Abstract
Acute administration of γ-aminobutyric acid B (GABAB) receptor agonists decreased nicotine, cocaine, ethanol, and heroin self-administration. GABAB receptor agonists also decreased cue-induced cocaine craving or seeking in humans and animals, respectively. The present study investigated the effects of repeated subcutaneous administration of the GABAB receptor agonist CGP44532 on nicotine- and food-maintained responding under a fixed ratio 5 schedule of reinforcement. The second part of the study determined whether contingent presentation of previously nicotine-associated cues reinstated extinguished nicotine-seeking behavior, and whether acute subcutaneous CGP44532 administration affected cue-induced reinstatement of extinguished nicotine-seeking behavior. The results indicated that repeated administration of 0.25 mg/kg CGP44532 selectively decreased nicotine self-administration compared to food-maintained responding during the first 7 days of treatment. Repeated administration of 0.5 mg/kg/day CGP44532 nonselectively decreased both nicotine- and food-maintained responding. Contingent presentation of previously nicotine-associated cues reinstated extinguished nicotine-seeking behavior. Further, acute CGP44532 administration (0.125 and 0.25 mg/kg) decreased cue-induced reinstatement of nicotine-seeking behavior. In summary, the present results indicated that 0.25 mg/kg/day CGP44532 selectively decreased nicotine self-administration compared to food-maintained responding, and acute administration of CGP44532 (0.125 and 0.25 mg/kg) dose-dependently decreased cue-induced reinstatement of nicotine-seeking behavior.
Similar content being viewed by others
Introduction
γ-Amino-butyric acid (GABA) is one of the major inhibitory neurotransmitters in the central nervous system (Sivilotti and Nistri, 1991), and has been implicated in the modulation of central reward processes (Willick and Kokkinidis, 1995; Macey et al, 2001; Panagis and Kastellakis, 2002). Mesolimbic dopaminergic neurons originating in the ventral tegmental area receive inhibitory GABA-ergic projections from the nucleus accumbens (Kalivas et al, 1993; Rahman and McBride, 2002) and the ventral pallidum (Walaas and Fonnum, 1979; Sugita et al, 1992; Klitenick et al, 1992; Kalivas et al, 1993). Inhibitory GABAB receptors are present on dopaminergic and glutamatergic neurons in the ventral tegmental area (Bowery et al, 1987; Margeta-Mitrovic et al, 1999; Liang et al, 2000; Wirtshafter and Sheppard, 2001). Further, there are inhibitory GABA-ergic interneurons in the ventral tegmental area (Rahman and McBride, 2002). Systemic administration of the GABAB agonist baclofen decreased the firing rate and burst firing of ventral tegmental area dopaminergic neurons (Olpe et al, 1977; Erhardt et al, 2002). Further, GABAB receptor activation in the ventral tegmental area reduced extracellular dopamine levels in the nucleus accumbens (Westerink et al, 1996). Together, these data demonstrate the inhibitory role of GABAB receptors in the mesolimbic circuitry.
Either of two GABAB receptor agonists, (3-amino-2[S]-hydroxypropyl)-methylphosphinic acid (CGP44532) or baclofen, decreased self-administration of cocaine (Roberts et al, 1996; Shoaib et al, 1998; Brebner et al, 1999, 2000a, 2000b; Corrigall et al, 2000; Di Ciano and Everitt, 2003; Campbell et al, 2002), methamphetamine (Ranaldi and Poeggel, 2002), heroin (Xi and Stein, 1999), alcohol (Colombo et al, 2000, 2002), and nicotine (Corrigall et al, 2000, 2001; Fattore et al, 2002; Paterson et al, 2004) in rats. Further, baclofen decreased morphine-induced locomotor stimulation (Leite-Morris et al, 2002) and methamphetamine-induced conditioned place preference (Li et al, 2001). In addition, CGP44532 attenuated cocaine-induced facilitation of brain reward function (Dobrovitsky et al, 2002). Finally, baclofen attenuated nicotine-, cocaine- and heroin-induced increases in nucleus accumbens dopamine levels (Fadda et al, 2003).
Most previous studies investigating the effects of GABA-ergic manipulations on drug self-administration focused on acute administration of GABAB ligands (see above). However, some studies investigated the effects of repeated administration of baclofen. A 14-day baclofen administration selectively reduced alcohol consumption (Colombo et al, 2000), and 3 days of baclofen administration decreased cocaine self-administration (Shoaib et al, 1998) in rats. In addition to animal studies, human studies also indicated that repeated baclofen may reduce alcohol consumption and cocaine use (Addolorato et al, 2000, 2002; Ling et al, 1998; Shoptaw et al, 2003). The first part of the present study investigated the effects of repeated administration of the GABAB receptor agonist CGP44532 on nicotine- and food-maintained responding for two reasons. First, an effective antismoking agent is likely to be administered chronically; therefore, it is important to investigate the potential for tolerance to the effects of CGP44532. Second, the long time course (5–10 days) of extinction of nicotine-seeking behavior seen in some studies (Corrigall and Coen, 1989; Donny et al, 1995; Chiamulera et al, 1996; Shaham et al, 1997; however, see Watkins et al, 1999) suggested that the effects of acute vs repeated administration of CGP44532 could differ. Two doses of CGP44532 were selected based on previous work (Paterson et al, 2004); one dose that nonselectively decreased nicotine- and food-maintained responding (0.5 mg/kg) and another dose (0.25 mg/kg) that had no effect on either nicotine (0.03 mg/kg/infusion)- or food-maintained responding when administered acutely.
When animals trained to self-administer a drug are given access to saline rather than drug, response rates dramatically decrease (Chiamulera et al, 1996; Shaham et al, 1997; See et al, 1999; Watkins et al, 1999). If specific environmental stimuli are present during drug self-administration but are removed during the extinction phase, then noncontingent (de Wit and Stewart, 1981) or contingent (See et al, 2001) presentation of previously drug-associated stimuli will induce responding levels similar to those seen for the drug itself, even in the continued absence of the drug. Shaham and co-workers classifed ‘cues’ into discrete cues that are paired with drug infusions (See, 2002), discriminative cues that predict the availability of drug (Weiss et al, 2000), and contextual cues (Crombag and Shaham, 2002) that are associated with drug availability. It is hypothesized that drug-associated cues play an important role in both relapse to drug use (for a review, see Shaham et al, 2003) and at least for nicotine, acquiring and maintaining self-administration behavior in rats (Caggiula et al, 2001, 2002), and smoking in humans (Balfour et al, 2000).
