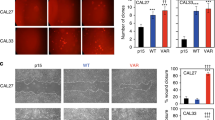
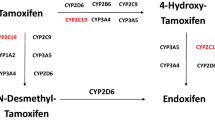
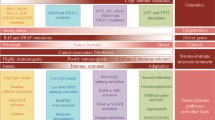
Abstract
The cytochromes P450 (CYPs) are key enzymes in cancer formation and cancer treatment. They mediate the metabolic activation of numerous precarcinogens and participate in the inactivation and activation of anticancer drugs. Since all CYPs that metabolize xenobiotics are polymorphic, much emphasis has been put on the investigation of a relationship between the distribution of specific variant CYP alleles and risk for different types of cancer, but a consistent view does not yet exist. This is to a great extent explained by the fact that the CYPs involved in activation of precarcinogens are in general not functionally polymorphic. This is in contrast to CYPs that are active in drug biotransformation where large interindividual differences in the capacity to metabolize therapeutic drugs are seen as a consequence of polymorphic alleles with altered function. This includes also some anticancer drugs like tamoxifen and cyclophosphamide metabolized by CYP2D6, CYP2C19 and CYP2B6. Some P450 forms are also selectively expressed in tumours, and this could provide a mechanism for drug resistance, but also future therapies using these enzymes as drug targets can be envisioned. This review gives an up-to-date description of our current knowledge in these areas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Agundez JA . (2004). Curr Drug Metab 5: 211–224.
Aklillu E, Carrillo JA, Makonnen E, Hellman K, Pitarque M, Bertilsson L et al. (2003). Mol Pharmacol 64: 659–669.
Aklillu E, Oscarson M, Hidestrand M, Leidvik B, Otter C, Ingelman-Sundberg M . (2002). Mol Pharmacol 61: 586–594.
Aklillu E, Ovrebo S, Botnen IV, Otter C, Ingelman-Sundberg M . (2005). Cancer Res 65: 5105–5111.
Aklillu E, Persson I, Bertilsson L, Johansson I, Rodrigues F, Ingelman-Sundberg M . (1996). J Pharmacol Exp Ther 278: 441–446.
Ariyoshi N, Miyamoto M, Umetsu Y, Kunitoh H, Dosaka-Akita H, Sawamura Y et al. (2002). Cancer Epidemiol Biomarkers Prev 11: 890–894.
Bournique B, Lemarie A . (2002). Drug Metab Dispos 30: 1149–1152.
Buters J, Quintanilla-Martinez L, Schober W, Soballa VJ, Hintermair J, Wolff T et al. (2003). Carcinogenesis 24: 327–334.
Buters JT, Sakai S, Richter T, Pineau T, Alexander DL, Savas U et al. (1999). Proc Natl Acad Sci USA 96: 1977–1982.
Chen JK, Capdevila J, Harris RC . (2001). Mol Cell Biol 21: 6322–6331.
Clarke R, Liu MC, Bouker KB, Gu Z, Lee RY, Zhu Y et al. (2003). Oncogene 22: 7316–7339.
Crewe HK, Notley LM, Wunsch RM, Lennard MS, Gillam EM . (2002). Drug Metab Dispos 30: 869–874.
Dachs GU, Tupper J, Tozer GM . (2005). Anticancer Drugs 16: 349–359.
Dai D, Zeldin DC, Blaisdell JA, Chanas B, Coulter SJ, Ghanayem BI et al. (2001). Pharmacogenetics 11: 597–607.
Daigo S, Takahashi Y, Fujieda M, Ariyoshi N, Yamazaki H, Koizumi W et al. (2002). Pharmacogenetics 12: 299–306.
Dally H, Edler L, Jager B, Schmezer P, Spiegelhalder B, Dienemann H et al. (2003). Pharmacogenetics 13: 607–618.
DeMichele A, Aplenc R, Botbyl J, Colligan T, Wray L, Klein-Cabral M et al. (2005). J Clin Oncol 23: 5552–5559.
Desta Z, Zhao X, Shin JG, Flockhart DA . (2002). Clin Pharmacokinet 41: 913–958.
Dhaini HR, Thomas DG, Giordano TJ, Johnson TD, Biermann JS, Leu K et al. (2003). J Clin Oncol 21: 2481–2485.
