Abstract
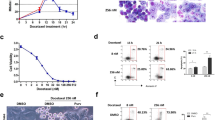
We have previously defined a homozygously deleted region at chromosome 1p36.2–p36.3 in human neuroblastoma cell lines, NB-1 and NB-C201, and identified six genes including DFF45/ICAD within this region. In this study, we found that NB-C201 cells are much more resistant to various genotoxic stresses such as cisplatin (CDDP) than CHP134 and SH-SY5Y cells that do not have the homozygous deletion. To examine a role(s) of DFF45 in the regulation of apoptosis in response to CDDP, we have established stably DFF45-expressing NB-C201 cell clones (DFF45-1 and DFF45-3) and a control cell clone (NB-C201-C) using a retrovirus-mediated gene transfer. In contrast to NB-C201-C cells, DFF45-3 cells displayed apoptotic nuclear fragmentation in response to CDDP. Although CDDP-induced proteolytic cleavage of procaspase-3 and DFF45 in DFF45-3 cells, we could not detect a typical apoptotic DNA fragmentation. Additionally, deletion analysis revealed that C-terminal region of DFF45 is required for inducing nuclear fragmentation. Unexpectedly, (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays demonstrated that DFF45 has undetectable effect on CDDP sensitivity of NB-C201 cells. Taken together, our present results suggest that DFF45/DFF40 system may be sufficient for CDDP-induced nuclear fragmentation but not DNA cleavage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Amler LC, Corvi R, Praml C, Savelyeva L, Le Paslier D, Schwab M . (1995). A reciprocal translocation (1;15)(36.2;q24) in a neuroblastoma cell line is accompanied by DNA duplication and may signal the site of a putative tumor suppressor gene. Oncogene 10: 1095–1101.
Brodeur GM, Seeger RC, Schwab M, Varmas HE, Bishop JM . (1984). Amplification of N-myc in untreated human neuroblastomas corelates with advanced disease stage. Science 224: 1121–1124.
Brodeur GM, Nakagawara A . (1992). Molecular basis of clinical heterogeneity in neuroblastoma. Am J Pediatr Hematol Oncol 14: 111–116.
Cheng NC, Van Roy N, Chan A, Beitsma M, Westerveld A, Speleman F et al. (1995). Deletion mapping in neuroblastoma cell lines suggests two distinct tumor suppressor genes in the 1p35–36 region, only one of which is associated with N-myc amplification. Oncogene 10: 291–297.
Enari M, Sakahira H, Yokoyama H, Okawa K, Iwamatsu A, Nagata S . (1998). A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 391: 43–50.
Gu J, Dong RP, Zhang C, McLaughlin DF, Wu MX, Schlossman SF . (1999). Functional interaction of DFF35 and DFF45 with caspase-activated DNA fragmentation nuclease DFF40. J Biol Chem 274: 20759–20762.
Halenbeck R, MacDonald H, Roulston A, Chen TT, Conroy L, Williams LT . (1998). CPAN, a human nuclease regulated by the caspase-sensitive inhibitor DFF45. Curr Biol 8: 537–540.
Hosoda M, Ozaki T, Miyazaki K, Hayashi S, Furuya S, Watanabe K et al. (2005). UFD2a mediates the proteasomal turnover of p73 without promoting p73 ubiquitination. Oncogene 24: 7156–7169.
Inohara N, Koseki T, Chen S, Benedict MA, Nunez G . (1999). Identification of regulatory and catalytic domains in the apoptosis nuclease DFF40/CAD. J Biol Chem 274: 270–274.
Liu X, Zou H, Slaughter C, Wang X . (1997). DFF, a heterodimeric protein that functions downstream of caspase-3 to trigger DNA fragmentation during apoptosis. Cell 89: 175–184.
Liu X, Li P, Widlak P, Zou H, Luo X, Garrard WT et al. (1998). The 40-kDa subunit of DNA fragmentation factor induces DNA fragmentation and chromatin condensation during apoptosis. Proc Natl Acad Sci USA 95: 8461–8466.
Lu C, Zhu F, Cho YY, Tang F, Zykova T, Ma WY et al. (2006). Cell apoptosis: requirement of H2AX in DNA ladder formation, but not for the activation of caspase-3. Mol Cell 23: 121–132.
Lu Z, Zhang C, Zhai Z . (2005). Nucleoplasmin regulates chromatin condensation during apoptosis. Proc Natl Acad Sci USA 102: 2778–2783.
Martinsson T, Sjoberg RM, Hedborg F, Kogner P . (1995). Deletion of chromosome 1p loci and microsatellite instability in neuroblastomas analyzed with short-tandem repeat polymorphisms. Cancer Res 55: 5681–5686.
Mukae N, Enari M, Sakahira H, Fukuda Y, Inazawa J, Tho H et al. (1998). Molecular cloning and characterization of human caspase- activated DNase. Proc Natl Acad Sci USA 95: 9123–9128.
Ohira M, Kageyama H, Mihara M, Furuta S, Machida T, Shishikura T et al. (2000). Identification and characterization of a 500-kb homozygously deleted region at 1p36.2–p36.3 in a neuroblastoma cell line. Oncogene 19: 4302–4307.
Raff MC . (1992). Social controls on cell survival and cell death. Nature 356: 397–400.
Sakahira H, Enari M, Nagata S . (1998). Cleavage of CAD inhibitor in CAD activation and DNA degradation during apoptosis. Nature 391: 96–99.
Schleiermacher G, Peter M, Michon J, Hugot JP, Vielh P, Zucker JM et al. (1994). Two distinct deleted regions on the short arm of chromosome 1 in neuroblastoma. Genes Chromosomes Cancer 10: 275–281.
Takeda O, Homma C, Maseki N, Sakurai M, Kanda N, Schwab M et al. (1994). There may be two tumor suppressor genes on chromosome arm 1p closely associated with biologically distinct subtypes of neuroblastoma. Genes Chromosomes Cancer 10: 30–39.
White PS, Maris JM, Beltinger C, Sulman E, Marshall HN, Fujimori M et al. (1995). A region of consistent deletion in neuroblastoma maps within human chromosome 1p36.2–36.3. Proc Natl Acad Sci USA 92: 5520–5524.
Wyllie AH . (1980). Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature 284: 555–556.
Yang HW, Chen YZ, Piao HY, Takita J, Soeda E, Hayashi Y . (2001). DNA fragmentation factor 45 (DFF45) gene at 1p36.2 is homozygously deleted and encodes variant transcripts in neuroblastoma cell line. Neoplasia 3: 165–169.
Zhang J, Wang X, Bove KE, Xu M . (1999). DNA fragmentation factor 45-deficient cells are more resistant to apoptosis and exhibit different dying morphology than wild-type control cells. J Biol Chem 274: 37450–37454.
Acknowledgements
This work was supported in part by a Grant-in-Aid from the Ministry of Health, Labour and Welfare for Third Term Comprehensive Control Research for Cancer, and a Grant-in-Aid for Cancer Research from the Ministry of Health, Labour and Welfare of Japan, a Grant-in-Aid for Scientific Research on Priority Areas from the Ministry of Education, Culture, Sports, Science and Technology, Japan, and a grant from Uehara Memorial Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takahashi, M., Ozaki, T., Takahashi, A. et al. DFF45/ICAD restores cisplatin-induced nuclear fragmentation but not DNA cleavage in DFF45-deficient neuroblastoma cells. Oncogene 26, 5669–5673 (2007). https://doi.org/10.1038/sj.onc.1210352
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210352
Keywords
This article is cited by
-
The role of the DFF40/CAD endonuclease in genomic stability
Apoptosis (2021)