The GABAB receptor agonist baclofen has been shown to decrease cue-induced activation of limbic areas in abstinent cocaine addicts (Childress et al, 2002). Further, acute administration of baclofen either systemically (Di Ciano and Everitt, 2003), or into the ventral tegmental area or nucleus accumbens core (Di Ciano and Everitt, 2004) decreased cue-maintained responding during the first drug-free interval under a second-order schedule of reinforcement in rats, demonstrating decreased drug-seeking behavior. The present study set out to establish first whether nicotine-seeking behavior was reinstated by contingent presentation of previously nicotine-associated cues, and second whether acute CGP44532 administration affected cue-induced reinstatement.
In summary, the first part of the present study determined the effects of repeated administration of the GABAB receptor agonist CGP44532 on nicotine- and food-maintained responding. The second part of the present study determined whether contingent presentation of previously nicotine-associated cues reinstated extinguished nicotine-seeking behavior, and the effects of acute CGP44532 administration on cue-induced reinstatement.
Methods
Subjects
Male Wistar rats (Charles River, Raleigh, NC) weighing 300–350 g upon arrival in the laboratory were group housed (two per cage) in a temperature- and humidity-controlled vivarium on a 12-h reverse light–dark cycle with unrestricted access to water except during testing. Rats were food-restricted to 12–20 g/day throughout the experiments (see below), with the exception of the surgery recovery and initial habituation periods. All behavioral testing occurred during the dark phase of the light–dark cycle. For 1 week after arrival in the vivarium, animals were allowed to habituate to their new environment (rats were handled twice during this week). All subjects, animal facilities, and experimental protocols were in accordance with NIH guidelines (1996) and guidelines from the Association for the Assessment and Accreditation of Laboratory Animal Care (AAALAC).
Drugs
(−)Nicotine hydrogen tartrate was purchased from Sigma (St Louis, MO). Nicotine doses are reported as free base concentrations. Nicotine hydrogen tartrate was dissolved in saline and the pH adjusted to 7.0 (±0.5) with sodium hydroxide. The solution was then filtered through a 0.22 μm syringe filter (Fisher Scientific, Pittsburgh, PA 15219) for sterilization purposes. CGP44532 was kindly donated by Novartis Pharma AG, dissolved in 0.9% saline and administered subcutaneously in a volume of 1 ml/kg, with a pretreatment time of 15 min.
Apparati
Intravenous nicotine self-administration and food-maintained responding took place in Plexiglas experimental chambers (25 × 31 × 24 cm; Med Associates, St Albans, VT), each housed in a sound-attenuated box (San Diego Instruments, San Diego, CA). One wall of the chamber contained two levers, measuring approximately 3 cm in width, and located approximately 3 cm above the metal grid floor of the chamber. All data collection and test session functions were controlled by a microcomputer and in-house software.
Surgery
Rats were anaesthetized with an isoflurane/oxygen mixture (1–3% isoflurane) and prepared with a catheter inserted into the right jugular vein. Catheters were constructed from a 14 cm piece of Silastic tubing (0.3 mm inside diameter × 0.63 mm outside diameter, Dow Corning) attached to a 22-gauge stainless steel guide cannula (Plastics One, Roanoke, VA), and attached to a molded base (Teets Denture Material, Co-Oral-Lite Mfg. Co., Diamond Springs, CA) and mesh (Small Parts Inc., Miami Lakes, FL). The mesh anchored the catheter base underneath the skin at the level of the scapulae. Animals were given 7 days to recover from surgery prior to being trained to lever press for 45 mg food pellets (described below). All animals received a 1-week course of antibiotic treatment, consisting of daily 0.2 ml infusions of Timentin (100 mg/ml). In addition, animals received a 0.1 ml infusion of heparinized saline (33.3 U/ml) before and after each self-administration session.
Food Training and Testing
Approximately 1 week after preparation with catheters, rats were food-restricted (5 g/day) for 48 h prior to starting food training. After the initiation of food training, animals received 12 g rat chow/day, at least 1 h after the end of the food training session. Initially, the animals received 45 mg food pellets at fixed intervals (one pellet every 12 s for 5 min, then 5 min break; this sequence was repeated during the initial 30 min session; total number of pellets delivered was 75) with no requirement to lever press. After this session, rats were allowed to press a lever to receive the food pellets on a fixed ratio 1 time-out 1 s (FR1 TO1 s) schedule of reinforcement. The schedule was progressively changed according to the sequence FR1 TO1 s, FR1 TO10 s, FR2 TO20 s, FR5 TO20 s, with sessions lasting 30 min. Animals moved through the sequence only after the successful acquisition of the previous schedule (defined as earning 50 pellets within the 30 min session).
An identical training procedure was used for the food-maintained responding subjects, except that after successful acquisition of food-maintained responding, the rats were allowed to respond for food (45 mg Noyes food pellet) on a FR5 TO20 s schedule (session length 1 h) 7 days/week, during which two levers (active and inactive) were present in the box. This testing procedure was identical in all parameters to the nicotine self-administration procedure. After successful acquisition of food-maintained responding, all rats were maintained on 20 g rat chow/day, given at least 1 h after testing.
Nicotine Self-Administration
After the successful completion of food training, rats were allowed to self-administer nicotine (0.03 mg/kg/infusion; free base) by switching the delivery of a food pellet for the delivery of a nicotine infusion. This dose of nicotine was selected because it leads to reliable and stable self-administration rates (Watkins et al, 1999). There were two levers present in the box, only one of which (the lever previously paired with food delivery) was paired with the delivery of a nicotine infusion (the active lever). Responses on the other lever were recorded but had no consequence (the inactive lever). Responding on the active lever resulted in delivery of the nicotine solution in a volume of 0.1 ml over a 1 s period (Razel syringe pump model A, Razel Scientific Instruments Inc., Stamford, CT). The delivery of an infusion was paired with a cue light, which was lit simultaneously with the initiation of the nicotine infusion and remained illuminated throughout the 20 s time-out period, during which responding was recorded but not reinforced. The delivery of an infusion was earned by responding five times on the active lever (FR5 TO 20 s). Rats were considered to have acquired stable nicotine self-administration when they pressed the active lever more than twice the number of times they pressed the inactive lever and received a minimum of six infusions/1 h session, with less than 20% variation in the number of infusions earned per session over three consecutive sessions. Rats were allowed to self-administer nicotine 7 days/week.