Downie D, McFadyen MC, Rooney PH, Cruickshank ME, Parkin DE, Miller ID et al. (2005). Clin Cancer Res 11: 7369–7375.
Ekins S, Vandenbranden M, Ring BJ, Gillespie JS, Yang TJ, Gelboin HV et al. (1998). J Pharmacol Exp Ther 286: 1253–1259.
Engels FK, Ten Tije AJ, Baker SD, Lee CK, Loos WJ, Vulto AG et al. (2004). Clin Pharmacol Ther 75: 448–454.
Friedman HS, Petros WP, Friedman AH, Schaaf LJ, Kerby T, Lawyer J et al. (1999). J Clin Oncol 17: 1516–1525.
Gambier N, Batt AM, Marie B, Pfister M, Siest G, Visvikis-Siest S . (2005). Pharmacogenomics J 5: 271–275.
Gervot L, Rochat B, Gautier JC, Bohnenstengel F, Kroemer H, de Berardinis V et al. (1999). Pharmacogenetics 9: 295–306.
Gharavi N, El-Kadi AO . (2004). Curr Drug Metab 5: 203–210.
Gibson P, Gill JH, Khan PA, Seargent JM, Martin SW, Batman PA et al. (2003). Mol Cancer Ther 2: 527–534.
Goh BC, Lee SC, Wang LZ, Fan L, Guo JY, Lamba J et al. (2002). J Clin Oncol 20: 3683–3690.
Gonzalez FJ . (2003). Drug Metab Rev 35: 319–335.
Goodwin B, Moore LB, Stoltz CM, McKee DD, Kliewer SA . (2001). Mol Pharmacol 60: 427–431.
Goria-Gatti L, Iannone A, Tomasi A, Poli G, Albano E . (1992). Carcinogenesis 13: 799–805.
Gribben JG, Ryan DP, Boyajian R, Urban RG, Hedley ML, Beach K et al. (2005). Clin Cancer Res 11: 4430–4436.
Guengerich FP, Parikh A, Turesky RJ, Josephy PD . (1999). Mutat Res 428: 115–124.
Han XM, Ouyang DS, Chen XP, Shu Y, Jiang CH, Tan ZR et al. (2002). Br J Clin Pharmacol 54: 540–543.
Hankinson O . (1995). Annu Rev Pharmacol Toxicol 35: 307–340.
Harris JW, Katki A, Anderson LW, Chmurny GN, Paukstelis JV, Collins JM . (1994). J Med Chem 37: 706–709.
Hayashi S, Watanabe J, Kawajiri K . (1991). J Biochem (Tokyo) 110: 559–565.
Henningsson A, Marsh S, Loos WJ, Karlsson MO, Garsa A, Mross K et al. (2005). Clin Cancer Res 11: 8097–8104.
Hesse LM, He P, Krishnaswamy S, Hao Q, Hogan K, von Moltke LL et al. (2004). Pharmacogenetics 14: 225–238.
Hirth J, Watkins PB, Strawderman M, Schott A, Bruno R, Baker LH . (2000). Clin Cancer Res 6: 1255–1258.
Huang Z, Raychowdhury MK, Waxman DJ . (2000a). Cancer Gene Ther 7: 1034–1042.
Huang Z, Roy P, Waxman DJ . (2000b). Biochem Pharmacol 59: 961–972.
Ingelman-Sundberg M . (2004a). Naunyn-Schmiedeberg's Arch Pharmacol 369: 89–104.
Ingelman-Sundberg M . (2004b). Trends Pharmacol Sci 25: 193–200.
Ingelman-Sundberg M . (2005). Pharmacogenomics J 5: 6–13.
Ingelman-Sundberg M, Rodriguez-Antona C . (2005). Philos Trans R Soc Lond B Biol Sci 360: 1563–1570.
Jacobson PA, Green K, Birnbaum A, Remmel RP . (2002). Cancer Chemother Pharmacol 49: 461–467.
Jiang JG, Chen CL, Card JW, Yang S, Chen JX, Fu XN et al. (2005). Cancer Res 65: 4707–4715.
Jin Y, Desta Z, Stearns V, Ward B, Ho H, Lee KH et al. (2005). J Natl Cancer Inst 97: 30–39.
Johansson I, Lundqvist E, Bertilsson L, Dahl ML, Sjoqvist F, Ingelman-Sundberg M . (1993). Proc Natl Acad Sci USA 90: 11825–11829.
