Abstract
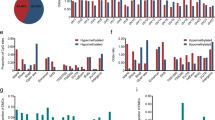
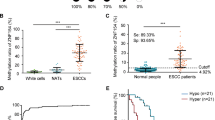
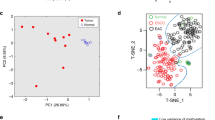
Epigenetic alterations and the resulting inactivation of tumor suppressor genes often contribute to the development of various cancers. To identify novel candidates that may be silenced by aberrant methylation in esophageal squamous-cell carcinoma (ESCC), we analysed ESCC cell lines by a recently developed method known as bacterial artificial chromosome array-based methylated CpG island amplification (BAMCA), and selected candidates through BAMCA-assisted strategy. In the course of this program, we identified frequent CpG methylation-dependent silencing of the gene encoding cellular retinoic acid binding protein 1 (CRABP1) in our panel of ESCC cell lines. Expression of CRABP1 mRNA was restored in gene-silenced ESCC cells after treatment with 5-aza 2′-deoxycytidine. The DNA methylation status of the CRABP1 CpG island with clear promoter activity correlated inversely with expression of this gene. CpG methylation of CRABP1 was frequently observed in primary ESCC tissues as well. Restoration of CRABP1 expression in ESCC cells lacking the protein reduced cell growth by inducing arrest at G0–G1, whereas knockdown of the gene in cells expressing CRABP1 promoted cell growth. Among 113 primary ESCC tumors, the absence of immunoreactive CRABP1 was significantly associated with de-differentiation of cancer cells and with distant lymph-node metastases in the patients. These results indicate that CRABP1 appears to have a tumor-suppressor function in esophageal epithelium, and its epigenetic silencing may play a pivotal role during esophageal carcinogenesis. Its expression status in biopsies or resected tumors might serve as an index for identifying ESCC patients for whom combined therapeutic modalities would be recommended.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
de Bruijn DR, Oerlemans F, Hendriks W, Baats E, Ploemacher R, Wieringa B et al. (1994). Normal development, growth and reproduction in cellular retinoic acid binding protein-I (CRABPI) null mutant mice. Differentiation 58: 141–148.
Dong D, Ruuska SE, Levinthal DJ, Noy N . (1999). Distinct roles for cellular retinoic acid-binding proteins I and II in regulating signaling by retinoic acid. J Biol Chem 274: 23695–23698.
Donovan M, Olofsson B, Gustafson AL, Dencker L, Eriksson U . (1995). The cellular retinoic acid binding proteins. J Steroid Biochem Mol Biol 53: 459–465.
Dowlatshahi K, Mehta RG, Levin B, Cerny WL, Skinner DB, Moon RC . (1984). Retinoic-acid-binding protein in normal and neoplastic human esophagus. Cancer 54: 308–311.
Egger G, Liang G, Aparicio A, Jones PA . (2004). Epigenetics in human disease and prospects for epigenetic therapy. Nature 429: 457–463.
Fiorella PD, Giguere V, Napoli JL . (1993). Expression of cellular retinoic acid-binding protein (type II) in Escherichia coli. Characterization and comparison to cellular retinoic acid-binding protein (type I). J Biol Chem 268: 21545–21552.
Gorry P, Lufkin T, Dierich A, Rochette-Egly C, Decimo D, Dolle P et al. (1994). The cellular retinoic acid binding protein I is dispensable. Proc Natl Acad Sci USA 91: 9032–9036.
Hawthorn L, Stein L, Varma R, Wiseman S, Loree T, Tan D . (2004). TIMP1 and SERPIN-A overexpression and TFF3 and CRABP1 underexpression as biomarkers for papillary thyroid carcinoma. Head Neck 26: 1069–1083.
Herman JG, Baylin SB . (2003). Gene silencing in cancer in association with promoter hypermethylation. N Engl J Med 349: 2042–2054.
Huang Y, de la Chapelle A, Pellegata NS . (2003). Hypermethylation, but not LOH, is associated with the low expression of MT1G and CRABP1 in papillary thyroid carcinoma. Int J Cancer 104: 735–744.
Imoto I, Yang ZQ, Pimkhaokham A, Tsuda H, Shimada Y, Imamura M et al. (2001). Identification of cIAP1 as a candidate target gene within an amplicon at 11q22 in esophageal squamous cell carcinomas. Cancer Res 61: 6629–6634.
Inazawa J, Inoue J, Imoto I . (2004). Comparative genomic hybridization (CGH)-arrays pave the way for identification of novel cancer-related genes. Cancer Sci 95: 559–563.
