Abstract
Objective:
To compare fracture rates in four diet groups (meat eaters, fish eaters, vegetarians and vegans) in the Oxford cohort of the European Prospective Investigation into Cancer and Nutrition (EPIC-Oxford).
Design:
Prospective cohort study of self-reported fracture risk at follow-up.
Setting:
The United Kingdom.
Subjects:
A total of 7947 men and 26 749 women aged 20–89 years, including 19 249 meat eaters, 4901 fish eaters, 9420 vegetarians and 1126 vegans, recruited by postal methods and through general practice surgeries.
Methods:
Cox regression.
Results:
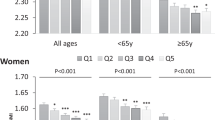
Over an average of 5.2 years of follow-up, 343 men and 1555 women reported one or more fractures. Compared with meat eaters, fracture incidence rate ratios in men and women combined adjusted for sex, age and non-dietary factors were 1.01 (95% CI 0.88–1.17) for fish eaters, 1.00 (0.89–1.13) for vegetarians and 1.30 (1.02–1.66) for vegans. After further adjustment for dietary energy and calcium intake the incidence rate ratio among vegans compared with meat eaters was 1.15 (0.89–1.49). Among subjects consuming at least 525 mg/day calcium the corresponding incidence rate ratios were 1.05 (0.90–1.21) for fish eaters, 1.02 (0.90–1.15) for vegetarians and 1.00 (0.69–1.44) for vegans.
Conclusions:
In this population, fracture risk was similar for meat eaters, fish eaters and vegetarians. The higher fracture risk in the vegans appeared to be a consequence of their considerably lower mean calcium intake. An adequate calcium intake is essential for bone health, irrespective of dietary preferences.
Sponsorship:
The EPIC-Oxford study is supported by The Medical Research Council and Cancer Research UK.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Allen NE, Appleby PN, Davey GK, Kaaks R, Rinaldi S, Key TJ (2002). The associations of diet with serum insulin-like growth factor I and its main binding proteins in 292 women meat-eaters, vegetarians, and vegans. Cancer Epidemiol Biomarkers Prev 11, 1441–1448.
Allen NE, Appleby PN, Davey GK, Key TJ (2000). Hormones and diet: low insulin-like growth factor-I but normal bioavailable androgens in vegan men. Br J Cancer 83, 95–97.
American Dietetic Association; Dietitians of Canada (2003). Position of the American Dietetic Association and Dietitians of Canada: Vegetarian diets. J Am Diet Assoc 103, 748–765.
Barr SI, Prior JC, Janelle KC, Lentle BC (1998). Spinal bone mineral density in premenopausal vegetarian and nonvegetarian women: cross-sectional and prospective comparisons. J Am Diet Assoc 98, 760–765.
Bingham SA, Gill C, Welch A, Day K, Cassidy A, Khaw KT et al. (1994). Comparison of dietary assessment methods in nutritional epidemiology: weighed records v. 24 h recalls, food-frequency questionnaires and estimated-diet records. Br J Nutr 72, 619–643.
Chen Z, Kooperberg C, Pettinger MB, Bassford T, Cauley JA, LaCroix AZ et al. (2004). Validity of self-report for fractures among a multiethnic cohort of postmenopausal women: results from the women's health initiative observational study and clinical trials. Menopause 11, 264–274.
Chiu JF, Lan SJ, Yang CY, Wang PW, Yao WJ, Su LH et al. (1997). Long-term vegetarian diet and bone mineral density in postmenopausal Taiwanese women. Calcif Tissue Int 60, 245–249.
Davey GK, Spencer EA, Appleby PN, Allen NE, Knox KH, Key TJ (2003). EPIC-Oxford: lifestyle characteristics and nutrient intakes in a cohort of 33 883 meat-eaters and 31 546 non meat-eaters in the UK. Public Health Nutr 6, 259–269.
Dawson-Hughes B (2003). Calcium and protein in bone health. Proc Nutr Soc 62, 505–509.
Department of Health (1991). Dietary Reference Values for Food Energy and Nutrients for the United Kingdom. Report on Health and Social Subjects: 41. HMSO: London.
Fontana L, Shew JL, Holloszy JO, Villareal DT (2005). Low bone mass in subjects on a long-term raw vegetarian diet. Arch Intern Med 165, 684–689.
Ginty F (2003). Dietary protein and bone health. Proc Nutr Soc 62, 867–876.
Holland B, Welch AA, Unwin ID, Buss DH, Paul AA, Southgate DAT (1991). McCance & Widdowson's The Composition of Foods. Royal Society of Chemistry: Cambridge, UK.
Institute of Medicine (1997). Dietary reference intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. National Academy Press: Washington, DC.