Jordan VC, Collins MM, Rowsby L, Prestwich G . (1977). J Endocrinol 76: 305–316.
Kaijser GP, Korst A, Beijnen JH, Bult A, Underberg WJ . (1993). Anticancer Res 13: 1311–1324.
Kaiser R, Sezer O, Papies A, Bauer S, Schelenz C, Tremblay PB et al. (2002). J Clin Oncol 20: 2805–2811.
Karlgren M, Gomez A, Stark K, Svärd J, Rodriguez-Antona C, Oliw E et al. (submitted).
Karlgren M, Miura S, Ingelman-Sundberg M . (2005). Toxicol Appl Pharmacol 207: 57–61.
Kirchheiner J, Brockmoller J . (2005). Clin Pharmacol Ther 77: 1–16.
Klein K, Lang T, Saussele T, Barbosa-Sicard E, Schunck WH, Eichelbaum M et al. (2005). Pharmacogenet Genomics 15: 861–873.
Komatsu T, Yamazaki H, Shimada N, Nakajima M, Yokoi T . (2000). Drug Metab Dispos 28: 1457–1463.
Kuehl P, Zhang J, Lin Y, Lamba J, Assem M, Schuetz J et al. (2001). Nat Genet 27: 383–391.
Kumarakulasingham M, Rooney PH, Dundas SR, Telfer C, Melvin WT, Curran S et al. (2005). Clin Cancer Res 11: 3758–3765.
Lacroix D, Sonnier M, Moncion A, Cheron G, Cresteil T . (1997). Eur J Biochem 247: 625–634.
Landi S, Gemignani F, Moreno V, Gioia-Patricola L, Chabrier A, Guino E et al. (2005). Pharmacogenet Genomics 15: 535–546.
Lang T, Klein K, Fischer J, Nussler AK, Neuhaus P, Hofmann U et al. (2001). Pharmacogenetics 11: 399–415.
Lang T, Klein K, Richter T, Zibat A, Kerb R, Eichelbaum M et al. (2004). J Pharmacol Exp Ther 311: 34–43.
Lee SJ, Usmani KA, Chanas B, Ghanayem B, Xi T, Hodgson E et al. (2003). Pharmacogenetics 13: 461–472.
Leong CO, Gaskell M, Martin EA, Heydon RT, Farmer PB, Bibby MC et al. (2003). Br J Cancer 88: 470–477.
Li AP, Kaminski DL, Rasmussen A . (1995). Toxicology 104: 1–8.
Libby RT, Smith RS, Savinova OV, Zabaleta A, Martin JE, Gonzalez FJ et al. (2003). Science 299: 1578–1581.
Lohr M, Hoffmeyer A, Kroger J, Freund M, Hain J, Holle A et al. (2001). Lancet 357: 1591–1592.
Malaiyandi V, Sellers EM, Tyndale RF . (2005). Clin Pharmacol Ther 77: 145–158.
Martinez C, Garcia-Martin E, Blanco G, Gamito FJ, Ladero JM, Agundez JA . (2005). Br J Clin Pharmacol 59: 62–69.
Masson LF, Sharp L, Cotton SC, Little J . (2005). Am J Epidemiol 161: 901–915.
Mathijssen RH, de Jong FA, van Schaik RH, Lepper ER, Friberg LE, Rietveld T et al. (2004). J Natl Cancer Inst 96: 1585–1592.
Mathijssen RH, Verweij J, de Bruijn P, Loos WJ, Sparreboom A . (2002). J Natl Cancer Inst 94: 1247–1249.
McFadyen MC, Breeman S, Payne S, Stirk C, Miller ID, Melvin WT et al. (1999). J Histochem Cytochem 47: 1457–1464.
McFadyen MC, Cruickshank ME, Miller ID, McLeod HL, Melvin WT, Haites NE et al. (2001a). Br J Cancer 85: 242–246.
McFadyen MC, McLeod HL, Jackson FC, Melvin WT, Doehmer J, Murray GI . (2001b). Biochem Pharmacol 62: 207–212.
McFadyen MC, Melvin WT, Murray GI . (2004). Mol Cancer Ther 3: 363–371.
Miksys S, Lerman C, Shields PG, Mash DC, Tyndale RF . (2003). Neuropharmacology 45: 122–132.
