Abstract
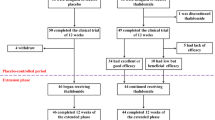
The pharmacokinetics of busulphan were studied in 23 thalassaemic children undergoing BMT. Patients received busulphan at a dose of either 16 mg/kg with cyclophosphamide and ATG (Group A) or 600 mg/m2 (with cyclophosphamide alone) (Group B) in 16 divided doses every 6 h over 4 days. Busulphan levels were analyzed by a modified GC-MS method. The dose of busulphan/kg for patients in group B was 64% (range 56–71%) higher than that for patients in group A. The mean AUC, Css, Cmax and MRV were significantly higher in group B as compared with group A for both doses 1 and 13. There was no significant difference in Vd/F, T1/2 and Kel between the two groups. A significant decrease in AUC and Css was found between 1st and 13th doses in group B, but not in group A. The Cl/F values in group A were significantly higher than those in group B after dose 1, but not after dose 13. No increase in toxicity due to the higher dose of busulphan was noted. We conclude that busulphan at 600 mg/m2 results in much higher systemic exposure to the drug as compared to 16 mg/kg, without increase in toxicity in children with beta thalassaemia major.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Poonkuzhali, B., Srivastava, A., Quernin, M. et al. Pharmacokinetics of oral busulphan in children with beta thalassaemia major undergoing allogeneic bone marrow transplantation. Bone Marrow Transplant 24, 5–11 (1999). https://doi.org/10.1038/sj.bmt.1701814
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1701814
Keywords
This article is cited by
-
Population pharmacokinetics of cyclophosphamide in patients with thalassemia major undergoing HSCT
Bone Marrow Transplantation (2012)
-
Carbonyl reductase 1 expression influences daunorubicin metabolism in acute myeloid leukemia
European Journal of Clinical Pharmacology (2012)
-
Stem cell transplantation in India
Bone Marrow Transplantation (2008)
-
Elevated Plasma Ferritin and Busulfan Pharmacodynamics During High-dose Chemotherapy Regimens in Children with Malignant Solid Tumors
Clinical Pharmacology & Therapeutics (2007)
-
Prospective validation of a novel IV busulfan fixed dosing for paediatric patients to improve therapeutic AUC targeting without drug monitoring
Cancer Chemotherapy and Pharmacology (2007)