Abstract
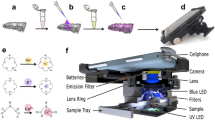
FLAME photometric analysis1 of cations in biological fluids and tissues is, because of its simplicity, rapidity and greater accuracy, replacing laborious chemical procedures. The vaporization of alkali metal solutions in constant amount per minute into a controlled low-temperature air–acetylene flame produces simple emission spectra, the light intensities of which, measured photo-electrically after slit isolation of the most sensitive line spectrum for the particular element (potassium red flame at 7665 A.), are practically proportional to their concentrations for low concentrations2.
Similar content being viewed by others
Article PDF
References
Berry, J. W., Chappell, D. G., and Barnes, R. B., Indust. Chem. Eng., Anal. Ed., 18, 19 (1946).
Bernstein, R. E., S.A. J. Med. Sci., 14, 163 (1949)
Parks, T. D., Johnson, H. O., and Lykken, L., Anal. Chem., 20, 822 (1948).
Cumings, J. N., J. Clin. Path., 1, 173 (1948).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BERNSTEIN, R. Serum Potassium by Internal Standard Flame Photometry. Nature 165, 649 (1950). https://doi.org/10.1038/165649a0
Issue Date:
DOI: https://doi.org/10.1038/165649a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.