Miyoshi Y, Ando A, Takamura Y, Taguchi T, Tamaki Y, Noguchi S . (2002). Int J Cancer 97: 129–132.
Miyoshi Y, Taguchi T, Kim SJ, Tamaki Y, Noguchi S . (2005). Breast Cancer 12: 11–15.
Murray GI, Taylor MC, McFadyen MC, McKay JA, Greenlee WF, Burke MD et al. (1997). Cancer Res 57: 3026–3031.
Nakajima M, Fujiki Y, Kyo S, Kanaya T, Nakamura M, Maida Y et al. (2005). J Clin Pharmacol 45: 674–682.
Nakajima M, Kwon JT, Tanaka N, Zenta T, Yamamoto Y, Yamamoto H et al. (2001). Clin Pharmacol Ther 69: 72–78.
Nebert DW, Dalton TP, Okey AB, Gonzalez FJ . (2004). J Biol Chem 279: 23847–23850.
Nelson DR, Zeldin DC, Hoffman SM, Maltais LJ, Wain HM, Nebert DW . (2004). Pharmacogenetics 14: 1–18.
Niemi M, Leathart JB, Neuvonen M, Backman JT, Daly AK, Neuvonen PJ . (2003). Clin Pharmacol Ther 74: 380–387.
Oscarson M . (2001). Drug Metab Dispos 29: 91–95.
Oscarson M, McLellan RA, Gullsten H, Yue QY, Lang MA, Bernal ML et al. (1999). FEBS Lett 448: 105–110.
Oyama T, Kagawa N, Kunugita N, Kitagawa K, Ogawa M, Yamaguchi T et al. (2004). Front Biosci 9: 1967–1976.
Ozdemir V, Kalowa W, Tang BK, Paterson AD, Walker SE, Endrenyi L et al. (2000). Pharmacogenetics 10: 373–388.
Paris PL, Kupelian PA, Hall JM, Williams TL, Levin H, Klein EA et al. (1999). Cancer Epidemiol Biomarkers Prev 8: 901–905.
Potter GA, Patterson LH, Wanogho E, Perry PJ, Butler PC, Ijaz T et al. (2002). Br J Cancer 86: 774–778.
Pozzi A, Macias-Perez I, Abair T, Wei S, Su Y, Zent R et al. (2005). J Biol Chem 280: 27138–27146.
Rae JM, Soukhova NV, Flockhart DA, Desta Z . (2002). Drug Metab Dispos 30: 525–530.
Rasmussen BB, Brix TH, Kyvik KO, Brosen K . (2002). Pharmacogenetics 12: 473–478.
Rebbeck TR, Jaffe JM, Walker AH, Wein AJ, Malkowicz SB . (1998). J Natl Cancer Inst 90: 1225–1229.
Riddick DS, Lee C, Ramji S, Chinje EC, Cowen RL, Williams KJ et al. (2005). Drug Metab Dispos 33: 1083–1096.
Rieger MA, Ebner R, Bell DR, Kiessling A, Rohayem J, Schmitz M et al. (2004). Cancer Res 64: 2357–2364.
Rochat B, Morsman JM, Murray GI, Figg WD, McLeod HL . (2001). J Pharmacol Exp Ther 296: 537–541.
Rodriguez-Antona C, Sayi JG, Gustafsson LL, Bertilsson L, Ingelman-Sundberg M . (2005). Biochem Biophys Res Commun 338: 299–305.
Rooseboom M, Commandeur JN, Vermeulen NP . (2004). Pharmacol Rev 56: 53–102.
Roy P, Waxman DJ . (2005). Toxicol In Vitro (in press).
Sachse C, Brockmoller J, Bauer S, Roots I . (1999). Br J Clin Pharmacol 47: 445–449.
Salmons B, Lohr M, Gunzburg WH . (2003). J Gastroenterol 38(Suppl 15): 78–84.
Savas U, Hsu MH, Griffin KJ, Bell DR, Johnson EF . (2005). Arch Biochem Biophys 436: 377–385.
Sim SC, Edwards RJ, Boobis AR, Ingelman-Sundberg M . (2005b). Pharmacogenet Genomics 15: 625–631.
