Abstract
Background/Objectives:
Growing evidence indicates that trans-fatty acids (TFA) adversely affect cardiovascular health. As part of the World Health Organization (WHO) Scientific Update on TFA, we reviewed the evidence for effects of TFA consumption on coronary heart disease (CHD).
Subjects/Methods:
We searched Medline publications examining TFA consumption and CHD risk factors or outcomes, emphasizing results of studies in humans. We evaluated and synthesized evidence from both controlled feeding trials evaluating risk factors and long-term observational studies evaluating risk factors or clinical outcomes, each of which have complementary strengths and limitations, to enable the most robust and reliable inferences of effects.
Results:
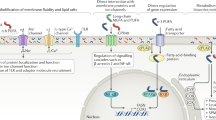
The effects of TFA consumption on risk factors most consistently seen in both controlled trials and observational studies included adverse lipid effects (for example ↑low-density lipoprotein cholesterol, ↓high-density lipoprotein cholesterol (HDL-C), ↑total/HDL-C ratio), proinflammatory effects (for example ↑tumor necrosis factor-α activity, ↑interleukin-6, ↑C-reactive protein) and endothelial dysfunction. These effects were most prominent in comparison with cis unsaturated fats; adverse effects on total/HDL-C and endothelial function were also seen in comparison with saturated fatty acids (SFA). TFA may also worsen insulin sensitivity, particularly among individuals predisposed to insulin resistance; possible effects on weight gain and diabetes incidence require further confirmation. Five retrospective case–control studies and four prospective cohort studies demonstrated positive associations between TFA consumption and CHD events. A meta-analysis of prospective studies indicated 24, 20, 27 and 32% higher risk of myocardial infarction (MI) or CHD death for every 2% energy of TFA consumption isocalorically replacing carbohydrate, SFA, cis monounsaturated fatty acids and cis polyunsaturated fatty acids, respectively. The differential effects of specific TFA isomers may be important but are less well established. The available evidence indicates that trans-18:1 and particularly trans-18:2 isomers have stronger CHD effects than trans-16:1 isomers. The limited data suggest that the experimental effects of ruminant and industrial TFA are similar when consumed in similar quantities, but very few persons consume such high levels of ruminant TFA, and observational studies do not support adverse CHD effects of ruminant TFA in amounts actually consumed.
Conclusions:
Controlled trials and observational studies provide concordant evidence that consumption of TFA from partially hydrogenated oils adversely affects multiple cardiovascular risk factors and contributes significantly to increased risk of CHD events. The public health implications of ruminant TFA consumption appear much more limited. The effects of specific TFA isomers require further investigation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Abbey M, Noakes M, Belling GB, Nestel PJ (1994). Partial replacement of saturated fatty acids with almonds or walnuts lowers total plasma cholesterol and low-density-lipoprotein cholesterol. Am J Clin Nutr 59, 995–999.
Akesson B, Johansson BM, Svensson M, Ockerman PA (1981). Content of trans-octadecenoic acid in vegetarian and normal diets in Sweden, analyzed by the duplicate portion technique. Am J Clin Nutr 34, 2517–2520.
Allison DB, Egan SK, Barraj LM, Caughman C, Infante M, Heimbach JT (1999). Estimated intakes of trans fatty and other fatty acids in the US population. J Am Diet Assoc 99, 166–174.
Almendingen K, Jordal O, Kierulf P, Sandstad B, Pedersen JI (1995). Effects of partially hydrogenated fish oil, partially hydrogenated soybean oil, and butter on serum lipoproteins and Lp(a) in men. J Lipid Res 36, 1370–1384.
Almendingen K, Seljeflot I, Sandstad B, Pedersen JI (1996). Effects of partially hydrogenated fish oil, partially hydrogenated soybean oil, and butter on hemostatic variables in men. Arterioscler Thromb Vasc Biol 16, 375–380.
Armstrong RA, Chardigny JM, Beaufrere B, Bretillon L, Vermunt SH, Mensink RP et al. (2000). No effect of dietary trans isomers of alpha-linolenic acid on platelet aggregation and haemostatic factors in European healthy men. The TRANSLinE study. Thromb Res 100, 133–141.
Aro A, Kardinaal AF, Salminen I, Kark JD, Riemersma RA, Delgado-Rodriguez M et al. (1995). Adipose tissue isomeric trans fatty acids and risk of myocardial infarction in nine countries: the EURAMIC study. Lancet 345, 273–278.
