Abstract
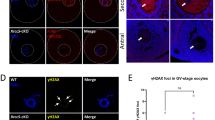
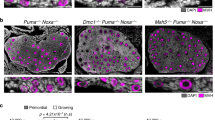
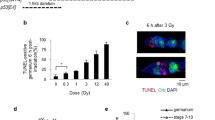
The early stage embryogenesis of higher eukaryotes lacks some of the damage response pathways such as G1/S checkpoint, G2/M checkpoint and apoptosis. We examined here the damage response of preimplantation stage embryos after fertilization with 6 Gy irradiated sperm. Sperm-irradiated embryos developed normally for the first 2.5 days, but started to exhibit a developmental delay at day 3.5. p21 was activated in the delayed embryos, which carried numerous micronuclei owing to delayed chromosome instability. Apoptosis was observed predominantly in the inner cell mass of the day 4.0 embryos. Sperm-irradiated p21−/− embryos lacked the delay, but chromosome instability and apoptosis were more pronounced than the corresponding p21 wild-type embryos. We conclude from the result that damage responses come in a stage-specific manner during preimplantation stage development; p53-dependent S checkpoint at the zygote stage, p21-mediated cell cycle arrest at the morula/blastocyst stages and apoptosis after the blastocyst stage in the inner cell mass.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Baus F, Gire V, Fisher D, Piette J, Dulic V . (2003). Permanent cell cycle exit in G2 phase after DNA damage in normal human fibroblasts. EMBO J 22: 3992–4002.
Clute P, Masui Y . (1997). Microtubule dependence of chromosome cycles in Xenopus laevis blastomeres under the influence of a DNA synthesis inhibitor, aphidicolin. Dev Biol 185: 1–13.
Fenech M . (1993). The cytokinesis-block micronucleus technique and its application to genotoxicity studies in human populations. Environ Health Perspect 101: 101–107.
Fleming TP, Sheth B, Fesenko I . (2001). Cell adhesion in the preimplantation mammalian embryo and its role in trophectoderm differentiation and blastocyst morphogenesis. Front Biosci 6: 2566–2576.
Friesner JD, Liu B, Culligan K, Britt AB . (2005). Ionizing radiation-dependent gamma-H2AX focus formation requires ataxia telangiectasia mutated and ataxia telangiectasia mutated and Rad3-related. Mol Biol Cell 16: D1000–D1007.
Gartel AL, Tyner AL . (2002). The role of the cyclin-dependent kinase inhibitor p21 in apoptosis. Mol Cancer Ther 1: 639–649.
Goldstein LS, Spindle AI, Pedersen RA . (1975). X-ray sensitivity of the preimplantation mouse embryo in vitro. Radiat Res 62: 276–287.
Gottifredi V, McKinney K, Poyurovsky MV, Prives C . (2004). Decreased p21 levels are required for efficient restart of DNA synthesis after S phase block. J Biol Chem 279: 5802–5810.
Hamatani T, Daikoku T, Wang H, Matsumoto H, Carter MG, Ko MS et al. (2004). Global gene expression analysis identifies molecular pathways distinguishing blastocyst dormancy and activation. Proc Natl Acad Sci USA 101: 10326–10331.
Hendzel MJ, Wei Y, Mancini MA, Van Hooser A, Ranalli T, Brinkley BR et al. (1997). Mitosis-specific phosphorylation of histone H3 initiates primarily within pericentromeric heterochromatin during G2 and spreads in an ordered fashion coincident with mitotic chromosome condensation. Chromosoma 106: 348–360.
Heyer BS, MacAuley A, Behrendtsen O, Werb Z . (2000). Hypersensitivity to DNA damage leads to increased apoptosis during early mouse development. Genes Dev 14: 2072–2084.
Ikegami R, Rivera-Bennetts AK, Brooker DL, Yager TD . (1997). Effect of inhibitors of DNA replication on early zebrafish embryos: evidence for coordinate activation of multiple intrinsic cell-cycle checkpoints at the mid-blastula transition. Zygote 5: 153–175.
Iliakis G, Wang H, Perrault AR, Boecker W, Rosidi B, Windhofer F et al. (2004). Mechanisms of DNA double strand break repair and chromosome aberration formation. Cytogenet Genome Res 104: 14–20.
Jascur T, Brickner H, Salles-Passador I, Barbier V, El Khissiin A, Smith B et al. (2005). Regulation of p21(WAF1/CIP1) stability by WISp39, a Hsp90 binding TPR protein. Mol Cell 17: 237–249.
Jeong YJ, Choi HW, Shin HS, Cui XS, Kim NH, Gerton GL et al. (2005). Optimization of real time RT-PCR methods for the analysis of gene expression in mouse eggs and preimplantation embryos. Mol Reprod Dev 71: 284–289.
Jurisicova A, Rogers I, Fasciani A, Casper RF, Varmuza S . (1998). Effect of maternal age and conditions of fertilization on programmed cell death during murine preimplantation embryo development. Mol Hum Reprod 4: 139–145.
