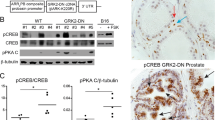
Abstract
The insulin-like growth factor-1 (IGF-1) signaling axis is important for cell growth, differentiation and survival and increased serum IGF is a risk factor for prostate and other cancers. To study IGF-1 action on the prostate, we created transgenic (PB-Des) mice that specifically express human IGF-1des in prostate epithelial cells. This encodes a mature isoform of IGF-1 with decreased affinity for IGF binding proteins (IGFBP) due to a 3-amino acid deletion in the N terminus. Expression of IGF-1des was sufficient to cause hyperplastic lesions in all mice, however the well-differentiated lesions did not progress to adenocarcinoma within a year. Remarkably, crossing the PB-Des mice to an established model of prostate cancer delayed progression of organ-confined tumors and emergence of metastatic lesions in young mice. While dissemination of metastatic lesions was widespread in old bigenic mice we did not detect IGF-1des in poorly differentiated primary tumors or metastatic lesions. Expression of endogenous IGF-1 and levels of P-Akt and P-Erk were reduced independent of age. These data suggest that increased physiologic levels of IGF-1 facilitate the emergence of hyperplastic lesions while imposing a strong IGF-1-dependent differentiation block. Selection against IGF-1 action appears requisite for progression of localized disease and metastogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Ahlen J, Wejde J, Brosjo O, von Rosen A, Weng WH, Girnita L et al. (2005). Insulin-like growth factor type 1 receptor expression correlates to good prognosis in highly malignant soft tissue sarcoma. Clin Cancer Res 11: 206–216.
Bendall SC, Stewart MH, Menendez P, George D, Vijayaragavan K, Werbowetski-Ogilvie T et al. (2007). IGF and FGF cooperatively establish the regulatory stem cell niche of pluripotent human cells in vitro. Nature 448: 1015–1021.
Burger PE, Xiong X, Coetzee S, Salm SN, Moscatelli D, Goto K et al. (2005). Sca-1 expression identifies stem cells in the proximal region of prostatic ducts with high capacity to reconstitute prostatic tissue. Proc Natl Acad Sci USA 102: 7180–7185.
Byrne RL, Leung H, Neal DE . (1996). Peptide growth factors in the prostate as mediators of stromal epithelial interaction. Br J Urol 77: 627–633.
Carboni JM, Lee AV, Hadsell DL, Rowley BR, Lee FY, Bol DK et al. (2005). Tumor development by transgenic expression of a constitutively active insulin-like growth factor I receptor. Cancer Res 65: 3781–3787.
Chan JM, Stampfer MJ, Giovannucci E, Gann PH, Ma J, Wilkinson P et al. (1998). Plasma insulin-like growth factor-I and prostate cancer risk: a prospective study. Science 279: 563–566.
Chan JM, Stampfer MJ, Ma J, Gann P, Gaziano JM, Pollak M et al. (2002). Insulin-like growth factor-I (IGF-I) and IGF binding protein-3 as predictors of advanced-stage prostate cancer. J Natl Cancer Inst 94: 1099–1106.
Chirgwin JM, Przybyla AE, MacDonald RJ, Rutter WJ . (1979). Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry 18: 5294–5299.
Chott A, Sun Z, Morganstern D, Pan J, Li T, Susani M et al. (1999). Tyrosine kinases expressed in vivo by human prostate cancer bone marrow metastases and loss of the type 1 insulin-like growth factor receptor. Am J Pathol 155: 1271–1279.
Colao A, Marzullo P, Spiezia S, Ferone D, Giaccio A, Cerbone G et al. (1999). Effect of growth hormone (GH) and insulin-like growth factor I on prostate diseases: an ultrasonographic and endocrine study in acromegaly, GH deficiency, and healthy subjects. J Clin Endocrinol Metab 84: 1986–1991.
Cunha GR, Donjacour AA . (1989). Mesenchymal-epithelial interactions in the growth and development of the prostate. Cancer Treat Res 46: 159–175.
Damon SE, Plymate SR, Carroll JM, Sprenger CC, Dechsukhum C, Ware JL et al. (2001). Transcriptional regulation of insulin-like growth factor-I receptor gene expression in prostate cancer cells. Endocrinology 142: 21–27.
DiGiovanni J, Bol DK, Wilker E, Beltran L, Carbajal S, Moats S et al. (2000a). Constitutive expression of insulin-like growth factor-1 in epidermal basal cells of transgenic mice leads to spontaneous tumor promotion. Cancer Res 60: 1561–1570.
DiGiovanni J, Kiguchi K, Frijhoff A, Wilker E, Bol DK, Beltran L et al. (2000b). Deregulated expression of insulin-like growth factor 1 in prostate epithelium leads to neoplasia in transgenic mice. Proc Natl Acad Sci USA 97: 3455–3460.
