Abstract
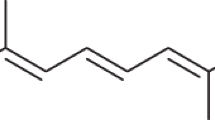
Two substances have been isolated from the urine of men which resemble the male hormone in their ability to stimulate the growth of the capon's comb1. One of these, androsterone, described in detail by Butenandt and Tscherning2, has -been produced artificially by Ruzicka and his collaborators3 by means of the oxidative decomposition of epi-dihydro-Cholesterol acetate, and has thereby been recognised as 3-epi-oxy-allo-aetio-cholanone-(17). A second oxyketone containing two hydrogen atoms less has been obtained by Butenandt and Dannenbaum4 from an unsaturated chloroketone, C19H27OC1, occurring in extracts of urine, by replacement of the chlorine atom by a hydroxyl group. On carrying out the analogous replacement reaction using the saturated chloroketone, androsterone was obtained. These reactions leave the question unanswered as to the position of the double bond and the steric position of the hydroxyl group.
Similar content being viewed by others
Article PDF
References
J. Pharm., 40, 27; 1930.
Z. physiol. Chem., 229, 167; 1934.
Helv. Chim. Acta, 17, 1389; 1934.
Z. physiol. Chem., 229, 192; 1934.
Helv. Chim. Acta, 18, 430; 1935.
Acta. brev. Neerl., 5, 1–2, 31; 1935.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
OPPENAUER, R. Synthesis of Dehydroandrosterone by the Decomposition of -Sitosterol from Soya Beans. Nature 135, 1039 (1935). https://doi.org/10.1038/1351039a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/1351039a0
This article is cited by
-
Die Wirkstoffe der männlichen Keimdrüse
Ergebnisse der Physiologie Biologischen Chemie und Experimentellen Pharmakologie (1936)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.