Abstract
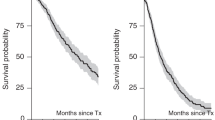
Following ASCT for multiple myeloma, it is unclear whether relapse is due solely to the presence of residual myeloma cells after myeloablation, or whether it is in part attributable to contamination of the stem cell harvest with viable malignant cells. Positive selection of CD34+ cells markedly reduces plasma cell contamination. We performed a case controlled analysis in which 15 patients with myeloma who underwent autologous PBSCT with CD34+cell selection using the Ceprate System (index group), were compared with 15 matched controls. All subjects received an identical preparative regimen. The median times to neutrophils ⩾0.5 × 109/l and unsupported platelets ⩾50 × 109/l were 14 and 23 days for the CD34+cell selected group and 11 (P = 0.03) and 14 (P = 0.029) for the case controls. Median follow-up of purged patients from autologous PBSCT was 32 months (range 18–43). At 36 months, the probability of PFS was 47 ± 14% and 46 ± 14% in the index and control groups (P = 0.44). The 3 year probability of OS was 69 ± 13% for the CD34+ cell selected arm and 66 ± 12.4% in unpurged patients (P = 0.91). Median PFS for the cell selected group is 24 months (CI 19.1–36.0), and 29 months for controls (CI 7.1–50.9). Eleven patients undergoing cell selection remain alive, seven of whom are progression free. At the same time-point after unpurged autologous PBSCT, the corresponding figures are 12 patients alive, with seven remaining progression free. Autologous PBSCT with CD34+ cell selection is both feasible and safe, but results in delayed engraftment as compared to case controls. The 3 year probability of PFS and OS in the cell selected arm was similar to that of the unpurged controls. Our findings indicate that autologous PBSCT with CD34+ cell selection appears not to have any favourable effect on disease progression. However, the results of this case controlled analysis should be cautiously interpreted, and the role of CD34+ selection in autologous PBSCT should be further investigated by large randomised trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gandhi, M., Jestice, H., Scott, M. et al. A comparison of CD34+ cell selected and unselected autologous peripheral blood stem cell transplantation for multiple myeloma: a case controlled analysis. Bone Marrow Transplant 24, 369–375 (1999). https://doi.org/10.1038/sj.bmt.1701938
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1701938
Keywords
This article is cited by
-
Dexamethasone, paclitaxel, etoposide, cyclophosphamide (d-TEC) and G-CSF for stem cell mobilisation in multiple myeloma
Bone Marrow Transplantation (2001)
-
Antibody responses to vaccinations given within the first two years after transplant are similar between autologous peripheral blood stem cell and bone marrow transplant recipients
Bone Marrow Transplantation (2001)
-
Stem cell transplantation for autoimmune diseases
Springer Seminars in Immunopathology (2001)
-
Lack of benefit of CD34+ cell selected over non-selected peripheral blood stem cell transplantation in multiple myeloma: results of a single center study
Leukemia (2000)
-
CD34: To select or not to select? That is the question
Leukemia (2000)