Abstract
Objective: To describe the influence of maternal weight and weight gain, placental volume and the rate of placental growth in early pregnancy on fetal dimensions measured sonographically.
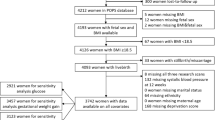
Design: In a prospective study, 712 women were recruited from the antenatal clinic of the University Hospital of the West Indies. Data analysis was confined to 374 women on whom measurements of the placental volume at 14, 17 and 20 weeks gestation were complete. Measurements of maternal anthropometry and fetal size (by ultrasound) were performed. Weight gain in pregnancy between the first antenatal visit (8–10 weeks) and 20 weeks gestation, and the rate of growth of the placenta between 14–17 and 17–20 weeks gestation were calculated.
Main outcome measures: Fetal anthropometry (abdominal and head circumferences, femoral length, and biparietal diameter) at 35 weeks gestation.
Results: Lower maternal weight at the first antenatal visit was associated with a significantly smaller placental volume at 17 and 20 weeks gestation (P<0.002 and <0.0001 respectively). In all women, maternal weight gain was directly related to fetal anthropometry. Placental volume at 14 weeks gestation and the rate of growth of the placenta between 17 and 20 weeks gestation were significantly related to all four fetal measurements.
Conclusion: This study has provided evidence that both placental volume, and the rate of placental growth may influence fetal size. These effects are evident in the first half of pregnancy, and appear to be mediated through maternal weight and weight gain.
Sponsorship: This study was supported by a grant from the Wellcome Trust, 183 Euston Road, London, England.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Abrams B & Selvin S (1995): Maternal weight gain pattern and birth weight. Obstet. Gynecol. 86, 63–69.
Andersson SW, Niklasson A, Lapidus L, Hallberg L, Bengtsson C & Hulthen L (2000): Sociodemographic characteristics influencing birth outcome in Sweden, 1908–1930. Birth variables in the population study of women in Guthenburg. J. Epidemiol. Community Health 54, 269–278.
Ananth CV, Peedicayil A & Savitz DA (1995): Effect of hypertensive disease in pregnancy on birthweight, gestational duration and small-for-gestational age births. Epidemiology 6, 391–395.
Anthony RV, Pratt Sl, Liang R & Holland MD (1995): Placental–fetal hormonal interactions: impact on fetal growth. J. Anim. Sci. 73, 1861–1871.
Baker J, Lui JP, Robertson EJ & Efstratiadis A (1993): Role of insulin-like growth factors in embryonic and postnatal growth. Cell 75, 73–82.
Barker DJP (1997): The fetal origins of coronary heart disease. Acta Paediatr. 422(Suppl.), S78–S82.
Barker DJP, Godfrey KM, Osmond C & Bull A (1992): The relationship of fetal length, ponderal index and head circumference to blood pressure and risk of hypertension in adult life. Paediatr. Perinat. Epidemiol. 6, 35–44.
Barker DJP, Hales CN, Fall CHD, Osmond C, Phipps K & Clark PMS (1993): Type 2 (non-insulin-dependent) diabetes mellitus, hypertension and hyperlipidaemia (syndrome X): relation to reduced fetal growth. Diabetologia. 36, 62–67.
Clapp III JF, Rizk KH, Appleby-Wineberg SK & Crass JR (1995): Second-trimester placental volumes predicts birth weight at term. J. Soc. Gynecol. Invest. 2, 19–22.
Clapp III JF, Kim H, Burciu B & Lopez B (2000): Beginning regular exercise in early pregnancy: effect on fetoplacental growth. Am. J. Obstet. Gynecol. 183, 1484–1488.
Clapp III JF, Kim H, Burciu B, Schmidt S, Petry K & Lopez B (2002): Continuing regular exercise during pregnancy: effect of exercise volume on fetoplacental growth. Am. J. Obstet. Gynecol. 186, 142–147.
Elford J, Whincup P & Shaper AG (1991): Early life experiences and cardiovascular disease: longitudinal and case–control studies. Int. J. Epidemiol. 20, 833–844.
England LJ, Kendrick JS, Wilson HG, Merritt RK, Gargiullo PM & Zahniser SC (2001): Effects of smoking reduction during pregnancy on the birth weight of term infants. Am. J. Epidemiol. 154, 694–701.
Ericson A, Eriksson M, Kallen B & Zetterstrom R (1989): Socio-economic variables and pregnancy outcome. Birthweight in singletons. Acta Paediatr. Scand. 360(Suppl.), S48–S55.
Forrester TE, Wilks RJ, Bennett FI, Simeon D, Osmond C, Allen M, Chung AP & Scott P (1996): Fetal growth and cardiovascular risk factors in Jamaican schoolchildren. BMJ 312, 156–160.
Gluckman PD, Breier BH, Oliver M, Harding J & Bassett N (1990): Fetal growth in late gestation—a constrained pattern of growth. Acta Paediatr. Scand. 367, 105–110.
Hay WW (1991): The placenta. Not just a conduit for maternal fuels. Diabetes 40, 44–50.
Hendricks CH (1964): Patterns of fetal and placental growth: the second half of normal pregnancy. Obstet. Gynecol. 24, 357–365.
Howe D, Wheeler & Perring S (1994): Measurement of placental volume with real time ultrasound in mid-pregnancy. J. Clin. Ultrasound 22, 77–83.