Key TJ, Appleby PN, Spencer EA, Roddam AW, Neale RE, Allen NE (2007). Calcium, diet and fracture risk: a prospective study of 1898 incident fractures among 34 696 British men and women. Public Health Nutr (in press).
Kohlenberg-Mueller K, Raschka L (2003). Calcium balance in young adults on a vegan and lactovegetarian diet. J Bone Miner Metab 21, 28–33.
Larsson CL, Johansson GK (2002). Dietary intake and nutritional status of young vegans and omnivores in Sweden. Am J Clin Nutr 76, 100–106.
Lau EM, Kwok T, Woo J, Ho SC (1998). Bone mineral density in Chinese elderly female vegetarians, vegans, lacto-vegetarians and omnivores. Eur J Clin Nutr 52, 60–64.
Lloyd T, Schaeffer JM, Walker MA, Demers LM (1991). Urinary hormonal concentrations and spinal bone densities of premenopausal vegetarian and nonvegetarian women. Am J Clin Nutr 54, 1005–1010.
Marsh AG, Sanchez TV, Michelsen O, Chaffee FL, Fagal SM (1988). Vegetarian lifestyle and bone mineral density. Am J Clin Nutr 48, 837–841.
New SA (2004). Do vegetarians have a normal bone mass? Osteoporos Int 15, 679–688.
Outila TA, Karkkainen MU, Seppanen RH, Lamberg-Allardt CJ (2000). Dietary intake of vitamin D in premenopausal, healthy vegans was insufficient to maintain concentrations of serum 25-hydroxyvitamin D and intact parathyroid hormone within normal ranges during the winter in Finland. J Am Diet Assoc 100, 434–441.
Parsons TJ, van Dusseldorp M, van der Vliet M, van de Werken K, Schaafsma G, van Staveren WA (1997). Reduced bone mass in Dutch adolescents fed a macrobiotic diet in early life. J Bone Miner Res 12, 1486–1494.
Prentice A (2004). Diet, nutrition and the prevention of osteoporosis. Public Health Nutr 7, 227–243.
Reed JA, Anderson JJ, Tylavsky FA, Gallagher Jr PN (1994). Comparative changes in radial-bone density of elderly female lacto-ovovegetarians and omnivores. Am J Clin Nutr 59, 1197S–1202S.
Siani V, Mohamed EI, Maiolo C, Di Daniele N, Ratiu A, Leonardi A et al. (2003). Body composition analysis for healthy Italian vegetarians. Acta Diabetol 40, S297–S298.
Smith AM (2006). Veganism and osteoporosis: a review of the current literature. Int J Nurs Pract 12, 302–306.
Stata Corporation (2005). Stata Statistical Software: Release 9.0. Stata Corporation. College Station: TX.
Tesar R, Notelovitz M, Shim E, Kauwell G, Brown J (1992). Axial and peripheral bone density and nutrient intakes of postmenopausal vegetarian and omnivorous women. Am J Clin Nutr 56, 699–704.
Waldmann A, Koschizke JW, Leitzmann C, Hahn A (2003). Dietary intakes and lifestyle factors of a vegan population in Germany: results from the German Vegan Study. Eur J Clin Nutr 57, 947–955.
Acknowledgements
The EPIC-Oxford study is supported by The Medical Research Council and Cancer Research UK. We thank the participants in EPIC-Oxford.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guarantor: P Appleby.
Contributors: PA performed the statistical analyses and wrote the manuscript. AR and NA contributed to the design of the study, interpretation of the results and to the writing of the manuscript. TK is the principal investigator of the EPIC-Oxford study and contributed to the manuscript writing.
Rights and permissions
About this article
Cite this article
Appleby, P., Roddam, A., Allen, N. et al. Comparative fracture risk in vegetarians and nonvegetarians in EPIC-Oxford. Eur J Clin Nutr 61, 1400–1406 (2007). https://doi.org/10.1038/sj.ejcn.1602659
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejcn.1602659
Keywords
This article is cited by
-
Plant-based diets and sports performance: a clinical review
Sport Sciences for Health (2023)
-
Plant-Based Diet: Is It as Good as an Animal-Based Diet When It Comes to Protein?
Current Nutrition Reports (2022)
-
Vegetarian and vegan diets and risks of total and site-specific fractures: results from the prospective EPIC-Oxford study
BMC Medicine (2020)
-
Sicherheit und Risiken vegetarischer und veganer Ernährung in Schwangerschaft, Stillzeit und den ersten Lebensjahren
Monatsschrift Kinderheilkunde (2019)
-
Bone turnover, calcium homeostasis, and vitamin D status in Danish vegans
European Journal of Clinical Nutrition (2018)