Experimental Procedures
Experiment 1: the effects of repeated CGP44532 administration on nicotine- and food-maintained responding
After successful acquisition of stable nicotine- or food-maintained responding, rats were assigned to one of six groups, balanced for mean daily operant response rates and body-weights. The groups were as follows: 0 mg/kg/day CGP44532 (nicotine: n=9; food: n=9), 0.25 mg/kg/day CGP44532 (nicotine: n=8; food: n=8), and 0.5 mg/kg/day CGP44532 (nicotine: n=10; food: n=7). CGP44532 was administered subcutaneously 15 min before daily testing sessions for 14 consecutive days. The pretreatment time was based on results from a previous study from our laboratory indicating reliable effects of CGP44532 on nicotine self-administration when administered 15 min prior to testing (Paterson et al, 2004), and another study indicating that CGP44532 (0.5 mg/kg i.p.), administered 10 min prior to testing, decreased cocaine-maintained responding for 4 h post-treatment (Brebner et al, 1999). Rats were allowed to respond for food or nicotine for 4 days after CGP44532 administration ceased.
Experiment 2: the effects of acute CGP44532 administration on cue-induced reinstatement of nicotine self-administration
After successful completion of experiment 1, nine rats were not allowed any access to nicotine self-administration for 9 days. Three rats were from the saline treatment group, four from the 0.25 mg/kg/day CGP44532 group, and one from the 0.5 mg/kg/day CGP445342 group, for a total of eight rats (one rat was excluded due to infection). The rats were again given access to nicotine for 5 days to re-establish stable nicotine self-administration under baseline conditions. After another 2 days with no access to nicotine self-administration, rats were once again allowed access to the self-administration apparatus on a daily basis but lever-pressing had no consequences (ie no infusion, no cue light illumination, and no syringe pump activation and noise). After all animals reached the predetermined criterion of extinction (defined as less than 30% of average nicotine-maintained responding per session for 3 consecutive days; extinction criterion selected based on previous nicotine self-administration extinction studies (Chiamulera et al, 1996; Shaham et al, 1997)), previously nicotine-associated cues were once again presented contingently (ie upon responding on the active lever) in the absence of nicotine for 1 day. Rats were then returned to the nicotine- and cue-absent condition until the re-establishment of extinction (less than 30% of average nicotine-maintained responding for 3 consecutive days). After re-establishment of extinction, testing with contingent cue delivery was conducted for 1 day. This process was repeated a total of three times. CGP44532 (0, 0.125, and 0.25 mg/kg) was administered subcutaneously 15 min before the test session, according to a randomized-order within-subjects design. The procedure used here was modified from the one used by See et al (2001), where repeated test sessions (two) consisting of contingent presentation of previously drug-associated cues with preceding and intervening periods of responding under extinction conditions were used to examine the effects of pharmacological manipulations on conditioned reinstatement of drug-seeking behavior. In summary, the time-line in the present study was as follows: 9 days ‘wash-out’ period after chronic CGP44532/saline administration; 5 days nicotine self-administration; 12 days extinction; 1 day test; 4–6 days extinction; 1 day test (CGP44532); 4–6 days extinction; 1 day test (CGP44532); 4–6 days extinction; 1 day test (CGP44532).
Data Analyses
In Experiment 1, active lever data were expressed as percent of baseline (mean of last 3 days of nicotine self-administration prior to treatment), while the inactive lever data were expressed as absolute number of responses. Body-weight data were also expressed as percent of baseline. In Experiment 2, all lever response data were expressed as absolute number of responses.
For Experiment 1, data were analyzed using two three-way analyses of variance (ANOVAs) with CGP44532 Dose (three levels) and Reinforcer (two levels) as between-subjects factors, and Days (18 levels; nine levels for body weight data) as the within-subjects factor. Also, a one-way ANOVA was used to further analyze inactive lever data with CGP44532 Dose (three levels) defined as the between-subjects factor. In addition, body-weight data were analyzed with two one-way ANOVAs with CGP44532 Dose (three levels) as between-subjects factors, and Days (9 levels) as the within-subjects factor. For Experiment 2, two one-way ANOVAs and a two-way ANOVA were used to analyze the extinction and reinstatement data, respectively. In the first ANOVA, Days (13 levels: baseline day, 12 days extinction) was the within-subjects factor. In the second one-way ANOVA, Cue (two levels) was the within-subjects factor. In the two-way ANOVA, CGP44532 Dose (three levels) and Cues (two levels) were the within-subjects factors. The two levels of Cue were the test days (cue present) and the mean response rate over the preceding 3 baseline days (nicotine- and cue-absent). Finally, a one-way ANOVA was used to compare baseline nicotine self-administration prior to CGP44532 treatment with self-administration prior to extinction for the eight rats used in both experiments to assess possible drift in responding levels during the course of the study. Where indicated by significant main or interaction effects, one- or two-way way follow-up ANOVAs or Newman–Keuls posthoc tests were conducted (Winer, 1971).
Results
Experiment 1: the Effects of Repeated CGP44532 Treatment on Nicotine- and Food-Maintained Responding
Table 1 shows the mean levels of responding by all experimental groups and for all reinforcers prior to the start of CGP44532 treatment. A three-way ANOVA indicated that CGP44532 treatment affected nicotine- and food-maintained responding differently across days (Reinforcer × Dose × Days interaction: F(34, 731)=1.76, p<0.05). Furthermore, there were significant interaction effects of Days × Dose (F(34, 731)=5.07, p<0.01), Days × Reinforcer (F(17, 731)=9.71, p<0.01), and significant main effects of Days (F(17, 731)=14.66, p<0.01) and Dose (F(2, 43)=29.22, p<0.01). The Newman–Keuls post hoc comparisons indicated that 0.25 mg/kg/day CGP44532 decreased nicotine-maintained responding relative to the saline group on days 1, 4, 5, 6, and 7 (see Figure 1a). Further, nicotine-maintained responding was significantly decreased in the 0.5 mg/kg/day CGP44532 group relative to the saline group on days 1 through 14 of treatment (see Figure 1a). Food-maintained responding was not significantly decreased in the 0.25 mg/kg/day CGP44532 group compared to the saline group at any time point. Food-maintained responding was significantly decreased in the 0.5 mg/kg/day CGP44532 group compared to the saline-treated group on treatment days 1 through 14, and on post-treatment day 1 (see Figure 1b).
The effects of repeated administration of the GABAB receptor agonist CGP44532 on nicotine- and food-maintained responding (Experiment 1). Panels a and b show the effects of 0, 0.25, and 0.5 mg/kg/day CGP44532 on responding for nicotine and food, respectively. The data are expressed as percentage of baseline performance (mean±SEM). Asterisks (*p<0.05, **p<0.01) indicate significant differences from the saline-treated group. Pound sign (#p<0.05) indicates significantly decreased responding for food on the first post-CGP44532 treatment day, compared to the vehicle control group. Arrows indicate the start and end of CGP44532 treatment.
