny Blood 1990 76: 1647–1656
Winston DJ . Infections in bone marrow transplant recipients. In: Mandell GL, Bennett JE, Dolin R (eds) Principles and Practice of Infectious Diseases Churchill Livingstone: New York 1995 2717–2722
Locksley RM, Wilson CB . Cell-mediated immunity and its role in host defense. In: Mandell GL, Bennett JE, Dolin R (eds) Principles and Practice of Infectious Diseases Churchill Livingstone: New York 1995 pp 102–149
Reusser P, Riddell SR, Meyers JD, Greenberg PD . Cytotoxic T lymphocyte response to cytomegalovirus after human allogeneic bone marrow transplantation: pattern of recovery and correlation with cytomegalovirus infection and disease Blood 1991 78: 1373–1380
Leblond V, Ben Othman T, Blanc C et al. Expansion of CD4+CD7− T cells, a memory subset with preferential interleukin-4 production, after bone marrow transplantation Transplantation 1997 64: 1453–1459
Reinhold U, Abken H . CD4+ CD7− T cells: a separate subpopulation of memory T cells? J Clin Immunol 1997 17: 265–271
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Vries, E., van Tol, M., van den Bergh, R. et al. Reconstitution of lymphocyte subpopulations after paediatric bone marrow transplantation. Bone Marrow Transplant 25, 267–275 (2000). https://doi.org/10.1038/sj.bmt.1702141
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1702141