Abstract
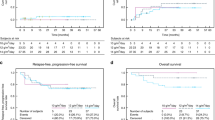
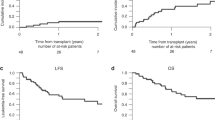
We conducted a prospective pilot study to assess the feasibility and safety of high-dose busulfan/melphalan as conditioning therapy prior to autologous PBPC transplantation in pediatric patients with high-risk solid tumors. From January 1995 to January 1999, 30 patients aged 2–21 years (median 8) were entered into the study. There were 14 females and 16 males. Diagnoses included neuroblastoma in 10 patients; Ewing’s sarcoma and peripheral neuroectodermal tumor (PNET) in 15 patients and rhabdomyosarcoma in five patients. Treatment consisted of busulfan 16 mg/kg, orally over 4 days (from days −5 to −2) in 6 hourly divided doses, and melphalan at a dose of 140 mg/m2 given by intravenous infusion over 5 min on day −1. G-CSF mobilized PBPC were used as autologous stem-cell rescue. One patient developed a single generalized convulsion during busulfan therapy. The most relevant non-hematologic toxicity was gastrointestinal, manifesting as grade 2–3 mucositis and diarrhea in 12 patients. Two patients died of procedure-related complications, one from veno-occlusive disease of liver and multiorgan failure and the other from adult respiratory distress syndrome. Probability of treatment-related mortality was 6.6 ± 4.5%. With a median follow-up of 18 months (range, 1–48), 19 patients are alive and disease-free, the actuarial EFS at 4 years being 55 ± 12% for the whole group. We conclude that high-dose busulfan/melphalan for autologous transplantation in children with solid tumors is feasible even in small patients. It is well-tolerated, with an acceptable transplant-related mortality and has proven antitumor activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Diaz, M., Vicent, M. & Madero, L. High-dose busulfan/melphalan as conditioning for autologous PBPC transplantation in pediatric patients with solid tumors. Bone Marrow Transplant 24, 1157–1159 (1999). https://doi.org/10.1038/sj.bmt.1702042
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1702042
Keywords
This article is cited by
-
Stability of Melphalan in 0.9 % Sodium Chloride Solutions Prepared in Polyvinyl Chloride Bags for Intravenous Injection
Drugs in R&D (2015)
-
Do pharmacokinetic polymorphisms explain treatment failure in high-risk patients with neuroblastoma?
European Journal of Clinical Pharmacology (2011)
-
Keratinocyte growth factor is effective in the prevention of intestinal mucositis in patients with hematological malignancies treated with high-dose chemotherapy and autologous hematopoietic SCT: a video-capsule endoscopy study
Bone Marrow Transplantation (2008)
-
High-dose therapy with hematopoietic stem cell rescue in patients with poor prognosis Ewing family tumors
Bone Marrow Transplantation (2008)
-
Elevated Plasma Ferritin and Busulfan Pharmacodynamics During High-dose Chemotherapy Regimens in Children with Malignant Solid Tumors
Clinical Pharmacology & Therapeutics (2007)