Aro A, Jauhiainen M, Partanen R, Salminen I, Mutanen M (1997). Stearic acid, trans fatty acids, and dairy fat: effects on serum and lipoprotein lipids, apolipoproteins, lipoprotein (a), and lipid transfer proteins in healthy subjects. Am J Clin Nutr 65, 1419–1426.
Ascherio A, Hennekens CH, Buring JE, Master C, Stampfer MJ, Willett WC (1994). Trans fatty acids intake and risk of myocardial infarction. Circulation 89, 94–101.
Ascherio A, Rimm EB, Giovannucci EL, Spiegelman D, Stampfer MJ, Willett WC (1996). Dietary fat and risk of coronary heart disease in men: cohort follow-up study in the United States. BMJ 313, 84–90.
Axen KV, Dikeakos A, Sclafani A (2003). High dietary fat promotes syndrome X in nonobese rats. J Nutr 133, 2244–2249.
Baer DJ, Judd JT, Clevidence BA, Tracy RP (2004). Dietary fatty acids affect plasma markers of inflammation in healthy men fed controlled diets: a randomized crossover study. Am J Clin Nutr 79, 969–973.
Baylin A, Kabagambe EK, Ascherio A, Spiegelman D, Campos H (2003). Adipose tissue alpha-linolenic acid and nonfatal acute myocardial infarction in Costa Rica. Circulation 107, 1586–1591.
Chardigny JM, Destaillats F, Malpuech-Brugère C, Moulin J, Bauman DE, Lock AL et al. (2008). Do industrially-produced and natural trans fatty acid sources have the same impact on cardiovascular diseases risk factors in healthy subjects? Results of the TRANSFACT Study. Am J Clin Nutr 87, 558–566.
Chavarro JE, Rich-Edwards JW, Rosner BA, Willett WC (2007). Dietary fatty acid intakes and the risk of ovulatory infertility. Am J Clin Nutr 85, 231–237.
Christiansen E, Schnider S, Palmvig B, Tauber-Lassen E, Pedersen O (1997). Intake of a diet high in trans monounsaturated fatty acids or saturated fatty acids. Effects on postprandial insulinemia and glycemia in obese patients with NIDDM. Diabetes Care 20, 881–887.
Clevidence BA, Judd JT, Schaefer EJ, Jenner JL, Lichtenstein AH, Muesing RA et al. (1997). Plasma lipoprotein (a) levels in men and women consuming diets enriched in saturated, cis-, or trans-monounsaturated fatty acids. Arterioscler Thromb Vasc Biol 17, 1657–1661.
Clifton PM, Keogh JB, Noakes M (2004). Trans fatty acids in adipose tissue and the food supply are associated with myocardial infarction. J Nutr 134, 874–879.
Colon-Ramos U, Baylin A, Campos H (2006). The relation between trans fatty acid levels and increased risk of myocardial infarction does not hold at lower levels of trans fatty acids in the Costa Rican food supply. J Nutr 136, 2887–2892.
Cuchel M, Schwab US, Jones PJ, Vogel S, Lammi-Keefe C, Li Z et al. (1996). Impact of hydrogenated fat consumption on endogenous cholesterol synthesis and susceptibility of low-density lipoprotein to oxidation in moderately hypercholesterolemic individuals. Metabolism 45, 241–247.
de Roos NM, Bots ML, Katan MB (2001). Replacement of dietary saturated fatty acids by trans fatty acids lowers serum HDL cholesterol and impairs endothelial function in healthy men and women. Arterioscler Thromb Vasc Biol 21, 1233–1237.
de Roos NM, Siebelink E, Bots ML, van Tol A, Schouten EG, Katan MB (2002). Trans monounsaturated fatty acids and saturated fatty acids have similar effects on postprandial flow-mediated vasodilation. Eur J Clin Nutr 56, 674–679.
Doney AS, Fischer B, Leese G, Morris AD, Palmer CN (2004). Cardiovascular risk in type 2 diabetes is associated with variation at the PPARG locus: a Go-DARTS study. Arterioscler Thromb Vasc Biol 24, 2403–2407.
European Food Safety Authority (2004). Opinion of the scientific panel on dietetic products, nutrition and allergies on a request from the Commission related to the presence of trans fatty acids in foods and the effect on human health of the consumption of trans fatty acids. EFSA J 81, 1–49.
