Abstract
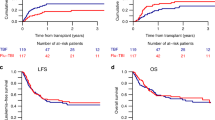
While high-dose chemotherapy and stem cell transplantation is associated with higher complete response rates than conventional chemotherapy in patients with metastatic breast cancer (MBC), its role in conferring a survival advantage is unproven. We report the results of a prospective phase II trial of 33 patients accrued between 1996 to 1998 with chemosensitive MBC, who received cyclophosphamide (Cy) 2000 mg/m2/day and carboplatin (Cb) 600 mg/m2/day for 3 consecutive days, followed by infusion of peripheral blood stem cells cultured in IL-2 for 24 h on day 0 as adoptive immunotherapy. Low-dose interleukin-2 (IL-2) was administered from day 0 to +4 and/or +7 to +11, +14 to +18, +21 to +25, then 5 days per month for 11 months to augment a graft-versus-tumor effect. The results of this study were compared to those of a historical control group treated with an identical high-dose Cb + Cy regimen with SCT but without IL-2 treatment. Only gastrointestinal (GI) toxicity was more frequent in the IL-2 cohort (P = 0.0031). At a median follow-up of 18.6 months, the median progression-free survival (PFS) is 9 months (2.4–40) and the median OS has not been reached yet. The Kaplan–Meier estimated 2 year PFS is 35%, compared with 17% in the control arm (P = 0.73), and the estimated 2 year OS is 78%, compared with 61% in the control arm (P = 0.22). Multivariate analysis showed that ER status was an independent predictor for OS and PFS, and less chemotherapy prior to HDCSCT predicted for a better PFS. These results show that augmenting HDC with IL-2 activated SCT is well-tolerated. Whether a therapeutic advantage is achievable in patients with MBC remains to be determined. Bone Marrow Transplantation (2000) 25, 19–24.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Guillaume T, Rubenstein DB, Symann M . Immune reconstitution and immunotherapy after hematopoietic stem cell transplantation Blood 1998 92: 1471–1490
Charak BS, Malloy B, Agah R et al. Interaction of various cytokines with interkeukin-2 in the generation of killer cells from human bone marrow: application in the purging of leukemia Leukemia Res 1991 9: 801–810
Charak BS, Brynes RK, Chogyoji M et al. Graft vesus leukemia effect of interleukin-2 activated bone marrow: correlation with eradication of residual disease Transplantation 1993 56: 31–37
Meehan KR, Verma UN, Cahill R et al. Interleukin-2-activated hematopoietic stem cell transplantation for breastcancer: investigation of dose level with clinical correlates Bone Marrow Transplant 1997 20: 643–651
Charak BS, Brynes RK, Katsuda S et al. Induction of graft versus leukemia effect in bone marrow transplantation: dosage and time schedule dependency of interleukin-2 therapy Cancer Res 1991 51: 2015–2020
Charak BS, Verma UN, Mazumder A . Immunomodulation in autologous bone marrow transplantation: experimental approaches. In: Spitzer T, Mazumder A (eds) Immunotherapy and Bone Marrow Transplantation Futura: Armonk, NY 1995 pp 35–58
Agah R, Malloy B, Kerner M et al. Generation and characterization of IL-2 activated bone marrow cells as potent graft versus tumor effectors in transplantation J Immunol 1989 143: 3093–3099
Meehan KR, Verma UN, Rajagopal C et al. Stem cell transplantation with chemoradiotherapy myeloablation and interleukin-2 J Infus Chemother 1996 6: 28–32
Lotze MT, Chang AE, Seipp CA et al. High dose recombinant IL-2 in the treatment of patients with disseminated cancer JAMA 1986 256: 3117–3124
Spitzer TR, Cirenza E, McAfee S et al. Phase I–II trial of high-dose cyclophosphamide, carboplatin and autologous bone marrow or peripheral blood stem cell rescue Bone Marrow Transplant 1995 15: 537–542
Kaplan EL, Meier P . Non parametric estimation from incomplete observations J Am Stat Assoc 1958 53: 457–481
Peto R, Peto J . Asymmetrically efficient rank invariant test procedures J R Stat Soc A 1972 35: 185–206
Cox DR . Regression models and life tables J R Stat Soc B 1972 34: 187–220
Hortobagyi GN . Treatment of breast cancer New Engl J Med 1998 339: 974–984
Rahman ZU, Frye DK, Smith TL et al. Results and long-term follow-up for 1581 patients with metastatic breast carcinoma treated with standard dose doxorubicin-containing chemotherapy Cancer 1999 85: 104–111
Robinson N, Benyunes MC, Thompson JA et al. Interleukin-2 after autologous stem cell transplantation for hematologic malignancy – a phase I/II study Bone Marrow Transplant 1997 19: 435–442
Miller JS, Tessmer-Tuck J, Pierson BA et al. Low dose subcutaneous interleukin-2 after autologous transplantation generates sustained in vivo natural killer activity Biol Blood Marrow Transplant 1997 3: 34–44
Soiffer RJ, Murray C, Shapiro C et al. Expansion and manipulation of natural killer cells in patients with metastatic cancer by low-dose continuous infusion and intermittent bolus administration of interleukin 2 Clin Cancer Res 1996 2: 493–499
Areman EM, Mazumder A, Kotula PL et al. Hematopoietic potential of IL-2 cultured peripheral blood stem cells from breast cancer patients Bone Marrow Transplant 1999 23: 27–33
Kopp WC, Holmlund JT . Cytokines and immunological monitoring. In Pinedo HM, Longo DL, Chabner BA (eds) Cancer Chemotherapy and Biological Response Modifiers Annual 16 Elsevier Science BV: Amsterdam 1996 pp 189–238
Salazar R, Sola C, Maroto P et al. Infectious complications in 126 patients treated with high-dose chemotherapy and autologous peripheral blood stem cell transplantation Bone Marrow Transplant 1999 23: 27–33
Hudis CA, Munster PN . High-dose therapy for breast cancer Semin Oncol 1999 26: 35–47
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Toh, H., McAfee, S., Sackstein, R. et al. High-dose cyclophosphamide + carboplatin and interleukin-2 (IL-2) activated autologous stem cell transplantation followed by maintenance IL-2 therapy in metastatic breast carcinoma – a phase II study. Bone Marrow Transplant 25, 19–24 (2000). https://doi.org/10.1038/sj.bmt.1702091
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1702091
Keywords
This article is cited by
-
Interleukin-2 and granulocyte–macrophage–colony-stimulating factor immunomodulation with high-dose chemotherapy and autologous hematopoietic stem cell transplantation for patients with metastatic breast cancer
International Journal of Hematology (2009)
-
Prognostic analysis of pre-transplant peripheral T-cell levels in patients receiving an autologous hematopoietic progenitor-cell transplant
Bone Marrow Transplantation (2005)
-
IL-2-based immunotherapy after autologous transplantation for lymphoma and breast cancer induces immune activation and cytokine release: a phase I/II trial
Bone Marrow Transplantation (2003)
-
High-dose carboplatin and regimen-related toxicity following autologous bone marrow transplant
Bone Marrow Transplantation (2002)
-
Administration of low-dose interleukin-2 plus G-CSF/EPO early after autologous PBSC transplantation: effects on immune recovery and NK activity in a prospective study in women with breast and ovarian cancer
Bone Marrow Transplantation (2002)