Abstract
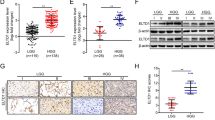
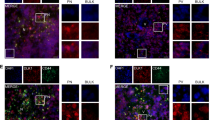
DLK1 (delta-like) is a transmembrane and secreted protein in the epidermal growth factor-like homeotic family. Although expressed widely during embryonic development, only a few tissues retain the expression in adults. Neuroendocrine tumors often highly express this protein; therefore, we hypothesized that brain tumors might also express it. This study found that the expression of DLK1 in gliomas was higher than that in normal brain (P<0.05). After stable transfection of a DLK1 cDNA expression vector into GBM cell lines, their proliferation was increased. Furthermore, they lost contact inhibition, had enhanced anchorage-independent growth in soft agar, and had significantly greater capacity to migrate. Western blot studies showed that expression of cyclin D1, CDK2, and E2F4 were increased, and Rb levels were decreased in these cells. DLK1 was found on the cell surface and secreted in the medium from the transfected GBM cells. DLK1-enriched condition medium stimulated the growth of glioblastoma multiforme cell lines and explants. DLK1 antibody blocked cell growth stimulated by DLK1. In summary, these results suggest that DLK1 may play a role in the formation or progression of gliomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
Abbreviations
- GBM:
-
glioblastoma multiforme
- T98G/w, U87/w, and U118/w:
-
wild-type GBM cell lines (T98G, U87, and U118)
- T98G/neo, U87/neo, and U118/neo:
-
T98G, U87, U118 cells transfected with neo-containing vector
- T98G/D, U87/D, and U118/D:
-
T98G, U87, U118 cells stably transfected with the DLK1 expression vector
- FACS:
-
fluorescence-activated cell sorting
References
Abdallah BM, Jensen CH, Gutierrez G, Leslie RG, Jensen TG, Kassem M . (2004). J Bone Miner Res 19: 841–852.
Baladron V, Ruiz-Hidalgo MJ, Nueda ML, Diaz-Guerra MJ, Garcia-Ramirez JJ, Bonvini E et al. (2005). Exp Cell Res 303: 343–359.
Baladron V, Ruiz-Hidalgo MJ, Bonvini E, Gubina E, Notario V, Laborda J . (2002). Biochem Biophys Res Commun 291: 193–204.
Bauer SR, Ruiz-Hidalgo MJ, Rudikoff EK, Goldstein J, Laborda J . (1998). Mol Cell Biol 18: 5247–5255.
Boney CM, Fiedorek FTJ, Paul SR, Gruppuso PA . (1996). Endocrinology 137: 2923–2928.
Capobianco AJ, Zagouras P, Blaumueller CM, Artavanis-Tsakonas S, Bishop JM . (1997). Mol Cell Biol 17: 6265–6273.
Carpenter G, Zendegui JG . (1986). Exp Cell Res 164: 1–10.
Cooper MJ, Hutchins GM, Cohen PS, Helman LJ, Mennie RJ, Israel MA . (1990). Cell Growth Differ 1: 149–159.
Edwards PA . (1999). Cancer Metast Rev 18: 175–180.
Ellisen LW, Bird J, West DC, Soreng AL, Reynolds TC, Smith SD et al. (1991). Cell 66: 649–661.
Engel J . (1989). FEBS Lett 251: 1–7.
Fan X, Mikolaenko I, Elhassan I, Ni X, Wang Y, Ball D et al. (2004). Cancer Res 64: 7787–7793.
Floridon C, Jensen CH, Thorsen P, Nielsen O, Sunde L, Westergaard JG et al. (2000). Differentiation 66: 49–59.
Gaetano C, Matsumoto K, Thiele CJ . (1992). Cancer Res 52: 4402–4407.
Garces C, Ruiz-Hidalgo MJ, Bonvini E, Goldstein J, Laborda J . (1999). Differentiation 64: 103–114.
Halder SK, Takemori H, Hatano O, Nonaka Y, Wada A, Okamoto M . (1998). Endocrinology 139: 3316–3328.
Hartman J, Muller P, Foster JS, Wimalasena J, Gustafsson JA, Strom A . (2004). Oncogene 23: 8826–8833.
Helman LJ, Thiele CJ, Linehan WM, Nelkin BD, Baylin SB, Israel MA . (1987). Proc Natl Acad Sci USA 84: 2336–2339.
Jensen CH, Teisner B, Hojrup P, Rasmussen HB, Madsen OD, Nielsen B et al. (1993). Hum Reprod 8: 635–641.
Kaneta M, Osawa M, Sudo K, Nakauchi H, Farr AG, Takahama Y . (2000). J Immunol 164: 256–264.