Finally, analysis of inactive lever data indicated a significant main effect of CGP44532 Dose (decreased responding; F(2, 43)=3.85, p<0.05), but no other significant main or interaction effects (data not shown). Further analysis of inactive lever data using a one-way ANOVA with Dose defined as the between-subjects factor indicated a significant main effect of Dose (F(2, 46)=4.7, p<0.05). The Newman–Keuls post hoc comparisons indicated that inactive lever responding was significantly decreased in the 0.5 mg/kg/day CGP44532 group compared to both the saline and the 0.25 mg/kg/day CGP44532 groups (data not shown).
Table 1 shows the mean body weights for all experimental groups prior to the start of CGP44532 treatment. A three-way ANOVA indicated that CGP44532 treatment affected body weights in the different treatment groups differently across days (Reinforcer × CGP44532 Dose × Days interaction: F(16, 344)=3.42, p<0.01). Furthermore, there were significant interaction effects of Days × Dose (F(16, 344)=2.53, p<0.05), Dose × Reinforcer (F(2, 43)=8.81, p<0.01), and Days × Reinforcer (F(8, 344)=37.07, p<0.01). Finally, there were significant main effects of Days (F(8, 344)=316.34, p<0.001), Dose (F(2, 43)=13.26, p<0.01)], and Reinforcer (F(1, 43)=132.21, p<0.001)]; (see Figure 2). A follow-up one-way ANOVA indicated that body weights in the nicotine-responding groups increased over time (significant main effect of Days F(8, 192)=83.47, p<0.01) regardless of CGP44532 Dose administered (no significant main effect of CGP44532 Dose or interaction effect). In contrast, body weights in the food-responding groups increased over time (Days: F(8, 152)=297.22, p<0.01)], with different rates of increase in different treatment groups (CGP44532 Dose: F(2, 19)=47.87, p<0.01; CGP44532 Dose × Days: F(16, 152)=3.93, p<0.01).
The effect of repeated administration of the GABAB receptor agonist CGP44532 on body weight (Experiment 1). The graph shows body-weight data expressed as a percentage of pretreatment baseline body weights over nine time points (corresponding to body weight assessed every second day). See Results section for description of statistically significant effects.
Experiment 2: the Effects of Acute CGP44532 Administration on Cue-Induced Reinstatement of Nicotine-Seeking Behavior
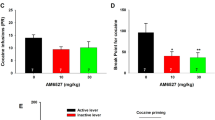
Comparison of nicotine self-administration during the baseline periods prior to the initiation of CGP44532 treatment (Experiment 1) and prior to extinction (Experiment 2) indicated no significant difference in baseline nicotine-maintained responding over the course of the study (F(1, 7)=3.8, NS). One-way ANOVA indicated that lever-pressing on the active lever significantly decreased during nicotine- and cue-absent extinction sessions (Days: F(12, 84)=16.16, p<0.001). The Newman–Keuls comparisons indicated that responding was significantly decreased compared to the last day of nicotine-maintained responding on days 2 through 12 of extinction in the absence of cues. Responding was significantly increased when previously drug-associated cues were presented contingently in the absence of nicotine (significant main effect of Cue: F(1, 7)=41.67, p<0.001; see Figure 3a). In contrast, analysis of inactive lever data indicated no significant main effect of Days (F(12, 84)=2.5, NS) or Cues (F(1, 7)=0.8, NS; data not shown).
The effect of acute administration of the GABAB receptor agonist CGP44532 on cue-induced reinstatement of nicotine-seeking behavior (Experiment 2). Panel a shows the extinction of nicotine-seeking behavior over 12 days (open square, ‘B’ on abscissa axis: nicotine self-administration; closed square: nicotine- and cue-absent nicotine-seeking), and the effect of contingent presentation of drug-associated cues on day 13 (open circle, ‘CS+’ on abscissa axis). The dotted line indicates the level of the predetermined criterion for extinction (ie 30% of nicotine self-administration active-lever-pressing). Asterisks (*p<0.05) indicate significant differences from nicotine self-administration baseline. The pound sign (#p<0.05) indicates significant difference between extinction (days 10, 11, and 12) and cue-induced reinstatement test day. Panel b shows the effects of acute administration of CGP44532 on cue-induced reinstatement of nicotine-seeking behavior. Asterisks (*p<0.05, **p<0.01) indicate significant differences from saline pretreatment. The pound sign (#p<0.05) indicates significant differences between the test day and the preceding 3-day baseline. In both panels, data are expressed as mean number of lever presses±SEM.
A two-way ANOVA indicated that contingent cue-presentation consistently increased active lever responding (Cues: F(1, 14)=6.75, p<0.05), and that administration of CGP44532 significantly decreased lever-pressing on the active lever when previously nicotine-associated cues were presented contingently in the absence of nicotine (Cues × CGP44532 Dose: F(2, 28)=5.06, p<0.05). There was no significant main effect of CGP44532 (F(2, 28)=2.39, NS). The Newman–Keuls comparisons indicated that responding after administration of 0.125 or 0.25 mg/kg CGP44532 was significantly decreased compared to responding after saline administration (see Figure 3b). Further, responding after saline administration was significantly increased compared to pretest baseline, unlike responding after administration of either CGP44532 Dose (see Figure 3b).
Discussion
The present results indicate that repeated administration of the GABAB receptor agonist CG44532 decreased nicotine- and food-maintained responding at the highest dose tested, although the lower dose of CGP44532 selectively decreased nicotine self-administration compared to food-maintained responding. In the second experiment, nicotine-seeking behavior that was extinguished in the absence of nicotine and previously nicotine-associated cues was reinstated when those cues were presented contingently, although no nicotine was available. Importantly, cue-induced reinstatement of nicotine-seeking behavior was significantly decreased after acute CGP44532 administration.
