Abstract
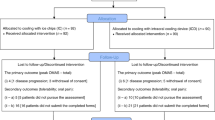
Mucositis remains an important problem following BMT and may delay discharge from hospital. Patient-controlled analgesia (PCA) systems have been reported to be of benefit in controlling BMT-associated mucositis. The present study comprised 65 patients (age range 16–68 years; 19 allografts, 29 peripheral blood stem cell autografts and 17 autologous bone marrow). Subjects were prospectively randomised to receive intravenous diamorphine for pain relief either by conventional continuous infusion (CI) or by PCA, using a Medex Walkman 440 delivery system. Each patient assessed his/her pain control and nausea daily by a visual analogue scale. Twenty-two patients did not require any diamorphine. Four patients required diamorphine for pain other than mucositis, and four patients failed PCA control. Of 35 assessable cases, no difference in pain control was noted between CI and PCA. However, PCA-controlled patients required significantly less diamorphine than CI controlled patients (mean, 131 ± 23 mg for PCA vs 296 ± 40 mg for CI; P = 0.001), and PCA required fewer days of diamorphine than CI (mean, 7.17 ± 0.66 days for PCA, 9.00 ± 0.65 days for CI; P = 0.03). Side-effects were minimal and equivalent in the two arms. The findings suggest that PCA and CI offer equivalent control of the pain of BMT-associated mucositis, but PCA requires less total consumption and duration of diamorphine therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pillitteri, L., Clark, R. Comparison of a patient-controlled analgesia system with continuous infusion for administration of diamorphine for mucositis. Bone Marrow Transplant 22, 495–498 (1998). https://doi.org/10.1038/sj.bmt.1701370
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1701370
Keywords
This article is cited by
-
Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients
Supportive Care in Cancer (2013)
-
Palliative care in BMT
Bone Marrow Transplantation (2009)
-
Opioids in pain management of blood-related malignancies
Annals of Hematology (2006)
-
Transdermal fentanyl in HSCT patients: an open trial using transdermal fentanyl for the treatment of oral mucositis pain
Bone Marrow Transplantation (2004)
-
A multivariable analysis of factors influencing mucositis after autologous progenitor cell transplantation
Bone Marrow Transplantation (2002)