Kinare AS, Natekar AS, Chinchwadkar MC, Yajnik CS, Coyaji KJ, Fall CH & Howe DT (2000): Low mid-pregnancy placental volume in rural Indian women: a cause for low birth weight. Am. J. Obstet. Gynecol. 182, 443–448.
Kirchengast S & Hartmann B (1998): Maternal prepregnancy weight status and pregnancy weight gain as major determinants for newborn weight and size. Ann. Hum. Biol. 25, 17–28.
Koupilova I, Leon DA, McKeigue PM & Lithell HO (1999): Is the effect of low birth weight on cardiovascular mortality mediated through high blood pressure? J. Hypertens. 17, 19–25.
Kramer MS (1987): Determinants of low birth weight: methodological assessment and meta-analysis Bull. World Health Organ. 65, 663–737.
Launer LJ, Hofman A & Grobbee DE (1993): Relation between birth weight and blood pressure: longitudinal study of infants and children. BMJ 307, 1451–1454.
Lauszus FF, Paludan J & Klebe JG (1999): Birthweight in women with potential gestational diabetes mellitus—an effect of obesity rather than glucose intolerance? Acta Obstet. Gynecol. Scand. 78, 520–525.
Law CM & Sheill AW (1996): Is blood pressure inversely related to birth weight? The strength of the evidence from a systematic review of the literature. J. Hypertens. 14, 935–941.
Leon DA, Lithell HO, Vagero D, Koupilova I, Mohsen R & Berglund L (1998): Reduced fetal growth rate and increased risk of death from ischaemic heart disease: cohort study of 15000 Swedish men and women born 1915–29. BMJ 317, 241–244.
Lithell HO, McKeigue PM, Berglund L, Mohsen R, Lithell UB & Leon DA (1996): Relation of size at birth to non-insulin dependent diabetes and insulin concentrations in men aged 50–60 years. BMJ 312, 406–410.
Lui JP, Baker J, Perkins AS, Robertson EJ & Efstratiadis A (1993): Mice carrying null mutations of the genes encoding insulin-like growth factor I (IGF-I) and type I IGF receptor (IGFIr). Cell 75, 59–72.
McCormick MC (1985): The contribution of low birth weight to infant mortality and childhood morbidity. N. Engl. J. Med. 312, 82–90.
Paneth N & Susser M (1995): Early origin of coronary heart disease (the “Barker hypothesis”). BMJ 310, 411–412.
Rich-Edwards JW, Colditz GA, Stampfer MJ, Willett WC, Gillman MW, Hennekens CH, Speizer FE & Manson JE (1999): Birthweight and risk of type 2 diabetes mellitus in adult women. Ann. Intern. Med. 130, 278–284.
Thame M, Wilks RJ, McFarlane-Anderson N, Bennett FI & Forrester TE (1997): Relationship between maternal nutritional status and infant's weight and body proportions at birth. Eur. J. Clin. Nutr. 51, 134–138.
Thame M, Osmond C, Wilks RJ, Bennett FI, McFarlane-Anderson & Forrester TE (2000): Blood pressure is related to placental volume and birth weight. Hypertension 35, 662–667.
Thame M, Osmond C, Wilks RJ, Bennett FI & Forrester TE (2001): Second trimester placental volume and infant size at birth. Obstet. Gynecol. 98, 279–283.
Tuntiseranee P, Olsen J, Chongsuvivatwong V & Limbutara S (1999): Socioeconomic and work related determinants of pregnancy outcome in southern Thailand. J. Epidemiol. Community Health 53, 624–629.
Wolf H, Oosting H & Treffers P (1989): Second-trimester placental volume measurement by ultrasound: prediction of fetal outcome. Am. J. Obstet. Gynecol. 160, 121–126.
Woods KA, Camacho Hubner C, Savage MO & Clark AJ (1996): Intrauterine growth retardation and postnatal growth failure associated with deletion of the insulin-like growth factor I gene. N. Engl. J. Med. 335, 1363–1367.
World Health Organization (1995): Maternal anthropometry and pregnancy outcome Bull. World Health Organ 73, 21–31.
Author information
Authors and Affiliations
Contributions
Contributors: All authors have read and approved submission of the manuscript, and each has made a unique contribution to the study. MT carried out the measurements on the subjects, supervised the technical staff and participated in the writing and analysis of the manuscript. CO provided statistical advice and participated in data analysis. RW participated in the design of the study and was the clinical epidemiologist assigned to conduct the study. FB participated in the design of the study and the preparation of the manuscript. TF was the principal investigator in all matters of the conduct of the study, including the manuscript preparation. The project was supported by the Wellcome Trust.
Corresponding author
Rights and permissions
About this article
Cite this article
Thame, M., Osmond, C., Bennett, F. et al. Fetal growth is directly related to maternal anthropometry and placental volume. Eur J Clin Nutr 58, 894–900 (2004). https://doi.org/10.1038/sj.ejcn.1601909
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejcn.1601909
Keywords
This article is cited by
-
Association of trimester-specific gestational weight gain with birth weight and fetal growth in a large community-based population
Archives of Gynecology and Obstetrics (2019)
-
Obesity, maternal smoking and SHBG in neonates
Diabetology & Metabolic Syndrome (2016)
-
Is the fetoplacental ratio a differential marker of fetal growth restriction in small for gestational age infants?
European Journal of Epidemiology (2015)
-
Size at birth and risk of breast cancer: update from a prospective population-based study
European Journal of Epidemiology (2015)
-
Effect of malaria on placental volume measured using three-dimensional ultrasound: a pilot study
Malaria Journal (2012)