Sim SC, Risinger C, Dahl M-L, Aklillu E, Christensen M, Bertilsson L et al. (2005a). Clin Pharm Ther (in press).
Spurdle AB, Goodwin B, Hodgson E, Hopper JL, Chen X, Purdie DM et al. (2002). Pharmacogenetics 12: 355–366.
Takada K, Arefayene M, Desta Z, Yarboro CH, Boumpas DT, Balow JE et al. (2004). Arthritis Rheum 50: 2202–2210.
Tanaka T, Tanimoto K, Otani K, Satoh K, Ohtaki M, Yoshida K et al. (2004). Int J Cancer 111: 617–626.
Taniguchi R, Kumai T, Matsumoto N, Watanabe M, Kamio K, Suzuki S et al. (2005). J Pharmacol Sci 97: 83–90.
Tayeb MT, Clark C, Haites NE, Sharp L, Murray GI, McLeod HL . (2003). Br J Cancer 88: 928–932.
Tayeb MT, Clark C, Sharp L, Haites NE, Rooney PH, Murray GI et al. (2002). Oncol Rep 9: 653–655.
Timm R, Kaiser R, Lotsch J, Heider U, Sezer O, Weisz K et al. (2005). Pharmacogenomics J 5: 365–373.
Tokizane T, Shiina H, Igawa M, Enokida H, Urakami S, Kawakami T et al. (2005). Clin Cancer Res 11: 5793–5801.
Tsuchiya K, Gatanaga H, Tachikawa N, Teruya K, Kikuchi Y, Yoshino M et al. (2004). Biochem Biophys Res Commun 319: 1322–1326.
Vineis P . (2002). Toxicology 181–182: 457–462.
Wang H, Faucette S, Sueyoshi T, Moore R, Ferguson S, Negishi M et al. (2003). J Biol Chem 278: 14146–14152.
Wang J, Pitarque M, Ingelman-Sundberg M . (2006). Biochem Biophys Res Commun 340: 491–497.
Wang J, Sönnerborg A, Rane A, Josephson F, Lundgren S, Ståhle L et al. (2005). Pharmacogenet Genomics (in press).
Westlind-Johnsson A, Bernhard Hauns D, Hermann R, Huennemeyer A, Lahu G, Nassr N et al. (2006). Clin Pharm Ther (in press).
Williams JA, Ring BJ, Cantrell VE, Jones DR, Eckstein J, Ruterbories K et al. (2002). Drug Metab Dispos 30: 883–891.
Xie HJ, Yasar U, Lundgren S, Griskevicius L, Terelius Y, Hassan M et al. (2003). Pharmacogenomics J 3: 53–61.
Yamamoto N, Tamura T, Murakami H, Shimoyama T, Nokihara H, Ueda Y et al. (2005). J Clin Oncol 23: 1061–1069.
Yasar U, Lundgren S, Eliasson E, Bennet A, Wiman B, de Faire U et al. (2002). Biochem Biophys Res Commun 299: 25–28.
Yengi LG, Xiang Q, Pan J, Scatina J, Kao J, Ball SE et al. (2003). Anal Biochem 316: 103–110.
Yu AM, Fukamachi K, Krausz KW, Cheung C, Gonzalez FJ . (2005). Endocrinology 146: 2911–2919.
Acknowledgements
The research at the author's laboratories is supported by The Swedish Research Council, The Swedish Cancer Foundation, NIH (NIGMS 1-R01 GM60548), by the ‘Ramon y Cajal’ program from the Spanish Ministry of Education and Science, and by a Marie Curie European Reintegration Grants of the European Community programme Structuring the European Research Area under contract number MERG-CG-6-2005-014881.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rodriguez-Antona, C., Ingelman-Sundberg, M. Cytochrome P450 pharmacogenetics and cancer. Oncogene 25, 1679–1691 (2006). https://doi.org/10.1038/sj.onc.1209377
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209377
Keywords
This article is cited by
-
Spatial metabolic heterogeneity of oligodendrogliomas at single-cell resolution
Brain Tumor Pathology (2023)
-
Bioinformatics screening the novel and promising targets of curcumin in hepatocellular carcinoma chemotherapy and prognosis
BMC Complementary Medicine and Therapies (2022)
-
Integration of liquid biopsy and pharmacogenomics for precision therapy of EGFR mutant and resistant lung cancers
Molecular Cancer (2022)