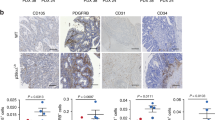
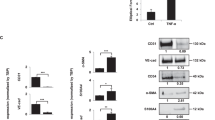
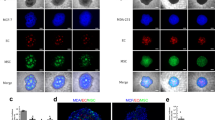
Abstract
Tumor microenvironment in carcinomas recruits mesenchymal cells with an abnormal proangiogenic and invasive phenotype. It is not clear whether mesenchymal tumor cells (MTCs) derive from the activation of mature fibroblasts or from their stem cell precursors. However, stromal cell activation in tumors resembles in several aspects the mesenchymal rearrangement which normally occurs during reparative processes such as wound healing. Mesenchymal stem cells (MSCs) play a crucial role in developmental and reparative processes and have extraordinary proangiogenic potential, on the basis of which they are thought to show great promise for the treatment of ischemic disorders. Here, we show that MTCs have proangiogenic potential and that they share the transcriptional expression of the best-known proangiogenic factors with MSCs. We also found that MTCs and MSCs have the same molecular signature for stemness-related genes, and that when co-implanted with cancer cells in syngeneic animals MSCs determine early tumor appearance, probably by favoring the angiogenic switch. Our data (1) reveal crucial aspects of the proangiogenic phenotype of MTCs, (2) strongly suggest their stem origin and (3) signal the risk of therapeutic use of MSCs in tumor-promoting conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Al Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF . (2003). Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA 100: 3983–3988.
Al Sayed AD, El Weshi AN, Tulbah AM, Rahal MM, Ezzat AA . (2006). Metaplastic carcinoma of the breast clinical presentation, treatment results and prognostic factors. Acta Oncol 45: 188–195.
Bhowmick NA, Neilson EG, Moses HL . (2004). Stromal fibroblasts in cancer initiation and progression. Nature 432: 332–337.
Bonnet D, Dick JE . (1997). Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 3: 730–737.
Burns JS, Abdallah BM, Guldberg P, Rygaard J, Schroder HD, Kassem M . (2005). Tumorigenic heterogeneity in cancer stem cells evolved from long-term cultures of telomerase-immortalized human mesenchymal stem cells. Cancer Res 65: 3126–3135.
Cao Y, Sun Z, Liao L, Meng Y, Han Q, Zhao RC . (2005). Human adipose tissue-derived stem cells differentiate into endothelial cells in vitro and improve postnatal neovascularization in vivo. Biochem Biophys Res Commun 332: 370–379.
De Wever O, Mareel M . (2003). Role of tissue stroma in cancer cell invasion. J Pathol 200: 429–447.
Dingli D, Michor F . (2006). Successful therapy must eradicate cancer stem cells. Stem Cells 24: 2603–2610.
Direkze NC, Hodivala-Dilke K, Jeffery R, Hunt T, Poulsom R, Oukrif D et al. (2004). Bone marrow contribution to tumor-associated myofibroblasts and fibroblasts. Cancer Res 64: 8492–8495.
Dvorak HF . (1986). Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N Engl J Med 315: 1650–1659.
Feinberg AP . (2007). Phenotypic plasticity and the epigenetics of human disease. Nature 447: 433–440.
Fiegl H, Millinger S, Goebel G, Muller-Holzner E, Marth C, Laird PW et al. (2006). Breast cancer DNA methylation profiles in cancer cells and tumor stroma: association with HER-2/neu status in primary breast cancer. Cancer Res 66: 29–33.
Folkman J, Watson K, Ingber D, Hanahan D . (1989). Induction of angiogenesis during the transition from hyperplasia to neoplasia. Nature 339: 58–61.
Galie M, D’Onofrio M, Montani M, Amici A, Calderan L, Marzola P et al. (2005a). Tumor vessel compression hinders perfusion of ultrasonographic contrast agents. Neoplasia 7: 528–536.
Galie M, Sorrentino C, Montani M, Micossi L, Di Carlo E, D’Antuono T et al. (2005b). Mammary carcinoma provides highly tumourigenic and invasive reactive stromal cells. Carcinogenesis 26: 1868–1878.
Galli R, Binda E, Orfanelli U, Cipelletti B, Gritti A, De Vitis S et al. (2004). Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res 64: 7011–7021.
Hayward SW, Wang Y, Cao M, Hom YK, Zhang B, Grossfeld GD et al. (2001). Malignant transformation in a nontumorigenic human prostatic epithelial cell line. Cancer Res 61: 8135–8142.
Hill R, Song Y, Cardiff RD, Van Dyke T . (2005). Selective evolution of stromal mesenchyme with p53 loss in response to epithelial tumorigenesis. Cell 123: 1001–1011.
Holmgren L, O’Reilly MS, Folkman J . (1995). Dormancy of micrometastases: balanced proliferation and apoptosis in the presence of angiogenesis suppression. Nat Med 1: 149–153.
Hu M, Yao J, Cai L, Bachman KE, van den BF, Velculescu V et al. (2005). Distinct epigenetic changes in the stromal cells of breast cancers. Nat Genet 37: 899–905.
Hung SC, Deng WP, Yang WK, Liu RS, Lee CC, Su TC et al (2005). Mesenchymal stem cell targeting of microscopic tumors and tumor stroma development monitored by noninvasive in vivo positron emission tomography imaging. Clin Cancer Res 11: 7749–7756.
Keith WN . (2004). From stem cells to cancer: balancing immortality and neoplasia. Oncogene 23: 5092–5094.
Knutson KL, Lu H, Stone B, Reiman JM, Behrens MD, Prosperi CM et al. (2006). Immunoediting of cancers may lead to epithelial to mesenchymal transition. J Immunol 177: 1526–1533.