Field AE, Willett WC, Lissner L, Colditz GA (2007). Dietary fat and weight gain among women in the Nurses’ Health Study. Obesity (Silver Spring) 15, 967–976.
Han SN, Leka LS, Lichtenstein AH, Ausman LM, Schaefer EJ, Meydani SN (2002). Effect of hydrogenated and saturated, relative to polyunsaturated, fat on immune and inflammatory responses of adults with moderate hypercholesterolaemia. J Lipid Res 43, 445–452.
He K, Merchant A, Rimm EB, Rosner BA, Stampfer MJ, Willett WC et al. (2003). Dietary fat intake and risk of stroke in male US healthcare professionals: 14 year prospective cohort study. BMJ 327, 777–782.
Hu FB, Stampfer MJ, Manson JE, Rimm EB, Colditz GA, Rosner BA et al. (1997). Dietary fat intake and the risk of coronary heart disease in women. N Eng J Med 337, 1491–1499.
Hu FB, Manson JE, Stampfer MJ, Colditz G, Liu S, Solomon CG et al. (2001a). Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N Engl J Med 345, 790–797.
Hu FB, Manson JE, Stampfer MJ, Colditz G, Liu S, Solomon CG et al. (2001b). Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N Engl J Med 345, 790–797.
Ibrahim A, Natrajan S, Ghafoorunissa R (2005). Dietary trans-fatty acids alter adipocyte plasma membrane fatty acid composition and insulin sensitivity in rats. Metabolism 54, 240–246.
Jakobsen MU, Overvad K, Dyerberg J, Heitmann BL (2007). Intake of ruminant trans fatty acids and risk of coronary heart disease. Int J Epidemiol 12 December 2007(e-pub ahead of print).
Johansson L, Borgejordet A, Pedersen JI (2006). [Trans fatty acids in the Norwegian diet]. Tidsskr Nor Laegeforen 126, 760–763.
Judd JT, Clevidence BA, Muesing RA, Wittes J, Sunkin ME, Podczasy JJ (1994). Dietary trans fatty acids: effects on plasma lipids and lipoproteins of healthy men and women. Am J Clin Nutr 59, 861–868.
Judd JT, Baer DJ, Clevidence BA, Muesing RA, Chen SC, Weststrate JA et al. (1998). Effects of margarine compared with those of butter on blood lipid profiles related to cardiovascular disease risk factors in normolipemic adults fed controlled diets. Am J Clin Nutr 68, 768–777.
Judd JT, Baer DJ, Clevidence BA, Kris-Etherton P, Muesing RA, Iwane M (2002). Dietary cis and trans monounsaturated and saturated FA and plasma lipids and lipoproteins in men. Lipids 37, 123–131.
Katan MB, Zock PL, Mensink RP (1995). Trans fatty acids and their effects on lipoproteins in humans. Annu Rev Nutr 15, 473–493.
Kavanagh K, Jones KL, Sawyer J, Kelley K, Carr JJ, Wagner JD et al. (2007). Trans fat diet induces abdominal obesity and changes in insulin sensitivity in monkeys. Obesity (Silver Spring) 15, 1675–1684.
Kim SH, Chunawala L, Linde R, Reaven GM (2006). Comparison of the 1997 and 2003 American diabetes association classification of impaired fasting glucose: impact on prevalence of impaired fasting glucose, coronary heart disease risk factors, and coronary heart disease in a community-based medical practice. J Am Coll Cardiol 48, 293–297.
Koh-Banerjee P, Chu NF, Spiegelman D, Rosner B, Colditz G, Willett W et al. (2003). Prospective study of the association of changes in dietary intake, physical activity, alcohol consumption, and smoking with 9-y gain in waist circumference among 16 587 US men. Am J Clin Nutr 78, 719–727.
Kromhout D, Feskens EJ, Bowles CH (1995). The protective effect of a small amount of fish on coronary heart disease mortality in an elderly population. Int J Epidemiol 24, 340–345.
L'Abbé MR, Stender S, Skeaff CM, Ghafoorunissa, Tavella M (2009). Approaches to removing trans fats in the food supply in industrialized and developing countries. Eur J Clin Nutr 63 (Suppl 2), S50–S67.
Lefevre M, Lovejoy JC, Smith SR, Delany JP, Champagne C, Most MM et al. (2005). Comparison of the acute response to meals enriched with cis- or trans-fatty acids on glucose and lipids in overweight individuals with differing FABP2 genotypes. Metabolism 54, 1652–1658.