Ito T, Shimada Y, Hashimoto Y, Kaganoi J, Kan T, Watanabe G et al. (2003). Involvement of TSLC1 in progression of esophageal squamous cell carcinoma. Cancer Res 63: 6320–6326.
Jones PA, Baylin SB . (2002). The fundamental role of epigenetic events in cancer. Nat Rev Genet 3: 415–428.
Kaifi JT, Yekebas EF, Schurr P, Obonyo D, Wachowiak R, Busch P et al. (2005). Tumor-cell homing to lymph nodes and bone marrow and CXCR4 expression in esophageal cancer. J Natl Cancer Inst 97: 1840–1847.
Kim MS, Yamashita K, Baek JH, Park HL, Carvalho AL, Osada M et al. (2006). N-methyl-D-aspartate receptor type 2B is epigenetically inactivated and exhibits tumor-suppressive activity in human esophageal cancer. Cancer Res 66: 3409–3418.
Kuroki T, Trapasso F, Yendamuri S, Matsuyama A, Alder H, Mori M et al. (2003). Allele loss and promoter hypermethylation of VHL, RAR-beta, RASSF1A, and FHIT tumor suppressor genes on chromosome 3p in esophageal squamous cell carcinoma. Cancer Res 63: 3724–3728.
Laird PW . (2003). The power and the promise of DNA methylation markers. Nat Rev Cancer 3: 253–266.
Mandelker DL, Yamashita K, Tokumaru Y, Mimori K, Howard DL, Tanaka Y et al. (2005). PGP9.5 promoter methylation is an independent prognostic factor for esophageal squamous cell carcinoma. Cancer Res 65: 4963–4968.
Means AL, Thompson JR, Gudas LJ . (2000). Transcriptional regulation of the cellular retinoic acid binding protein I gene in F9 teratocarcinoma cells. Cell Growth Differ 11: 71–82.
Misawa A, Inoue J, Sugino Y, Hosoi H, Sugimoto T, Hosoda F et al. (2005). Methylation-associated silencing of the nuclear receptor 1I2 gene in advanced-type neuroblastomas, identified by bacterial artificial chromosome array-based methylated CpG island amplification. Cancer Res 65: 10233–10242.
Miyazaki T, Kato H, Shitara Y, Yoshikawa M, Tajima K, Masuda N et al. (2000). Mutation and expression of the metastasis suppressor gene KAI1 in esophageal squamous cell carcinoma. Cancer 89: 955–962.
Miyazaki T, Kato H, Fukuchi M, Nakajima M, Kuwano H . (2003). EphA2 overexpression correlates with poor prognosis in esophageal squamous cell carcinoma. Int J Cancer 103: 657–663.
Ogino S, Cantor M, Kawasaki T, Brahmandam M, Kirkner G, Weisenberger DJ et al. (2006). CpG island methylator phenotype (CIMP) of colorectal cancer is best characterized by quantitative DNA methylation analysis and prospective cohort studies. Gut 55: 1000–1006.
Ohtsu A . (2004). Chemoradiotherapy for esophageal cancer: current status and perspectives. Int J Clin Oncol 9: 444–450.
Parkin DM, Bray F, Ferlay J, Pisani P . (2005). Global Cancer Statistics, 2002. CA Cancer J Clin 55: 74–108.
Pfoertner S, Goelden U, Hansen W, Toepfer T, Geffers R, Ukena SN et al. (2005). Cellular retinoic acid binding protein I: expression and functional influence in renal cell carcinoma. Tumour Biol 26: 313–323.
Pimkhaokham A, Shimada Y, Fukuda Y, Kurihara N, Imoto I, Yang ZQ et al. (2000). Nonrandom chromosomal imbalances in esophageal squamous cell carcinoma cell lines: possible involvement of the ATF3 and CENPF genes in the 1q32 amplicon. Jpn J Cancer Res 91: 1126–1133.
Roder JD, Busch R, Stein HJ, Fink U, Siewert JR . (1994). Ratio of invaded to removed lymph node are predictors of survival in squamous cell carcinoma of the esophagus. Br J Surg 81: 410–413.
Shibata H, Matsubara O, Wakiyama H, Tanaka S . (2001). The role of cyclin-dependent kinase inhibitor p27 in squamous cell carcinoma of the esophagus. Pathol Res Pract 197: 157–164.
Shibagaki I, Shimada Y, Wagata T, Ikenaga M, Imamura M, Ishizaki K . (1994). Allelotype analysis of esophageal squamous cell carcinoma. Cancer Res 54: 2996–3000.