In a previous study from this laboratory (Paterson et al, 2004), 0.25 mg/kg CGP44532 administered acutely had no significant effect on self-administration of 0.03 mg/kg/infusion nicotine, but did significantly and selectively decrease self-administration of 0.01 mg/kg/infusion nicotine compared to food-maintained responding. In the present study, nicotine self-administration was significantly decreased on day 1 and days 4–7 of treatment with 0.25 mg/kg/day CGP44532, while food-maintained responding was unaffected. Further, administration of 0.5 mg/kg/day CGP44532 significantly decreased nicotine self-administration and food-maintained responding throughout the 14 days of repeated CGP44532 administration, consistent with the nonspecific effect of acutely administered 0.5 mg/kg CGP44532 (Paterson et al, 2004). After termination of repeated administration of CGP44532, nicotine self-administration immediately returned to levels close to pre-CGP44532 levels of responding. In summary, administration of 0.25 mg/kg CGP445342 significantly decreased nicotine self-administration for the majority of the first 7 days of treatment, without affecting food-maintained responding at any time point. In contrast, 0.5 mg/kg/day CGP44532 consistently decreased both nicotine self-administration and food-maintained responding during the entire 14 days of repeated CGP44532 treatment. The nicotine self-administration data in the present study indicate the development of tolerance to the effects of repeated administration of CGP44532. The effects of 0.25 mg/kg/day CGP44532 were no longer significant after day 7 of treatment. The effects of 0.5 mg/kg/day CGP445342 remained significant throughout the 14 days of treatment, although nicotine self-administration behavior increased slowly over the last 4 or 5 days of treatment. Previous studies indicated that repeated administration of the GABAB receptor agonist baclofen resulted in downregulation of GABAB receptor function in rat cortex, spinal cord (Malcangio et al, 1995), and hippocampus (Wetherington and Lambert, 2002) without associated decreases in GABAB binding sites, mRNA, or protein levels for specific GABAB receptor subtypes (Lehmann et al, 2003). These data suggest a possible explanation for the tolerance to the effects of CGP44532 observed in the present study. Alternatively, nicotine may exert effects on GABAB receptor function or number. Nicotine exerts effects on GABA-ergic neurotransmission (Mansvelder et al, 2002) via nicotinic receptors located on GABA-ergic interneurones in the ventral tegmental area, and repeated nicotine administration decreases GABAB receptor expression (Li et al, 2002). Repeated administration of psychostimulants such as cocaine or amphetamine is associated with decreased functional coupling of GABAB receptors to G-proteins (Kushner and Unterwald, 2001; Zhang et al, 2000), decreases in G-proteins levels (Nestler et al, 1990; Striplin and Kalivas, 1992, 1993), increased presynaptic GABAB heteroreceptor function (Giorgetti et al, 2002), and elevated extracellular GABA levels (Xi et al, 2003). Thus, it is possible that nicotine self-administration in the present study decreased GABAB receptor function or number prior to, and during, CGP44532 treatment, resulting in the development of tolerance.
The food responding group exposed to 0.5 mg/kg/day CGP44532 exhibited significantly decreased responding on the first post-treatment day, in contrast to the nicotine self-administration groups that returned to pre-CGP44532 levels of responding. Although the effects of the GABAB receptor agonist baclofen on food-maintained responding have been mixed (Ebenezer and Pringle, 1992; Brebner et al, 1999; Colombo et al, 2000; Paterson et al, 2004), these prior studies have generally focused on acute rather than repeated baclofen administration. The observed post-treatment decrease in food-maintained responding in the present study may be explained by a conditioning effect that was not occurring with for nicotine or by CGP44532-induced changes in GABAB receptor activity (see above) in the absence of nicotine-induced changes in GABAB receptor activity. The greater body weights in the food- vs nicotine-responding groups, and the greater body weights exhibited in the saline- vs CGP44532-treated food-responding groups, are most likely due to the additional food obtained via operant responding and the suppression of food-maintained responding during CGP44532 treatment, respectively, because all groups were maintained on 20 g rat chow/day throughout the entire experiment.
The present data are similar to the suppressant effects of repeated baclofen administration on alcohol self-administration in rats (Colombo et al, 2000) that appeared to decrease over the course of a 14-day treatment. In contrast to the present data, however, food-maintained responding in the Colombo study was initially decreased in the high dose baclofen group, but this effect disappeared after approximately 7 days of treatment. The observed differences in the effects of repeated baclofen on food-maintained responding between the Colombo study (2000) and the present study is likely due to the fact that the rats in the Colombo study were entirely dependent on operant responding to obtain food. Similarly, a previous study that indicated a selective effect of baclofen on cocaine- vs food-maintained responding used a procedure in which rats relied on operant responding for all food (Brebner et al, 1999). In the present study, all rats received 20 g of rat chow/day throughout the testing phase. Finally, the present results are consistent with the decreases in nicotine self-administration observed after acute administration of baclofen (Corrigall et al, 2000, 2001; Fattore et al, 2002; Paterson et al, 2004), CGP44532 (Paterson et al, 2004), or the GABA transaminase inhibitor γ-vinyl-GABA (vigabatrin; Paterson and Markou, 2002).
Although the effects of 0.25 mg/kg/day CGP44532 appeared to be selective for nicotine-maintained responding over the first 7 days of treatment, it should be noted that responding for basic physiologically relevant reinforcers, such as food, has previously been found to be more difficult to disrupt than responding for drug rewards (eg Negus and Mello, 2003; for a review, see McMillan and Katz, 2002). Nonetheless, there has not been a thorough evaluation of the predictive validity of these apparent reinforcer-specific effects (McMillan and Katz, 2002). Thus, it is difficult to draw conclusions regarding the wider implications of effects on drug- vs food-maintained responding. Food-maintained responding occurred at much higher rates than nicotine-maintained responding in the present study, and therefore may have been differentially affected by the drug manipulations. Pioneering work by Dews (1955) indicated that the effects of drug pretreatments depended on the schedules of reinforcement used, and the rates of responding (for a review, see McMillan and Katz, 2002). Thus, the specific effect of 0.25 mg/kg/day CGP44532 on nicotine- vs food-maintained responding observed in the present study may be due to differences in the rates of reinforced responding or to selective blockade of nicotine vs food reward. Considering only the nicotine self-administration data in the present study, however, 0.25 mg/kg/day CGP44532 (which selectively decreased nicotine-maintained responding) had no effect on inactive lever presses; in contrast, 0.5 mg/kg/day CGP44532 (which nonselectively decreased nicotine-maintained responding) significantly decreased inactive lever presses also.