Lemaitre RN, King IB, Raghunathan TE, Pearce RM, Weinmann S, Knopp RH et al. (2002). Cell membrane trans fatty acids and the risk of primary cardiac arrest. Circulation 105, 697–701.
Lemaitre RN, King IB, Mozaffarian D, Sotoodehnia N, Rea TD, Kuller LH et al. (2006). Plasma phospholipid trans fatty acids, fatal ischemic heart disease, and sudden cardiac death in older adults: the cardiovascular health study. Circulation 114, 209–215.
Lichtenstein AH, Ausman LM, Jalbert SM, Schaefer EJ (1999). Effects of different forms of dietary hydrogenated fats on serum lipoprotein cholesterol levels. N Engl J Med 340, 1933–1940.
Lichtenstein AH, Erkkila AT, Lamarche B, Schwab US, Jalbert SM, Ausman LM (2003). Influence of hydrogenated fat and butter on CVD risk factors: remnant-like particles, glucose and insulin, blood pressure and C-reactive protein. Atherosclerosis 171, 97–107.
Lopez-Garcia E, Schulze MB, Meigs JB, Manson JE, Rifai N, Stampfer MJ et al. (2005). Consumption of trans fatty acids is related to plasma biomarkers of inflammation and endothelial dysfunction. J Nutr 135, 562–566.
Louheranta AM, Turpeinen AK, Vidgren HM, Schwab US, Uusitupa MI (1999). A high-trans fatty acid diet and insulin sensitivity in young healthy women. Metabolism 48, 870–875.
Lovejoy JC, Smith SR, Champagne CM, Most MM, Lefevre M, DeLany JP et al. (2002). Effects of diets enriched in saturated (palmitic), monounsaturated (oleic), or trans (elaidic) fatty acids on insulin sensitivity and substrate oxidation in healthy adults. Diabetes Care 25, 1283–1288.
Mauger JF, Lichtenstein AH, Ausman LM, Jalbert SM, Jauhiainen M, Ehnholm C et al. (2003). Effect of different forms of dietary hydrogenated fats on LDL particle size. Am J Clin Nutr 78, 370–375.
Meijer GW, van Tol A, van Berkel TJ, Weststrate JA (2001). Effect of dietary elaidic versus vaccenic acid on blood and liver lipids in the hamster. Atherosclerosis 157, 31–40.
Mensink RP, Katan MB (1990). Effect of dietary trans fatty acids on high-density and low-density lipoprotein cholesterol levels in healthy subjects. N Engl J Med 323, 439–445.
Mensink RP, Zock PL, Katan MB, Hornstra G (1992). Effect of dietary cis- and trans fatty acids on serum lipoprotein (a) levels in humans. J Lipid Res 33, 1493–1501.
Mensink RP, Zock PL, Kester AD, Katan MB (2003). Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials. Am J Clin Nutr 77, 1146–1155.
Meyer KA, Kushi LH, Jacobs Jr DR, Folsom AR (2001). Dietary fat and incidence of type 2 diabetes in older Iowa women. Diabetes Care 24, 1528–1535.
Moloney F, Yeow TP, Mullen A, Nolan JJ, Roche HM (2004). Conjugated linoleic acid supplementation, insulin sensitivity, and lipoprotein metabolism in patients with type 2 diabetes mellitus. Am J Clin Nutr 80, 887–895.
Mozaffarian D, Pischon T, Hankinson SE, Rifai N, Joshipura K, Willett WC et al. (2004a). Dietary intake of trans fatty acids and systemic inflammation in women. Am J Clin Nutr 79, 606–612.
Mozaffarian D, Rimm EB, King IB, Lawler RL, McDonald GB, Levy WC (2004b). Trans fatty acids and systemic inflammation in heart failure. Am J Clin Nutr 80, 1521–1525.
Mozaffarian D, Katan MB, Ascherio A, Stampfer MJ, Willett WC (2006). Trans fatty acids and cardiovascular disease. N Engl J Med 354, 1601–1613.
Mozaffarian D, Willett WC (2007). Trans fatty acids and cardiovascular risk: a unique cardiometabolic imprint? Curr Atheroscler Rep 9, 486–493.
Mozaffarian D, Clarke R (2009). Quantitative effects on cardiovascular risk factors and coronary heart disease risk of replacing partially hydrogenated vegetable oils with other fats and oils. Eur J Clin Nutr 63 (Suppl 2), S22–S33.