Evangelou AI, Winter SF, Huss WJ, Bok RA, Greenberg NM . (2004). Steroid hormones, polypeptide growth factors, hormone refractory prostate cancer, and the neuroendocrine phenotype. J Cell Biochem 91: 671–683.
Forbes K, Gillette K, Kelley LA, Sehgal I . (2004). Increased levels of urokinase plasminogen activator receptor in prostate cancer cells derived from repeated metastasis. World J Urol 22: 67–71.
Foulstone E, Prince S, Zaccheo O, Burns JL, Harper J, Jacobs C et al. (2005). Insulin-like growth factor ligands, receptors, and binding proteins in cancer. J Pathol 205: 145–153.
Gingrich JR, Barrios RJ, Morton RA, Boyce BF, DeMayo FJ, Finegold MJ et al. (1996). Metastatic prostate cancer in a transgenic mouse. Cancer Res 56: 4096–4102.
Greenberg NM, DeMayo F, Finegold MJ, Medina D, Tilley WD, Aspinall JO et al. (1995). Prostate cancer in a transgenic mouse. Proc Natl Acad Sci USA 92: 3439–3443.
Greenberg NM, DeMayo FJ, Sheppard PC, Barrios R, Lebovitz R, Finegold M et al. (1994). The rat probasin gene promoter directs hormonally and developmentally regulated expression of a heterologous gene specifically to the prostate in transgenic mice. Mol Endocrinol 8: 230–239.
Hadsell DL, Greenberg NM, Fligger JM, Baumrucker CR, Rosen JM . (1996). Targeted expression of des(1-3) human insulin-like growth factor I in transgenic mice influences mammary gland development and IGF-binding protein expression. Endocrinology 137: 321–330.
Hadsell DL, Murphy KL, Bonnette SG, Reece N, Laucirica R, Rosen JM . (2000). Cooperative interaction between mutant p53 and des(1-3)IGF-I accelerates mammary tumorigenesis. Oncogene 19: 889–898.
Hudson DL . (2004). Epithelial stem cells in human prostate growth and disease. Prostate Cancer Prostatic Dis 7: 188–194.
Huss WJ, Gray DR, Greenberg NM, Mohler JL, Smith GJ . (2005). Breast cancer resistance protein-mediated efflux of androgen in putative benign and malignant prostate stem cells. Cancer Res 65: 6640–6650.
Kaplan PJ, Mohan S, Cohen P, Foster BA, Greenberg NM . (1999). The insulin-like growth factor axis and prostate cancer: lessons from the transgenic adenocarcinoma of mouse prostate (TRAMP) model. Cancer Res 59: 2203–2209.
Kaplan-Lefko PJ, Chen TM, Ittmann MM, Barrios RJ, Ayala GE, Huss WJ et al. (2003). Pathobiology of autochthonous prostate cancer in a pre-clinical transgenic mouse model. Prostate 55: 219–237.
Kwabi-Addo B, Giri D, Schmidt K, Podsypanina K, Parsons R, Greenberg N et al. (2001). Haploinsufficiency of the PTEN tumor suppressor gene promotes prostate cancer progression. Proc Natl Acad Sci USA 98: 11563–11568.
Le Roith D . (2003). The insulin-like growth factor system. Exp Diabesity Res 4: 205–212.
Le Roith D, Roberts Jr CT . (2003). The insulin-like growth factor system and cancer. Cancer Lett 195: 127–137.
MacGrogan D, Bookstein R . (1997). Tumour suppressor genes in prostate cancer. Semin Cancer Biol 8: 11–19.
Majeed N, Blouin MJ, Kaplan-Lefko PJ, Barry-Shaw J, Greenberg NM, Gaudreau P et al. (2005). A germ line mutation that delays prostate cancer progression and prolongs survival in a murine prostate cancer model. Oncogene 24: 4736–4740.
Mehrian-Shai R, Chen CD, Shi T, Horvath S, Nelson SF, Reichardt JK et al. (2007). Insulin growth factor-binding protein 2 is a candidate biomarker for PTEN status and PI3K/Akt pathway activation in glioblastoma and prostate cancer. Proc Natl Acad Sci USA 104: 5563–5568.
Nakamura M, Miyamoto S, Maeda H, Zhang SC, Sangai T, Ishii G et al. (2004). Low levels of insulin-like growth factor type 1 receptor expression at cancer cell membrane predict liver metastasis in Dukes' C human colorectal cancers. Clin Cancer Res 10: 8434–8441.
Oh Y, Muller HL, Lee DY, Fielder PJ, Rosenfeld RG . (1993). Characterization of the affinities of insulin-like growth factor (IGF)-binding proteins 1–4 for IGF-I, IGF-II, IGF-I/insulin hybrid, and IGF-I analogs. Endocrinology 132: 1337–1344.