The results in the second part of the present study indicated that nicotine-seeking behavior is reinstated by contingent presentation of previously drug-associated cues, as shown previously for cocaine (eg See et al, 1999, 2001). The long time course of extinction seen in the present study (12 days) is consistent with previous reports (Corrigall and Coen, 1989; Donny et al, 1995, 1999; Chiamulera et al, 1996). It has also been shown previously that nicotine-seeking behavior was reinstated by noncontingent delivery of nicotine and by physical stress (Chiamulera et al, 1996; Shaham et al, 1997), again similarly to cocaine self-administration (for a review, see Shaham et al, 2003). The demonstration of reinstatement of nicotine-seeking induced by contingent presentation of nicotine-associated cues in the present study is consistent with a previous study in rats (Caggiula et al, 2001), and with the role of cues in maintaining smoking behavior (Rose et al, 1985, 1993) and contributing to relapse (Droungas et al, 1995; Niaura et al, 1998; Due et al, 2002) in humans. Administration of CGP44532 in the present study significantly decreased cue-induced reinstatement of nicotine-seeking behavior in rats. Similarly, baclofen was recently shown to decrease cocaine- and heroin-seeking behavior during the first, drug-free, interval of responding under a second-order schedule of reinforcement (Di Ciano and Everitt, 2003). Interestingly, baclofen administration decreased cue-induced craving for cocaine in humans (Childress et al, 2002). Although the subjects were receiving repeated baclofen treatment, two patients who had not taken their medication on the test day exhibited cue-induced craving at a level similar to controls, perhaps suggesting an acute rather than repeated effect of baclofen.
In summary, the results of the present study indicated that repeated administration of CGP44532 had different effects on nicotine- vs food-maintained responding. Taken together, the results indicated that 0.25 mg/kg/day CGP44532 selectively decreased nicotine self-administration compared to food-maintained responding during the majority of the first 7 days of treatment, while 0.5 mg/kg/day CGP44532 consistently decreased both nicotine- and food-maintained responding throughout the 14 days of treatment. Further, contingent presentation of previously nicotine-associated cues reinstated extinguished nicotine-seeking behavior. Finally, acute administration of CGP44532 significantly attenuated cue-induced reinstatement of nicotine-seeking behaviour. Thus, the present data suggest that GABAB receptor agonists may have potential as a medication for smoking cessation and for preventing relapse in human smokers triggered by previously nicotine-associated cues.
References
Addolorato G, Caputo F, Capristo E, Colombo G, Gessa GL, Gasbarrini G (2000). Ability of baclofen in reducing alcohol craving and intake: II – preliminary clinical evidence. Alcohol: Clin Exp Res 24: 67–71.
Addolorato G, Caputo F, Capristo E, Domenicali M, Bernardi M, Janiri L et al (2002). Baclofen efficacy in reducing alcohol craving and intake: a preliminary double-blind randomized controlled study. Alcohol Alcohol 37: 504–508.
Balfour DJK, Wright AE, Benwell MEM, Birrell CE (2000). The putative role of extra-synaptic mesolimbic dopamine in the neurobiology of nicotine dependence. Behav Brain Res 113: 73–83.
Bowery NG, Hudson AL, Price GW (1987). GABAA and GABAB receptor site distribution in the rat central nervous system. Neuroscience 20: 365–383.
Brebner K, Froestl W, Andrews M, Phelan R, Roberts DCS (1999). The GABAB agonist CGP44532 decreases cocaine self-administration in rats: demonstration using a progressive-ratio and a discrete trials procedure. Neuropharmacology 38: 1797–1804.
Brebner K, Phelan R, Roberts DCS (2000a). Effect of baclofen on cocaine self-administration in rats reinforced under fixed-ratio 1 and progressive-ratio schedules. Psychopharmacology 148: 314–321.
Brebner K, Phelan R, Roberts DCS (2000b). Intra-VTA baclofen attenuates cocaine self-administration on a progressive ratio schedule of reinforcement. Pharmacol Biochem Behav 66: 857–862.
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA et al (2001). Cue dependency of nicotine self-administration and smoking. Pharmacol Biochem Behav 70: 515–530.
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA et al (2002). Environmental stimuli promote the acquisition of nicotine self-administration in rats. Psychopharmacology 163: 230–237.
Campbell UC, Morgan AD, Carroll ME (2002). Sex differences in the effects of baclofen on the acquisition of intravenous cocaine self-administration in rats. Drug Alcohol Depend 66: 61–69.
Chiamulera C, Borgo C, Falchetto S, Valerio E, Tessari M (1996). Nicotine reinstatement of nicotine self-administration after long-term extinction. Psychopharmacology 127: 102–107.
Childress AR, Franklin T, Listerud J, Acton P, O'Brien CP (2002). Neuroimaging of cocaine craving states: cessation, stimulant administration, and drug cue paradigms. In: The College of Drug Dependence: Proceedings of the 64th Annual Meeting Abstracts. Quebec City, Quebec, Canada.
Colombo G, Agabio R, Carai MA, Lobina C, Pani M, Reali R et al (2000). Ability of baclofen in reducing alcohol intake and withdrawal severity: I-Preclinical evidence. Alcohol Clin Exp Res 24: 58–66.
Colombo G, Serra S, Brunetti G, Atzori G, Pani M, Vacca G et al (2002). The GABAB receptor agonists baclofen and CGP 44532 prevent acquisition of alcohol drinking behaviour in alcohol-preferring rats. Alcohol Alcohol 37: 499–503.
Corrigall WA, Coen KM (1989). Nicotine maintains robust self-administration in rats on a limited-access schedule. Psychopharmacology 99: 473–478.
Corrigall WA, Coen KM, Adamson KL, Chow BLC, Zhang J (2000). Response of nicotine self-administration in the rat to manipulations of mu-opioid and gamma-aminobutyric acid receptors in the ventral tegmental area. Psychopharmacology 149: 107–114.
Corrigall WA, Coen KM, Zhang J, Adamson KL (2001). GABA mechanisms in the pedunculopontine tegmental nucleus influence particular aspects of nicotine self-administration selectively in the rat. Psychopharmacology 158: 190–197.
Crombag HS, Shaham Y (2002). Renewal of drug seeking by contextual cues after prolonged extinction in rats. Behav Neurosci 116: 169–173.
de Wit H, Stewart J (1981). Reinstatement of cocaine-reinforced responding in the rat. Psychopharmacology 75: 134–143.
Dews PB (1955). Studies on behavior. I. Differential sensitivity to pentobarbital of pecking performance in pigeons depending on the schedule of reward. J Pharmacol Exp Ther 138: 393–401.
Di Ciano P, Everitt BJ (2003). The GABAB receptor agonist baclofen attenuates cocaine- and heroin-seeking behavior by rats. Neuropsychopharmacology 28: 510–518.
Di Ciano P, Everitt BJ (2004). Contribution of the ventral tegmental area to cocaine-seeking maintained by a drug-paired conditioned stimulus in rats. Eur J Neurosci 19: 1661–1667.
Dobrovitsky V, Pimentel P, Duarte A, Froestl W, Stellar JR, Trzcinska M (2002). CGP44532, a GABAB receptor agonist, is hedonically neutral and reduces cocaine-induced enhancement of reward. Neuropharmacology 42: 626–632.
Donny EC, Caggiula AR, Knopf S, Brown C (1995). Nicotine self-administration in rats. Psychopharmacology 122: 390–394.