Shimada Y, Imamura M, Wagata T, Yamaguchi N, Tobe T . (1992). Characterization of 21 newly established esophageal cancer cell lines. Cancer 69: 277–284.
Shirakawa Y, Naomoto Y, Kimura M, Kawashima R, Yamatsuji T, Tamaki T et al. (2000). Topological analysis of p21WAF1/CIP1 expression in esophageal squamous dysplasia. Clin Cancer Res 6: 541–550.
Sobin LH, Wittekind CH (ed). (2002). International Union Against Cancer (UICC): TNM Classification of Malignant Tumors 6th edn. Wiley-Liss: New York.
Sonoda I, Imoto I, Inoue J, Shibata T, Shimada Y, Chin K et al. (2004). Frequent silencing of low density lipoprotein receptor-related protein 1B (LRP1B) expression by genetic and epigenetic mechanisms in esophageal squamous cell carcinoma. Cancer Res 64: 3741–3747.
Stein HJ, Feith M, Bruecher BL, Naehrig J, Sarbia M, Siewert JR . (2005). Early esophageal cancer: pattern of lymphatic spread and prognostic factors for long-term survival after surgical resection. Ann Surg 242: 566–573.
Suzuki H, Gabrielson E, Chen W, Anbazhagan R, van Engeland M, Weijenberg MP et al. (2002). A genomic screen for genes upregulated by demethylation and histone deacetylase inhibition in human colorectal cancer. Nat Genet 31: 141–149.
Toyota M, Ho C, Ahuja N, Jair KW, Li Q, Ohe-Toyota M et al. (1999). Identification of differentially methylated sequences in colorectal cancer by methylated CpG island amplification. Cancer Res 59: 2307–2312.
Venepally P, Reddy LG, Sani BP . (1996). Characterization of cellular retinoic acid-binding protein I from chick embryo and its ligand binding properties. Arch Biochem Biophys 336: 231–239.
Wei LN, Chang L . (1996). Promoter and upstream regulatory activities of the mouse cellular retinoic acid-binding protein-I gene. J Biol Chem 271: 5073–5078.
Wei LN, Lee CH . (1994). Demethylation in the 5′-flanking region of mouse cellular retinoic acid binding protein-I gene is associated with its high level of expression in mouse embryos and facilitates its induction by retinoic acid in P19 embryonal carcinoma cells. Dev Dyn 201: 1–10.
Xiong Z, Laird PW . (1997). COBRA: a sensitive and quantitative DNA methylation assay. Nucleic Acids Res 25: 2532–2534.
Yamashita K, Upadhyay S, Osada M, Hoque MO, Xiao Y, Mori M et al. (2002). Pharmacologic unmasking of epigenetically silenced tumor suppressor genes in esophageal squamous cell carcinoma. Cancer Cell 2: 485–495.
Acknowledgements
This work is supported by Grants-in-aid for scientific research on priority areas and 21st Century center of excellence program for molecular destruction and reconstitution of tooth and bone from the Ministry of Education, Culture, Sports, Science, and Technology, Japan and a grant-in-aid from core research for evolutional science and technology (CREST) of the Japan science and technology corporation (JST).
We are grateful to Professor Yusuke Nakamura (Human Genome Center, Institute of Medical Science, The University of Tokyo) for continuous encouragement throughout this work. We also thank Ayako Takahashi, and Rumi Mori for technical assistance, and Yoriko Fukukawa for secretarial assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Tanaka, K., Imoto, I., Inoue, J. et al. Frequent methylation-associated silencing of a candidate tumor-suppressor, CRABP1, in esophageal squamous-cell carcinoma. Oncogene 26, 6456–6468 (2007). https://doi.org/10.1038/sj.onc.1210459
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210459
Keywords
This article is cited by
-
Common molecular features of H3K27M DMGs and PFA ependymomas map to hindbrain developmental pathways
Acta Neuropathologica Communications (2023)
-
CRABP1-CaMKII-Agrn regulates the maintenance of neuromuscular junction in spinal motor neuron
Cell Death & Differentiation (2022)
-
ACVR1C/SMAD2 signaling promotes invasion and growth in retinoblastoma
Oncogene (2019)
-
CRABP1, C1QL1 and LCN2 are biomarkers of differentiated thyroid carcinoma, and predict extrathyroidal extension
BMC Cancer (2018)
-
The novel 19q13 KRAB zinc-finger tumour suppressor ZNF382 is frequently methylated in oesophageal squamous cell carcinoma and antagonises Wnt/β-catenin signalling
Cell Death & Disease (2018)