Platz EA, Pollak MN, Leitzmann MF, Stampfer MJ, Willett WC, Giovannucci E . (2005). Plasma insulin-like growth factor-1 and binding protein-3 and subsequent risk of prostate cancer in the PSA era. Cancer Causes Control 16: 255–262.
Plymate SR, Tennant MK, Culp SH, Woodke L, Marcelli M, Colman I et al. (2004). Androgen receptor (AR) expression in AR-negative prostate cancer cells results in differential effects of DHT and IGF-I on proliferation and AR activity between localized and metastatic tumors. Prostate 61: 276–290.
Pollak MN, Schernhammer ES, Hankinson SE . (2004). Insulin-like growth factors and neoplasia. Nat Rev Cancer 4: 505–518.
Reiss K, Wang JY, Romano G, Furnari FB, Cavenee WK, Morrione A et al. (2000). IGF-I receptor signaling in a prostatic cancer cell line with a PTEN mutation. Oncogene 19: 2687–2694.
Renehan AG, Zwahlen M, Minder C, O'Dwyer ST, Shalet SM, Egger M . (2004). Insulin-like growth factor (IGF)-I, IGF binding protein-3, and cancer risk: systematic review and meta-regression analysis. Lancet 363: 1346–1353.
Schnarr B, Strunz K, Ohsam J, Benner A, Wacker J, Mayer D . (2000). Down-regulation of insulin-like growth factor-I receptor and insulin receptor substrate-1 expression in advanced human breast cancer. Int J Cancer 89: 506–513.
Shaneyfelt T, Husein R, Bubley G, Mantzoros CS . (2000). Hormonal predictors of prostate cancer: a meta-analysis. J Clin Oncol 18: 847–853.
Shappell SB, Thomas GV, Roberts RL, Herbert R, Ittmann MM, Rubin MA et al. (2004). Prostate pathology of genetically engineered mice: definitions and classification. The consensus report from the Bar Harbor meeting of the Mouse Models of Human Cancer Consortium Prostate Pathology Committee. Cancer Res 64: 2270–2305.
Wennbo H, Gebre-Medhin M, Gritli-Linde A, Ohlsson C, Isaksson OG, Tornell J . (1997a). Activation of the prolactin receptor but not the growth hormone receptor is important for induction of mammary tumors in transgenic mice. J Clin Invest 100: 2744–2751.
Wennbo H, Kindblom J, Isaksson OG, Tornell J . (1997b). Transgenic mice overexpressing the prolactin gene develop dramatic enlargement of the prostate gland. Endocrinology 138: 4410–4415.
Wu X, Jin C, Wang F, Yu C, McKeehan WL . (2003). Stromal cell heterogeneity in fibroblast growth factor-mediated stromal-epithelial cell cross-talk in premalignant prostate tumors. Cancer Res 63: 4936–4944.
Xin L, Lawson DA, Witte ON . (2005). The Sca-1 cell surface marker enriches for a prostate-regenerating cell subpopulation that can initiate prostate tumorigenesis. Proc Natl Acad Sci USA 102: 6942–6947.
Yakar S, Pennisi P, Wu Y, Zhao H, LeRoith D . (2005). Clinical relevance of systemic and local IGF-I. Endocr Dev 9: 11–16.
Acknowledgements
We would like to thank Tsuey-Ming Chen for help with immunoblotting, Diane Gentry and Caroline Castile for animal care and histology support and Jeffrey M Rosen and Stephen Plymate for their many helpful insights and constructive commentaries. This work was supported by grants from the National Cancer Institute CA82807 and CA84296 (to NMG), CA74589 (to PJK-L), and The Prostate Cancer Foundation (to PJK-L and NMG).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Kaplan-Lefko, P., Sutherland, B., Evangelou, A. et al. Enforced epithelial expression of IGF-1 causes hyperplastic prostate growth while negative selection is requisite for spontaneous metastogenesis. Oncogene 27, 2868–2876 (2008). https://doi.org/10.1038/sj.onc.1210943
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210943
Keywords
This article is cited by
-
Simvastatin inhibits prostatic hyperplasia in rats with metabolic syndrome
International Urology and Nephrology (2022)
-
KLF5 inhibits STAT3 activity and tumor metastasis in prostate cancer by suppressing IGF1 transcription cooperatively with HDAC1
Cell Death & Disease (2020)
-
Circulating IGF-1 promotes prostate adenocarcinoma via FOXO3A/BIM signaling in a double-transgenic mouse model
Oncogene (2019)
-
Modeling prostate cancer in mice: something old, something new, something premalignant, something metastatic
Cancer and Metastasis Reviews (2013)
-
Developmental and oncogenic effects of Insulin-like Growth Factor-I in Ptc1+/- mouse cerebellum
Molecular Cancer (2010)