Donny EC, Caggiula AR, Mielke MM, Booth S, Gharib MA, Hoffman A et al (1999). Nicotine self-administration in rats on a progressive-ratio schedule of reinforcement. Psychopharmacology 147: 135–142.
Droungas A, Ehrman RN, Childress AR, O'Brien CP (1995). Effects of smoking cues and cigarette availability on craving and smoking behavior. Addict Behav 20: 657–673.
Due DL, Huettel SA, Hall WG, Rubin DC (2002). Activation in mesolimbic and visuospatial neural circuits elicited by smoking cues: evidence from functional magnetic resonance imaging. Am J Psychiatry 159: 954–960.
Ebenezer IS, Pringle AK (1992). The effect of systemic administration of baclofen on food intake in rats. Neuropharmacology 31: 39–42.
Erhardt S, Mathe JM, Chergui K, Engberg G, Svensson TH (2002). GABAB receptor-mediated modulation of the firing pattern of ventral tegmental area dopamine neurons in vivo. Naunyn-Schmiedeberg's Arch Pharmacol 365: 173–180.
Fadda P, Scherma M, Fresu A, Collu M, Fratta W (2003). Baclofen antagonizes nicotine-, cocaine-, and morphine-induced dopamine release in the nucleus accumbens of rat. Synapse 50: 1–6.
Fattore L, Cossu G, Martellotta MC, Fratta W (2002). Baclofen antagonizes intravenous self-administration of nicotine in mice and rats. Alcohol Alcohol 37: 495–498.
Giorgetti M, Hotsenpiller G, Froestl W, Wolf ME (2002). In vivo modulation of ventral tegmental area dopamine and glutamate efflux by local GABAB receptors is altered after repeated amphetamine treatment. Neuroscience 109: 585–595.
Kalivas PW, Churchill L, Klitenick MA (1993). GABA and enkephalin projection from the nucleus accumbens and ventral pallidum to the ventral tegmental area. Neuroscience 57: 1047–1060.
Klitenick MA, De Witte P, Kalivas PW (1992). Regulation of somatodendritic dopamine release in the ventral tegmental area by opioids and GABA: an in vivo microdialysis study. J Neurosci 12: 2623–2632.
Kushner SA, Unterwald EM (2001). Chronic cocaine administration decreases the functional coupling of GABAB receptors in the rat ventral tegmental area as measured by baclofen-stimulated 35S-GTPγS binding. Life Sci 69: 1093–1102.
Lehmann A, Matteson JP, Edlund A, Johansson T, Ekstrand AJ (2003). Effects of repeated administration of baclofen to rats on GABAB receptor binding sites and subunit expression in the brain. Neurochem Res 28: 387–393.
Leite-Morris KA, Fukudome EY, Kaplan GB (2002). Opiate-induced motor stimulation is regulated by gamma-aminobutyric acid type B receptors found in the ventral tegmental area in mice. Neurosci Lett 317: 119–122.
Li SM, Yin LL, Ren YH, Pan LS, Zheng JW (2001). GABAB receptor agonist baclofen attenuates the development and expression of d-methamphetamine-induced place preference in rats. Life Sci 70: 349–356.
Li SP, Park MS, Bahk JY, Kim MO (2002). Chronic nicotine and smoking exposure decreases GABA(B1) receptor expression in the rat hippocampus. Neurosci Lett 334: 135–139.
Liang F, Hatanaka Y, Saito H, Yamamori T, Hashikawa T (2000). Differential expression of gamma-aminobutyric acid type B receptor-1a and -1b mRNA variants in GABA and non-GABAergic neurons of the rat brain. J Comp Neurol 416: 475–495.
Ling W, Shoptaw S, Majewska D (1998). Baclofen as a cocaine anti-craving medication: a preliminary clinical study. Neuropsychopharmacology 18: 403–404.
Macey DJ, Froestl W, Koob GF, Markou A (2001). Both GABAB receptor agonist and antagonists decreased brain stimulation reward in the rat. Neuropharmacology 40: 676–685.
Malcangio M, Libri V, Teoh H, Constanti A, Bowery NG (1995). Chronic (−)baclofen or CGP 36742 alters GABAB receptor sensitivity in rat brain and spinal cord. Neuroreport 6: 399–403.
Mansvelder HD, Keath JR, McGehee DS (2002). Synaptic mechanisms underlie nicotine-induced excitability of brain reward areas. Neuron 33: 905–919.
Margeta-Mitrovic M, Mitrovic I, Riley RC, Jan LY, Basbaum AI (1999). Immunohistochemical localization of GABAB receptors in the rat central nervous system. J Comp Neurol 405: 299–321.
McMillan SE, Katz JL (2002). Continuing implications of the early evidence against the drive-reduction hypothesis of the behavioral effects of drugs. Psychopharmacology 163: 251–264.
Negus SS, Mello NK (2003). Effects of chronic D-amphetamine treatment on cocaine- and food-maintained responding under a second-order schedule in rhesus monkeys. Drug Alcohol Depend 70: 39–52.
Nestler EJ, Terwilliger RZ, Walker JR, Sevarino KA, Duman RS (1990). Chronic cocaine treatment decreases levels of G protein subunits Gi α and Go α in discrete regions of rat brain. J Neurochem 55: 1079–1082.
Niaura R, Shadel WG, Abrams DB, Monti PM, Rohsenow DJ, Sirota A (1998). Individual differences in cue reactivity among smokers trying to quit: effects of gender and cue type. Addict Behav 23: 209–224.
Olpe HR, Koella WP, Wolf P, Haas HL (1977). The action of baclofen on neurons of the substantia nigra and of the ventral tegmental area. Brain Res 134: 577–580.
Panagis G, Kastellakis A (2002). The effects of ventral tegmental administration of GABAA, GABAB, NMDA and AMPA receptor agonists in ventral pallidum self-stimulation. Behav Brain Res 131: 115–123.
Paterson NE, Froestl W, Markou A (2004). The GABAB receptor agonists baclofen and CGP44532 decreased nicotine self-administration in the rat. Psychopharmacology 172: 179–186.
Paterson NE, Markou A (2002). Increased GABA neurotransmission via administration of gamma-vinyl GABA decreased nicotine self-administration in the rat. Synapse 44: 252–253.
Rahman S, McBride WJ (2002). Involvement of GABA and cholinergic receptors in the nucleus accumbens on feedback control of somatodendritic dopamine release in the ventral tegmental area. J Neurochem 80: 646–654.
Ranaldi R, Poeggel K (2002). Baclofen decreases methamphetamine self-administration in rats. Neuroreport 13: 1107–1110.
