Abstract
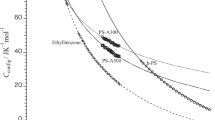
IN examining the properties of polymer-solvent systems, considerable attention has been given in the last decade to the evaluation of the configurational contribution to the partition function. It has been shown1 that the greater part of the divergence of these systems from ideal behaviour is due to the vast differences in the sizes and shapes of the molecules of the polymer and of the solvent. This configurational contribution exercises a dominating influence on the thermodynamical properties of polymer-solvent systems. The rubber–benzene system has been most thoroughly investigated experimentally. The theory reproduces the observed behaviour of the free energy, and also the general trend of the entropy at ordinary concentrations (see ref. 1, sections 4.6, 4.9, 4.10 and 6.11) ; but in very dilute solution, the experimental measurements show a feature which is not reproduced by the theory. This is made clear by the theoretical (a) and experimental (b) curves shown by the full lines in the diagram. In this note, the effect responsible for the dip in the experimental curve in dilute solution is determined ; its effect is shown by the dotted curve in the diagram. From this it can be seen that the new theoretical curve reproduces the shape of the experimental curve throughout the whole concentration-range.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Miller, A. R., "The Theory of Solutions of High Polymers", Ch. 4 (Clarendon Press, Oxford, 1948).
Miller, A. R., Proc. Camb. Phil. Soc., 39, 54 (1943).
Schallamach, A., Trans. Farad. Soc., 42A, 180 (1946). Gee, G., and Orr, W. J. C., Trans. Farad. Soc., 42, 507 (1946).
Miller, A. R., Proc. Camb. Phil. Soc., 43, 422 (1947).
Guggenheim, E. A., Proc. Roy. Soc., A, 183, 203 (1944).
Miller, A. R., Aust. J. Sci. Res., A, 1, 319 (1948).
Gee, G., J. Chim. Phys., 44, 66 (1947).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MILLER, A. Entropy of Dilution of a Polymer-Solvent System. Nature 163, 838–839 (1949). https://doi.org/10.1038/163838a0
Issue Date:
DOI: https://doi.org/10.1038/163838a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.