Abstract
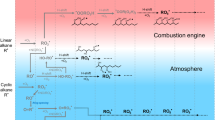
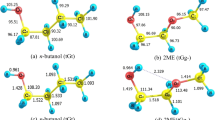
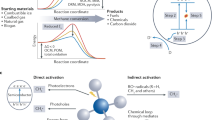
THE observations referred to by Prof. W. A. Bone in NATURE of Aug. 1 (p. 188) with regard to the source of alcohols in the combustion products of paraffin hydrocarbons were based on experimental evidence accumulated in Great Britain and other countries from the point of view of the peroxide mechanism of combustion; compare, for example, in the case of methane, the work of Wartenburg and Sieg,1 who concluded that methane first forms the moloxide CH4(O2). It is interesting to note that the earlier supporters of the peroxide theory of oxidation had discovered the presence of alcohols in the oxidation products of hydrocarbons, but it was considered that these alcohols had been derived directly from the peroxides or from the esters2 and not at all from a primary hydroxylation process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ber. 53, 2192; 1920. A short bibliography of the subject of the oxidation of fuel vapours is given in R. and M. (Air Ministry), 1374; 1930.
Grün, Ber., 53, 987; 1920: Kelber, Ber., 53, 1567; 1920.
Callendar and others, Engineering, R. and M. (Air Ministry), 1092; 1926.
Mardles, J.C.S., 872; 1928.
Comptes rendus, 19, 158; 1931. Ann. des Comb. Liquides, 5, 915; 1930.
Mardles, J. C. S., 872; 1928. Gill, Mardles, and Tett, Trans. Far. Soc., 24, 574; 1928. Brunner, Helvetica Chemica Acta, 13, 197; 1930.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MARDLES, E. The Slow Combustion of Methane and Ethane. Nature 128, 304–305 (1931). https://doi.org/10.1038/128304b0
Issue Date:
DOI: https://doi.org/10.1038/128304b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.