Roberts DCS, Andrews MM, Vickers GJ (1996). Baclofen attenuates the reinforcing effects of cocaine in rats. Neuropsychopharmacology 15: 417–423.
Rose JE, Behm FM, Levin ED (1993). Role of nicotine dose and sensory cues in the regulation of smoke intake. Pharmacol Biochem Behav 44: 891–900.
Rose JE, Tashkin DP, Ertle A, Zinser MC, Lafer R (1985). Sensory blockade of smoking satisfaction. Pharmacol Biochem Behav 23: 289–293.
See RE (2002). Neural substrates of conditioned-cued relapse to drug-seeking behavior. Pharmacol Biochem Behav 71: 517–529.
See RE, Grimm JW, Kruzich PJ, Rustay N (1999). The importance of a compound stimulus in conditioned drug-seeking behavior following one week of extinction from self-administered cocaine in rats. Drug Alcohol Depend 57: 41–49.
See RE, Kruzich PJ, Grimm JW (2001). Dopamine, but not glutamate, receptor blockade in the basolateral amygdala attenuates conditioned reward in a rat model of relapse to cocaine-seeking behavior. Psychopharmacology 154: 301–310.
Shaham Y, Adamson LK, Grocki S, Corrigall WA (1997). Reinstatement and spontaneous recovery of nicotine seeking in rats. Psychopharmacology 130: 396–403.
Shaham Y, Shalev U, Lu L, De Wit H, Stewart J (2003). The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology 168: 3–20.
Shoaib M, Swanner LS, Beyer CE, Goldberg SR, Schindler CW (1998). The GABAB agonist baclofen modifies cocaine self-administration in rats. Behav Pharmacol 9: 195–206.
Shoptaw S, Yang X, Rotheram-Fuller EJ, Hsieh Y-CM, Kintaudi PC, Charavustra VC et al (2003). Randomized placebo-controlled trial of baclofen for cocaine dependence: preliminary effects for individuals with chronic patterns of cocaine use. J Clin Psychiatry 64: 1440–1448.
Sivilotti L, Nistri A (1991). GABA receptor mechanisms in the central nervous system. Prog Neurobiol 36: 35–92.
Striplin CD, Kalivas PW (1992). Correlation between behavioral sensitization to cocaine and G protein ADP-ribosylation in the ventral tegmental area. Brain Res 579: 181–186.
Striplin CD, Kalivas PW (1993). Robustness of G protein changes in cocaine sensitization shown with immunoblotting. Synapse 14: 10–15.
Sugita S, Johnson SW, North RA (1992). Synaptic inputs to GABAA and GABAB receptors originate from discrete afferent neurons. Neurosci Lett 134: 207–211.
Walaas I, Fonnum F (1979). The distribution and origin of glutamate decarboxylase and choline acetyltransferase in ventral pallidum and other basal forebrain regions. Brain Res 177: 325–336.
Watkins SS, Epping-Jordan MP, Koob GF, Markou A (1999). Blockade of nicotine self-administration with nicotinic antagonists in rats. Pharmacol Biochem Behav 62: 743–751.
Weiss F, Maldonado-Vlaar CS, Parsons LH, Kerr TM, Smith DL, Ben-Shahar O (2000). Control of cocaine-seeking behavior by drug-associated stimuli in rats: effects on recovery of extinguished operant-responding and extracellular dopamine levels in amygdala and nucleus accumbens. Proc Natl Acad Sci USA 97: 4321–4326.
Westerink BHC, Kwint HF, De Vries JB (1996). The pharmacology of mesolimbic dopamine neurons: a dual-probe microdialysis study in the ventral tegmental area and nucleus accumbens of the rat brain. J Neurosci 16: 2605–2611.
Wetherington JP, Lambert NA (2002). GABA(B) receptor activation desensitizes postsynaptic GABA(B) and A(1) adenosine responses in rat hippocampal neurones. J Physiol 544 (Part 2): 459–467.
Willick ML, Kokkinidis L (1995). The effects of ventral tegmental administration of GABAA, GABAB and NMDA receptor agonists on medial forebrain bundle self-stimulation. Behav Brain Res 70: 31–36.
Winer BJ (1971). Statistical Principles in Experimental Design, 2nd edn. McGraw-Hill: New York.
Wirtshafter D, Sheppard AC (2001). Localization of GABAB receptors in midbrain monoamine containing neurons in the rat. Brain Res Bull 56: 1–5.
Xi Z-X, Ramamoorthy S, Shen H, Lake R, Samuvel DJ, Kalivas PW (2003). GABA transmission in the nucleus accumbens is altered after withdrawal from repeated cocaine. J Neurosci 23: 3498–3505.
Xi Z-X, Stein EA (1999). Baclofen inhibits heroin self-administration behavior and mesolimbic dopamine release. J Pharmacol Exp Ther 290: 1369–1374.
Zhang K, Tarazi FI, Campbell A, Baldessarini RJ (2000). GABAB receptors: altered coupling to G-proteins in rats sensitized to amphetamine. Neuroscience 101: 5–10.
Acknowledgements
This is publication number 16367-NP from The Scripps Research Institute. We express our gratitude to Robert Lintz for technical support and Michael Arends for editorial assistance. This study was supported by NIMH/NIDA Grant U01 MH69062, NIDA Grant DA11946, and a Novartis Research Grant to AM.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paterson, N., Froestl, W. & Markou, A. Repeated Administration of the GABAB Receptor Agonist CGP44532 Decreased Nicotine Self-Administration, and Acute Administration Decreased Cue-Induced Reinstatement of Nicotine-Seeking in Rats. Neuropsychopharmacol 30, 119–128 (2005). https://doi.org/10.1038/sj.npp.1300524
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.npp.1300524
Keywords
This article is cited by
-
Dorsomedial prefrontal cortex neurons encode nicotine-cue associations
Neuropsychopharmacology (2019)
-
KK-92A, a novel GABAB receptor positive allosteric modulator, attenuates nicotine self-administration and cue-induced nicotine seeking in rats
Psychopharmacology (2017)
-
Endogenous opioid system: a promising target for future smoking cessation medications
Psychopharmacology (2017)
-
Attenuation of nicotine-taking and nicotine-seeking behavior by the mGlu2 receptor positive allosteric modulators AZD8418 and AZD8529 in rats
Psychopharmacology (2016)
-
GABA Levels in The Dorsal Anterior Cingulate Cortex Associated with Difficulty Ignoring Smoking-Related Cues in Tobacco-Dependent Volunteers
Neuropsychopharmacology (2013)