Abstract
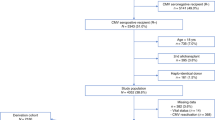
At our institution, the cytomegalovirus (CMV) prophylaxis protocol for allogeneic bone marrow transplant (BMT) recipients who are CMV-seropositive or receive marrow from a CMV-seropositive donor consists of a surveillance bronchoscopy approximately 35 days post-transplant. Patients with a positive surveillance bronchoscopy for CMV receive pre-emptive ganciclovir. In order to determine the utility of other screening methods for CMV, we prospectively performed weekly CMV antigenemia, and blood, urine and throat cultures from time of engraftment to day 120 post-BMT in 126 consecutive patients. Pre-emptive ganciclovir was given to 11/81 patients (13.6%) because of a positive surveillance bronchoscopy for CMV. Results of CMV blood, urine and throat cultures and the antigenemia assay done prior to or at the time of the surveillance bronchoscopy were analyzed for their ability to predict the bronchoscopy result. The antigenemia test had the highest positive and negative predictive values (72% and 96%, respectively). The ability of these tests to predict CMV disease was evaluated in the 70 patients with a negative surveillance bronchoscopy who did not receive pre-emptive ganciclovir. Of 19 cases of active CMV disease, CMV antigenemia was positive in 15 patients (79%) a mean of 34 days preceding symptoms. Blood cultures were positive in 14/19 patients (74%) a mean of 31 days before onset of disease. CMV antigenemia is useful for predicting the surveillance bronchoscopy result, and also predicts the development of CMV disease in the majority of patients missed by the surveillance bronchoscopy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Humar, A., O’Rourke, K., Lipton, J. et al. The clinical utility of CMV surveillance cultures and antigenemia following bone marrow transplantation. Bone Marrow Transplant 23, 45–51 (1999). https://doi.org/10.1038/sj.bmt.1701525
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1701525
Keywords
This article is cited by
-
Active human herpesvirus infections in adults with systemic lupus erythematosus and correlation with the SLEDAI score
Advances in Rheumatology (2020)
-
Cytomegalovirus viral load monitoring after allogeneic bone marrow transplantation in patients receiving antiviral prophylaxis
Bone Marrow Transplantation (2003)
-
Utility of antigenemia test for cytomegalovirus detection in hematopoietic transplant recipients: report of experiences from Medellín, Colombia
Bone Marrow Transplantation (2003)
-
High frequency of positive surveillance for cytomegalovirus (CMV) by PCR in allograft recipients at low risk of CMV
Bone Marrow Transplantation (2001)
-
Screening for cytomegalovirus (CMV) infection in allogeneic bone marrow transplantation using a quantitative whole blood polymerase chain reaction (PCR) method: analysis of potential risk factors for CMV infection
Bone Marrow Transplantation (2001)