Krampera M, Marconi S, Pasini A, Galie M, Rigotti G, Mosna F et al. (2007). Induction of neural-like differentiation in human mesenchymal stem cells derived from bone marrow, fat, spleen and thymus. Bone 40: 382–390.
Kurose K, Gilley K, Matsumoto S, Watson PH, Zhou XP, Eng C . (2002). Frequent somatic mutations in PTEN and TP53 are mutually exclusive in the stroma of breast carcinomas. Nat Genet 32: 355–357.
Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J et al. (1994). A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 367: 645–648.
Li C, Heidt DG, Dalerba P, Burant CF, Zhang L, Adsay V et al. (2007). Identification of pancreatic cancer stem cells. Cancer Res 67: 1030–1037.
McAlhany SJ, Ressler SJ, Larsen M, Tuxhorn JA, Yang F, Dang TD et al. (2003). Promotion of angiogenesis by ps20 in the differential reactive stroma prostate cancer xenograft model. Cancer Res 63: 5859–5865.
Miranville A, Heeschen C, Sengenes C, Curat CA, Busse R, Bouloumie A . (2004). Improvement of postnatal neovascularization by human adipose tissue-derived stem cells. Circulation 110: 349–355.
Moody SE, Perez D, Pan TC, Sarkisian CJ, Portocarrero CP, Sterner CJ et al. (2005). The transcriptional repressor Snail promotes mammary tumor recurrence. Cancer Cell 8: 197–209.
Naumov GN, Akslen LA, Folkman J . (2006). Role of angiogenesis in human tumor dormancy: animal models of the angiogenic switch. Cell Cycle 5: 1779–1787.
Orimo A, Gupta PB, Sgroi DC, Arenzana-Seisdedos F, Delaunay T, Naeem R et al. (2005). Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121: 335–348.
Pennacchietti S, Michieli P, Galluzzo M, Mazzone M, Giordano S, Comoglio PM . (2003). Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell 3: 347–361.
Petersen OW, Lind NH, Gudjonsson T, Villadsen R, Ronnov-Jessen L, Bissell MJ . (2001). The plasticity of human breast carcinoma cells is more than epithelial to mesenchymal conversion. Breast Cancer Res 3: 213–217.
Planat-Benard V, Silvestre JS, Cousin B, Andre M, Nibbelink M, Tamarat R et al. (2004). Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation 109: 656–663.
Ponti D, Costa A, Zaffaroni N, Pratesi G, Petrangolini G, Coradini D et al. (2005). Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res 65: 5506–5511.
Reya T, Morrison SJ, Clarke MF, Weissman IL . (2001). Stem cells, cancer, and cancer stem cells. Nature 414: 105–111.
Rigotti G, Marchi A, Galie M, Baroni G, Benati D, Krampera M et al. (2007). Clinical treatment of radiotherapy tissue damages by lipoaspirates transplant: a healing process mediated by adipose-derived adult stem cells (ASCs). Plast Reconstr Surg 119: 1409–1422.
Ronnov-Jessen L, Petersen OW, Bissell MJ . (1996). Cellular changes involved in conversion of normal to malignant breast: importance of the stromal reaction. Physiol Rev 76: 69–125.
Rubio D, Garcia-Castro J, Martin MC, de la FR, Cigudosa JC, Lloyd AC et al. (2005). Spontaneous human adult stem cell transformation. Cancer Res 65: 3035–3039.
Shipitsin M, Campbell LL, Argani P, Weremowicz S, Bloushtain-Qimron N, Yao J et al. (2007). Molecular definition of breast tumor heterogeneity. Cancer Cell 11: 259–273.
Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T et al. (2004). Identification of human brain tumour initiating cells. Nature 432: 396–401.
Thiery JP, Sleeman JP . (2006). Complex networks orchestrate epithelial–mesenchymal transitions. Nat Rev Mol Cell Biol 7: 131–142.
Wolf D, Rumpold H, Koeck R, Gunsilius E . (2005). Mesenchymal stem cells: potential precursors for tumor stroma and targeted-delivery vehicles for anticancer agents. J Natl Cancer Inst 97: 540–541.
Yang F, Tuxhorn JA, Ressler SJ, McAlhany SJ, Dang TD, Rowley DR . (2005). Stromal expression of connective tissue growth factor promotes angiogenesis and prostate cancer tumorigenesis. Cancer Res 65: 8887–8895.
Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H et al. (2002). Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13: 4279–4295.
Acknowledgements
This work was supported by grants from the Fondazione Cassa di Risparmio di Verona, Vicenza e Belluno and the Ministero dell’Università e della Ricerca Scientifica.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Galiè, M., Konstantinidou, G., Peroni, D. et al. Mesenchymal stem cells share molecular signature with mesenchymal tumor cells and favor early tumor growth in syngeneic mice. Oncogene 27, 2542–2551 (2008). https://doi.org/10.1038/sj.onc.1210920
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210920
Keywords
This article is cited by
-
Obesity and prostate cancer — microenvironmental roles of adipose tissue
Nature Reviews Urology (2023)
-
MicroRNA-100 shuttled by mesenchymal stem cell-derived exosomes suppresses in vitro angiogenesis through modulating the mTOR/HIF-1α/VEGF signaling axis in breast cancer cells
Cellular Oncology (2017)
-
Polyphenol-rich strawberry extract (PRSE) shows in vitro and in vivo biological activity against invasive breast cancer cells
Scientific Reports (2016)
-
CXCL1 mediates obesity-associated adipose stromal cell trafficking and function in the tumour microenvironment
Nature Communications (2016)
-
Resveratrol reduces IL-6 and VEGF secretion from co-cultured A549 lung cancer cells and adipose-derived mesenchymal stem cells
Tumor Biology (2016)