Abstract
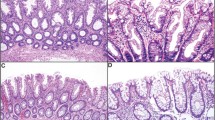
Serrated colorectal carcinomas (CRCs) are morphologically different from conventional CRCs and have been proposed to follow a distinct pathway of CRC formation. Despite studies of single molecular events in this tumor type, the diagnosis of serrated CRC relies on morphology and the putative unique biological character of these tumors has not been established. Here we show that the gene expression profiling of 37 CRCs separated serrated and conventional CRCs into two distinct branches in unsupervised hierarchical clustering (P-value 7.8 × 10−7), and revealed 201 differentially expressed genes representing potential biomarkers for serrated CRC. Immunohistochemistry was utilized to verify the key findings in the 37 CRCs examined by expression profiling, and a separate validation set of 37 serrated and 86 conventional CRCs was examined to evaluate the candidate biomarkers in an extended sample material. Ephrin receptor B2, hypoxia-inducible factor 1-alpha and patched appeared as proteins important for genesis of serrated CRC. This study establishes serrated CRCs as a biologically distinct subclass of CRC and represents a step forward in the molecular classification of these cancers. The study also provides a platform to understand the molecular basis of serrated CRC and in long term may contribute to the development of specific treatment options for this tumor type.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Accession codes
References
Aaltonen LA, Salovaara R, Kristo P, Canzian F, Hemminki A, Peltomaki P et al. (1998). N Engl J Med 338: 1481–1487.
Alazzouzi H, Davalos V, Kokko A, Domingo E, Woerner SM, Wilson AJ et al. (2005). Cancer Res 65: 10170–10173.
Arango D, Laiho P, Kokko A, Alhopuro P, Sammalkorpi H, Salovaara R et al. (2005). Gastroenterology 129: 874–884.
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM et al. (2000). Nat Genet 25: 25–29.
Batlle E, Bacani J, Begthel H, Jonkheer S, Gregorieff A, van de Born M et al. (2005). Nature 435: 1126–1130.
Batlle E, Henderson JT, Beghtel H, van den Born MM, Sancho E, Huls G et al. (2002). Cell 111: 251–263.
Benjamini Y, Hochberg Y . (1995). J Roy Stat Soc Ser B 57: 289–300.
Chan AO, Issa JP, Morris JS, Hamilton SR, Rashid A . (2002). Am J Pathol 160: 529–536.
Goldstein NS . (2006). Am J Clin Pathol 125: 146–153.
Golub TR, Slonim DK, Tamayo P, Huard C, Gaasenbeek M, Mesirov JP et al. (1999). Science 286: 531–537.
Hahn H, Wicking C, Zaphiropoulous PG, Gailani MR, Shanley S, Chidambaram A et al. (1996). Cell 85: 841–851.
Harris AL . (2002). Nat Rev Cancer 2: 38–47.
Hawkins NJ, Bariol C, Ward RL . (2002). Pathology 34: 548–555.
Hawkins NJ, Ward RL . (2001). J Natl Cancer Inst 93: 1307–1313.
Herman JG, Graff JR, Myohanen S, Nelkin BD, Baylin SB . (1996). Proc Natl Acad Sci USA 93: 9821–9826.
Higuchi T, Jass JR . (2004). J Clin Pathol 57: 682–686.
Hoffman EP, Awad T, Palma J, Webster T, Hubbell E, Warrington JA et al. (2004). Nat Rev Genet 5: 229–237.
Huusko P, Ponciano-Jackson D, Wolf M, Kiefer JA, Azorsa DO, Tuzmen S et al. (2004). Nat Genet 36: 979–983.
Iino H, Jass JR, Simms LA, Young J, Leggett B, Ajioka Y et al. (1999). J Clin Pathol 52: 5–9.
Jass JR, Whitehall VL, Young J, Leggett BA . (2002). Gastroenterology 123: 862–876.
Johnson RL, Rothman AL, Xie J, Goodrich LV, Bare JW, Bonifas JM et al. (1996). Science 272: 1668–1671.
Kambara T, Simms LA, Whitehall VL, Spring KJ, Wynter CV, Walsh MD et al. (2004). Gut 53: 1137–1144.
Kullander K, Klein R . (2002). Nat Rev Mol Cell Biol 3: 475–486.
Lazarus R, Junttila OE, Karttunen TJ, Makinen MJ . (2005). Am J Clin Pathol 123: 349–359.
Li LC, Dahiya R . (2002). Bioinformatics 18: 1427–1431.