Kadhim MA, Macdonald DA, Goodhead DT, Lorimore SA, Marsden SJ, Wright EG . (1992). Transmission of chromosomal instability after plutonium alpha-particle irradiation. Nature 355: 738–740.
Kamjoo M, Brison DR, Kimber SJ . (2002). Apoptosis in the preimplantation mouse embryo: effect of strain difference and in vitro culture. Mol Reprod Dev 61: 67–77.
Liu S, Bishop WR, Liu M . (2003). Differential effects of cell cycle regulatory protein p21(WAF1/Cip1) on apoptosis and sensitivity to cancer chemotherapy. Drug Resist Updat 6: 183–195.
Malashicheva AB, Kislyakova TV, Aksenov ND, Osipov KA, Pospelov VA . (2000). F9 embryonal carcinoma cells fail to stop at G1/S boundary of the cell cycle after gamma-irradiation due to p21WAF1/CIP1 degradation. Oncogene 19: 3858–3865.
Morgan WF . (2003). Non-targeted and delayed effects of exposure to ionizing radiation: I. Radiation-induced genomic instability and bystander effects in vitro. Radiat Res 159: 567–580.
Mukherjee AB . (1976). Cell cycle analysis and X-chromosome inactivation in the developing mouse. Proc Natl Acad Sci USA 73: 1608–1611.
Newport J, Kirschner M . (1982). A major developmental transition in early Xenopus embryos: II. Control of the onset of transcription. Cell 30: 687–696.
Raff JW, Glover DM . (1988). Nuclear and cytoplasmic mitotic cycles continue in Drosophila embryos in which DNA synthesis is inhibited with aphidicolin. J Cell Biol 107: 2009–2019.
Shimura T, Inoue M, Taga M, Shiraishi K, Uematsu N, Takei N et al. (2002a). p53-dependent S-phase damage checkpoint and pronuclear cross talk in mouse zygotes with X-irradiated sperm. Mol Cell Biol 22: 2220–2228.
Shimura T, Toyoshima M, Taga M, Shiraishi K, Uematsu N, Inoue M et al. (2002b). The novel surveillance mechanism of the Trp53-dependent s-phase checkpoint ensures chromosome damage repair and preimplantation-stage development of mouse embryos fertilized with x-irradiated sperm. Radiat Res 158: 735–742.
Taga M, Shiraishi K, Shimura T, Uematsu N, Kato T, Nishimune Y et al. (2000). The effect of caffeine on p53-dependent radioresponses in undifferentiated mouse embryonal carcinoma cells after X-ray and UV-irradiations. J Radiat Res (Tokyo) 41: 227–241.
Taylor WR, Stark GR . (2001). Regulation of the G2/M transition by p53. Oncogene 20: 1803–1815.
Toyoshima M, Shimura T, Adiga SK, Taga M, Shiraishi K, Inoue M et al. (2005). Transcription-independent suppression of DNA synthesis by p53 in sperm-irradiated mouse zygotes. Oncogene 24: 3229–3235.
Wright SJ . (1999). Sperm nuclear activation during fertilization. Curr Top Dev Biol 46: 133–178.
Zeng F, Baldwin DA, Schultz RM . (2004). Transcript profiling during preimplantation mouse development. Dev Biol 272: 483–496.
Zhou BB, Elledge SJ . (2000). The DNA damage response: putting checkpoints in perspective. Nature 408: 433–439.
Ziomek CA, Johnson MH . (1980). Cell surface interaction induces polarization of mouse 8-cell blastomeres at compaction. Cell 21: 935–942.
Acknowledgements
SKA is the recipient of Japan Society for the Promotion of Science (JSPS) fellowship (ID: P02268). This work was supported by Grant-in-Aid from the Ministry of Education, Science, Sports, Culture and Technology (MEXT), Japan to ON The work was also supported by a grant from the Nuclear Safety Research Association, Tokyo.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Adiga, S., Toyoshima, M., Shiraishi, K. et al. p21 provides stage specific DNA damage control to preimplantation embryos. Oncogene 26, 6141–6149 (2007). https://doi.org/10.1038/sj.onc.1210444
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210444
Keywords
This article is cited by
-
Lineage regulators TFAP2C and NR5A2 function as bipotency activators in totipotent embryos
Nature Structural & Molecular Biology (2024)
-
Sperm DNA integrity does play a crucial role for embryo development after ICSI, notably when good-quality oocytes from young donors are used
Biological Research (2022)
-
Sperm DNA damage compromises embryo development, but not oocyte fertilisation in pigs
Biological Research (2022)
-
Pak2 reduction induces a failure of early embryonic development in mice
Reproductive Biology and Endocrinology (2021)
-
Involvement of CDKN1A (p21) in cellular senescence in response to heat and irradiation stress during preimplantation development
Cell Stress and Chaperones (2020)