Muller H, Jordal O, Kierulf P, Kirkhus B, Pedersen JI (1998a). Replacement of partially hydrogenated soybean oil by palm oil in margarine without unfavorable effects on serum lipoproteins. Lipids 33, 879–887.
Muller H, Jordal O, Seljeflot I, Kierulf P, Kirkhus B, Ledsaak O et al. (1998b). Effect on plasma lipids and lipoproteins of replacing partially hydrogenated fish oil with vegetable fat in margarine. Br J Nutr 80, 243–251.
Mutanen M, Aro A (1997). Coagulation and fibrinolysis factors in healthy subjects consuming high stearic or trans fatty acid diets. Thromb Haemost 77, 99–104.
Natarajan S, Ibrahim A, Ghafoorunissa (2005). Dietary trans fatty acids alter diaphragm phospholipid fatty acid composition, triacylglycerol content and glucose transport in rats. Br J Nutr 93, 829–833.
Neubauer S, Welte R, Beiche A, Koenig HH, Buesch K, Leidl R (2006). Mortality, morbidity and costs attributable to smoking in Germany: update and a 10-year comparison. Tob Control 15, 464–471.
Oh K, Hu FB, Manson JE, Stampfer MJ, Willett WC (2005). Dietary fat intake and risk of coronary heart disease in women: 20 years of follow-up of the nurses’ health study. Am J Epidemiol 161, 672–679.
Oomen CM, Ocke MC, Feskens EJ, van Erp-Baart MA, Kok FJ, Kromhout D (2001). Association between trans fatty acid intake and 10-year risk of coronary heart disease in the Zutphen Elderly Study: a prospective population-based study. Lancet 357, 746–751.
Pietinen P, Ascherio A, Korhonen P, Hartman AM, Willett WC, Albanes D et al. (1997). Intake of fatty acids and risk of coronary heart disease in a cohort of Finnish men. The Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study. Am J Epidemiol 145, 876–887.
Riserus U, Basu S, Jovinge S, Fredrikson GN, Arnlov J, Vessby B (2002). Supplementation with conjugated linoleic acid causes isomer-dependent oxidative stress and elevated C-reactive protein: a potential link to fatty acid-induced insulin resistance. Circulation 106, 1925–1929.
Riserus U, Vessby B, Arnlov J, Basu S (2004). Effects of cis-9, trans-11 conjugated linoleic acid supplementation on insulin sensitivity, lipid peroxidation, and proinflammatory markers in obese men. Am J Clin Nutr 80, 279–283.
Roberts TL, Wood DA, Riemersma RA, Gallagher PJ, Lampe FC (1995). Trans isomers of oleic and linoleic acids in adipose tissue and sudden cardiac death. Lancet 345, 278–282.
Salmeron J, Hu FB, Manson JE, Stampfer MJ, Colditz GA, Rimm EB et al. (2001). Dietary fat intake and risk of type 2 diabetes in women. Am J Clin Nutr 73, 1019–1026.
Sun Q, Ma J, Campos H, Hankinson SE, Manson JE, Stampfer MJ et al. (2007). A prospective study of trans fatty acids in erythrocytes and risk of coronary heart disease. Circulation 115, 1858–1865.
Tholstrup T, Sandstrom B, Hermansen JE, Holmer G (1998). Effect of modified dairy fat on postprandial and fasting plasma lipids and lipoproteins in healthy young men. Lipids 33, 11–21.
Tholstrup T, Raff M, Basu S, Nonboe P, Sejrsen K, Straarup EM (2006). Effects of butter high in ruminant trans and monounsaturated fatty acids on lipoproteins, incorporation of fatty acids into lipid classes, plasma C-reactive protein, oxidative stress, hemostatic variables, and insulin in healthy young men. Am J Clin Nutr 83, 237–243.
Thomas LH, Winter JA, Scott RG (1983a). Concentration of 18:1 and 16:1 transunsaturated fatty acids in the adipose body tissue of decedents dying of ischaemic heart disease compared with controls: analysis by gas liquid chromatography. J Epidemiol Community Health 37, 16–21.
Thomas LH, Winter JA, Scott RG (1983b). Concentration of transunsaturated fatty acids in the adipose body tissue of decedents dying of ischaemic heart disease compared with controls. J Epidemiol Community Health 37, 22–24.
Tricon S, Burdge GC, Jones EL, Russell JJ, El-Khazen S, Moretti E et al. (2006). Effects of dairy products naturally enriched with cis-9, trans-11 conjugated linoleic acid on the blood lipid profile in healthy middle-aged men. Am J Clin Nutr 83, 744–753.