Longacre TA, Fenoglio-Preiser CM . (1990). Am J Surg Pathol 14: 524–537.
Mäkinen MJ, George SM, Jernvall P, Makela J, Vihko P, Karttunen TJ . (2001). J Pathol 193: 286–294.
Mullor JL, Sanchez P, Altaba AR . (2002). Trends Cell Biol 12: 562–569.
Narita T, Saitoh K, Kameda T, Kuroiwa A, Mizutani M, Koike C et al. (2000). Development 127: 981–988.
Oldak M, Grzela T, Lazarczyk M, Malejczyk J, Skopinski P . (2001). Int J Mol Med 8: 445–452.
Pages G, Pouyssegur J . (2005). Cardiovasc Res 65: 564–573.
Park SJ, Rashid A, Lee JH, Kim SG, Hamilton SR, Wu TT . (2003). Am J Pathol 162: 815–822.
Prestridge DS . (1995). J Mol Biol 249: 923–932.
Rashid A, Houlihan PS, Booker S, Petersen GM, Giardiello FM, Hamilton SR . (2000). Gastroenterology 119: 323–332.
Salovaara R, Loukola A, Kristo P, Kaariainen H, Ahtola H, Eskelinen M et al. (2000). J Clin Oncol 18: 2193–2200.
Sawyer EJ, Cerar A, Hanby AM, Gorman P, Arends M, Talbot IC et al. (2002). Gut 51: 200–206.
Schwartz RA . (1978). N Engl J Med 299: 49.
Semenza GL . (2003). Nat Rev Cancer 3: 721–732.
Taipale J, Beachy PA . (2001). Nature 411: 349–354.
Tamayo P, Slonim D, Mesirov J, Zhu Q, Kitareewan S, Dmitrovsky E et al. (1999). Proc Natl Acad Sci USA 96: 2907–2912.
Tateyama H, Li W, Takahashi E, Miura Y, Sugiura H, Eimoto T . (2002). Am J Surg Pathol 26: 249–256.
Tuppurainen K, Makinen JM, Junttila O, Liakka A, Kyllonen AP, Tuominen H et al. (2005). J Pathol 207: 285–294.
Yashiro M, Laghi L, Saito K, Carethers JM, Slezak P, Rubio C et al. (2005). Cancer Epidemiol Biomarkers Prev 14: 2253–2256.
Zeeberg BR, Feng W, Wang G, Wang MD, Fojo AT, Sunshine M et al. (2003). Genome Biol 4: R28.
Zhong H, De Marzo AM, Laughner E, Lim M, Hilton DA, Zagzag D et al. (1999). Cancer Res 59: 5830–5835.
Acknowledgements
We thank Sini Marttinen and Riitta Vuento for assistance. The study was supported by grants from the European Commission (QLG2-CT-2001-01861), Finnish Cancer Society, the Academy of Finland, Sigrid Juselius Foundation, Ida Montin Foundation, Jalmari and Rauha Ahokas Foundation, Emil Aaltonen Foundation, Finnish Cultural Foundation, Maud Kuistila Foundation, The Finnish Oncology Foundation, Research and Science Foundation of Farmos, Foundation for Technological Advancement, Instrumentarium Science Foundation, Oulu University Hospital and Helsinki University Central Hospital. This work was carried out at the Center of Excellence in Translational Genome-Scale Biology of the Academy of Finland.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Rights and permissions
About this article
Cite this article
Laiho, P., Kokko, A., Vanharanta, S. et al. Serrated carcinomas form a subclass of colorectal cancer with distinct molecular basis. Oncogene 26, 312–320 (2007). https://doi.org/10.1038/sj.onc.1209778
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209778
Keywords
This article is cited by
-
Enhanced SREBP2-driven cholesterol biosynthesis by PKCλ/ι deficiency in intestinal epithelial cells promotes aggressive serrated tumorigenesis
Nature Communications (2023)
-
Lymphocyte antigen 6G6D-mediated modulation through p38α MAPK and DNA methylation in colorectal cancer
Cancer Cell International (2022)
-
SGAClust: Semi-supervised Graph Attraction Clustering of gene expression data
Network Modeling Analysis in Health Informatics and Bioinformatics (2022)
-
ARNT deficiency represses pyruvate dehydrogenase kinase 1 to trigger ROS production and melanoma metastasis
Oncogenesis (2021)
-
A Hierarchical Clustering algorithm based on Silhouette Index for cancer subtype discovery from genomic data
Neural Computing and Applications (2020)