Abstract
Background:
Weight control programs for obese children monitor change in body mass index (BMI) adjusted for age. However, change can be measured in several ways: raw (kg/m2) units, percentage, z-scores or centiles. The suitability of the different measures is not known.
Aim:
To identify the optimal BMI measure for change, whose short-term variability is most consistent for children across the spectrum of adiposity.
Setting:
An Italian kindergarten.
Subjects:
A total of 135 (66 female) children aged 29–68 months at baseline, with BMI measured three times over a 9-month period.
Methods:
Each child's short-term variability in adiposity was summarized by the standard deviation (s.d.) of BMI and BMI % adjusted for age, and BMI z-score and BMI centile. The s.d.'s were then compared in obese and nonobese children, and also correlated with each child's baseline BMI z-score.
Results:
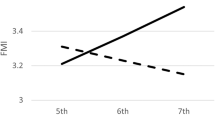
The within-child s.d.s of BMI z-score and BMI centile were significantly smaller in obese than nonobese children, while the s.d.s of BMI and BMI % were similar in the two groups. Also, the within-child s.d.s of z-score and centile, and to a lesser extent BMI %, were significantly inversely correlated with baseline z-score, whereas the s.d. of BMI was not. The changes in adiposity over time, as assessed by the four measures, were very highly correlated with each other, particularly for BMI with BMI %.
Discussion:
Even though BMI z-score is optimal for assessing adiposity on a single occasion, it is not necessarily the best scale for measuring change in adiposity, as the within-child variability over time depends on the child's level of adiposity. Better alternatives are BMI itself or BMI %. Our results underscore the importance of using a relatively stable method to assess adiposity change when following children at risk of obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Barlow SE, Trowbridge FL, Klish WS & Dietz WH (2002): Treatment of child and adolescent obesity: reports from pediatricians, pediatric nurse practitioners and registered dietitians. Pediatrics 110, 229–235.
Cole TJ (1997): Growth monitoring with the British 1990 growth reference. Arch Dis. Child 76, 47–49.
Cole TJ (2000): Sympercents: symmetric percentage differences on the 100 loge scale simplify the presentation of log transformed data. Stat. Med. 19, 3109–3125.
Ellis KJ (2000): Human body composition. In vivo methods. Physiol. Rev. 80, 649–680.
Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, Flegal KM, Guo SS, Wei R, Mei Z, Curtin LR, Roche AF & Johnson CL (2000): CDC Growth Charts: United States. Hyattsville, MD: National Center for Health Statistics.
Maynard LM, Wisemandle W, Roche AF, Chumlea WC, Guo SS & Siervogel RM (2001): Childhood body composition in relation to body mass index. Pediatrics 107, 344–350.
Pietrobelli A, Faith MS, Allison DB, Gallagher D, Chiumello G & Heymsfield SB (1998): Body mass index as a measure of adiposity among children and adolescents: a validation study. J. Pediatr. 132, 204–210.
Power C, Lake JK & Cole TJ (1997): Measurement and long term health risks of child and adolescent fatness. Int. J. Obes. 21, 507–526.
Zannolli R & Morgese G (1996): Distribution of BMI in children: prevalence of wasting and fattening conditions. Ann. Hum. Biol. 23, 63–69.
Zemel BS, Riley EM & Stallings VA (1997): Evaluation of methodology for nutritional assessment in children: anthropometry, body composition, and energy expenditure. Annu. Rev. Nutr. 17, 211–235.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guarantor: A Pietrobelli.
Contributors: AP designed the study and collected the data. MSF, MH, AP and TJC analysed the data, wrote the manuscript and gave final approval of the manuscript.
Rights and permissions
About this article
Cite this article
Cole, T., Faith, M., Pietrobelli, A. et al. What is the best measure of adiposity change in growing children: BMI, BMI %, BMI z-score or BMI centile?. Eur J Clin Nutr 59, 419–425 (2005). https://doi.org/10.1038/sj.ejcn.1602090
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejcn.1602090
Keywords
This article is cited by
-
A long-term follow-up of treatment for young children with obesity: a randomized controlled trial
International Journal of Obesity (2023)
-
Longitudinal association between density of retail food stores and body mass index in Mexican school children and adolescents
International Journal of Obesity (2023)
-
Early vascular ageing phenotypes and urinary targeted metabolomics in children and young adults: the ExAMIN Youth SA and African-PREDICT studies
Amino Acids (2023)
-
Multiple beneficial effects of 1-year nutritional–behavioral intervention on anthropometric and metabolic parameters in overweight and obese boys
Journal of Endocrinological Investigation (2023)
-
Face-to-face physical activity incorporated into dietary intervention for overweight/obesity in children and adolescents: a Bayesian network meta-analysis
BMC Medicine (2022)