Tsai CJ, Leitzmann MF, Willett WC, Giovannucci EL (2005). Long-term intake of trans-fatty acids and risk of gallstone disease in men. Arch Intern Med 165, 1011–1015.
Valenzuela A, Uauy R (1999). Consumption pattern of dietary fats in Chile: n−6 and n−3 fatty acids. Int J Food Sci Nutr 50, 127–133.
van Dam RM, Rimm EB, Willett WC, Stampfer MJ, Hu FB (2002). Dietary patterns and risk for type 2 diabetes mellitus in US men. Ann Intern Med 136, 201–209.
van Tol A, Zock PL, van Gent T, Scheek LM, Katan MB (1995). Dietary trans fatty acids increase serum cholesterylester transfer protein activity in man. Atherosclerosis 115, 129–134.
Vega-Lopez S, Ausman LM, Jalbert SM, Erkkila AT, Lichtenstein AH (2006). Palm and partially hydrogenated soybean oils adversely alter lipoprotein profiles compared with soybean and canola oils in moderately hyperlipidemic subjects. Am J Clin Nutr 84, 54–62.
Vermunt SH, Beaufrere B, Riemersma RA, Sebedio JL, Chardigny JM, Mensink RP et al. (2001). Dietary trans alpha-linolenic acid from deodorised rapeseed oil and plasma lipids and lipoproteins in healthy men: the TRANSLinE Study. Br J Nutr 85, 387–392.
Willett WC, Stampfer MJ, Manson JE, Colditz GA, Speizer FE, Rosner BA et al. (1993). Intake of trans fatty acids and risk of coronary heart disease among women. Lancet 341, 581–585.
Willett WC, Leibel RL (2002). Dietary fat is not a major determinant of body fat. Am J Med 113 (Suppl 9B), S47–S59.
World Cancer Research Fund (2007). Food, Nutrition, Physical Activity, and the Prevention of Cancer: a Global Perspective. American Institute for Cancer Research: Washington DC.
Zapolska-Downar D, Kosmider A, Naruszewicz M (2005). Trans fatty acids induce apoptosis in human endothelial cells. J Physiol Pharmacol 56, 611–625.
Zock PL, Katan MB (1992). Hydrogenation alternatives: effects of trans fatty acids and stearic acid versus linoleic acid on serum lipids and lipoproteins in humans. J Lipid Res 33, 399–410.
Acknowledgements
We thank Dr Robert Clarke, Dr Richard Deckelbaum, Dr Ghafoorunissa, Dr Martijn B Katan, Professor Jim Mann, Dr Ulf Risérus, Dr Thomas Sanders, Professor Ricardo Uauy and Dr Bengt Vessby for their helpful comments during the preparation of this paper. We also acknowledge the helpful comments received from Panel on Dietetic Products, Nutrition and Allergies (NDA), European Food Safety Authority (EFSA), in particular, Dr Silvia Valtueña Martinez, Dr Pilar Rodríguez Iglesias, Dr Wolfgang Gelbman and Dr Leng Heng. Dr Mozaffarian was supported by a grant from the National Heart, Lung, and Blood Institute, National Institutes of Health (K08-HL-075628). We thank Jemma Hopewell and Paul Sherliker for their assistance in statistical analyses and figure production.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mozaffarian, D., Aro, A. & Willett, W. Health effects of trans-fatty acids: experimental and observational evidence. Eur J Clin Nutr 63 (Suppl 2), S5–S21 (2009). https://doi.org/10.1038/sj.ejcn.1602973
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejcn.1602973
Keywords
This article is cited by
-
Positive associations between different circulating trans fatty acids (TFAs) and urinary albumin excretion among adults in the U.S.: a population-based study
Lipids in Health and Disease (2023)
-
Trans-fatty acid blood levels of industrial but not natural origin are associated with cardiovascular risk factors in patients with HFpEF: a secondary analysis of the Aldo-DHF trial
Clinical Research in Cardiology (2023)
-
Lifestyle Medicine as a Treatment for Resistant Hypertension
Current Hypertension Reports (2023)
-
Molecular effects of the consumption of margarine and butter varying in trans fat composition: a parallel human intervention study
Lipids in Health and Disease (2022)
-
Social marketing-based interventions to promote healthy nutrition behaviors: a systematic review protocol
Systematic Reviews (2021)