Laborda J . (2000). Histol Histopathol 15: 119–129.
Laborda J, Sausville EA, Hoffman T, Notario V . (1993). J Biol Chem 268: 3817–3820.
Lee YL, Helman L, Hoffman T, Laborda J . (1995). Biochim Biophys Acta 1261: 223–232.
Mark M, Rijli FM, Chambon P . (1997). Pediatr Res 42: 421–429.
Moon YS, Smas CM, Lee K, Villena JA, Kim KH, Yun EJ et al. (2002). Mol Cell Biol 22: 5585–5592.
Moore KA, Pytowski B, Witte L, Hicklin D, Lemischka IR . (1997). Proc Natl Acad Sci USA 94: 4011–4016.
Muller P, Kietz S, Gustafsson JA, Strom A . (2002). J Biol Chem 277: 28376–28379.
Munker R, Norman A, Koeffler HP . (1986). J Clin Invest 78: 424–430.
Muskavitch MA, Hoffmann FM . (1990). Curr Top Dev Biol 24: 289–328.
Nicolas M, Wolfer A, Raj K, Kummer JA, Mill P, van Noort M et al. (2003). Nat Genet 33: 416–421.
Samulewicz SJ, Seitz A, Clark L, Heber-Katz E . (2002). Wound Repair Regen 10: 215–221.
Schmidt JV, Matteson PG, Jones BK, Guan XJ, Tilghman SM . (2000). Genes Dev 14: 1997–2002.
Simpson P . (1995). Nature 375: 736–737.
Smas CM, Green D, Sul HS . (1994). Biochemistry 33: 9257–9265.
Smas CM, Sul HS . (1993). Cell 73: 725–734.
Sriuranpong V, Borges MW, Ravi RK, Arnold DR, Nelkin BD, Baylin SB et al. (2001). Cancer Res 61: 3200–3205.
Stockhausen MT, Sjolund J, Manetopoulos C, Axelson H . (2005). Br J Cancer 92: 751–759.
Takada S, Paulsen M, Tevendale M, Tsai CE, Kelsey G, Cattanach BM et al. (2002). Hum Mol Genet 11: 77–86.
Takada S, Tevendale M, Baker J, Georgiades P, Campbell E, Freeman T et al. (2000). Curr Biol 10: 1135–1138.
Tornehave D, Jensen CH, Teisner B, Larsson LI . (1996). Histochem Cell Biol 106: 535–542.
Tornehave D, Jansen P, Teisner B, Rasmussen HB, Chemnitz J, Moscoso G . (1993). Anat Embryol (Berl) 187: 335–341.
van Limpt V, Chan A, Caron H, Sluis PV, Boon K, Hermus MC et al. (2000). Med Pediatr Oncol 35: 554–558.
Wylie AA, Murphy SK, Orton TC, Jirtle RL . (2000). Genome Res 10: 1711–1718.
Yin D, Xie D, Hofmann WK, Miller CW, Black KL, Koeffler HP . (2002). Oncogene 21: 8372–8378.
Acknowledgements
This work was supported in part by National Institutes of Health, Cindy and Alan Horn Fund, Inger Fund, Parker Hughes Trust, NSFC30470980, and a grant from the Maxine Dunitz Neurosurgical Institute, Cedars-Sinai Medical Center. HPK is a member of the Molecular Biology Institute and Jonsson Comprehensive Cancer Center at UCLA, and holds the endowed Mark Goodson Chair of Oncology Research at Cedars-Sinai Medical Center/UCLA School of Medicine.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yin, D., Xie, D., Sakajiri, S. et al. DLK1: increased expression in gliomas and associated with oncogenic activities. Oncogene 25, 1852–1861 (2006). https://doi.org/10.1038/sj.onc.1209219
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209219
Keywords
This article is cited by
-
Hypoxia-induced release, nuclear translocation, and signaling activity of a DLK1 intracellular fragment in glioma
Oncogene (2020)
-
Delta-like 1 homologue promotes tumorigenesis and epithelial-mesenchymal transition of ovarian high-grade serous carcinoma through activation of Notch signaling
Oncogene (2019)
-
Soluble delta-like 1 homolog (DLK1) stimulates angiogenesis through Notch1/Akt/eNOS signaling in endothelial cells
Angiogenesis (2018)
-
fosB-Null Mice Display Impaired Adult Hippocampal Neurogenesis and Spontaneous Epilepsy with Depressive Behavior
Neuropsychopharmacology (2013)
-
Effects of varying Notch1 signal strength on embryogenesis and vasculogenesis in compound mutant heterozygotes
BMC Developmental Biology (2010)