Abstract
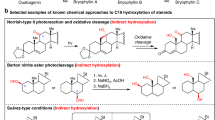
EARLY in 1948, I discussed by letter with Prof. L. F. Fieser the stereochemical course of reactions at, or adjacent to, C17 of the steroid nucleus. The experimental facts may be summarized as follows1.  Steroids with normal configuration at C14 undergo reactions exemplified by A, B, C and D to afford largely or almost exclusively a single 17-epimeride in which the persisting extracyclic C17-valency bond is β-orientated:
Steroids with normal configuration at C14 undergo reactions exemplified by A, B, C and D to afford largely or almost exclusively a single 17-epimeride in which the persisting extracyclic C17-valency bond is β-orientated: 
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Cf. Fieser, L. F., and Fieser, M., “Natural Products Related to Phenanthrene”, 410, 411 (3rd edit. New York: Reinhold, 1949).
Dostrovsky, Ingold and Hughes, J. Chem. Soc., 173 (1946).
Whitmore and Rothrock, J. Amer. Chem. Soc., 54, 343 (1932).
Quayle and Norton, J. Amer. Chem. Soc., 62, 1170 (1940).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SHOPPEE, C. Stereochemical Course of Reactions at Steroid C17. Nature 166, 107–108 (1950). https://doi.org/10.1038/166107a0
Issue Date:
DOI: https://doi.org/10.1038/166107a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.