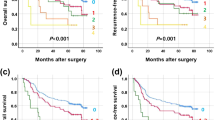
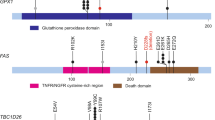
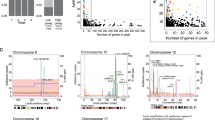
Abstract
Array-based comparative genomic hybridization (CGH-array) has a powerful potential for high-throughput identification of genetic aberrations in cell genomes. We identified a homozygous loss of ADAM23 (2q33.3) in the course of a program to screen a panel of gastric cancer (GC) cell lines (1/32, 3.1%) for genomic copy-number aberrations using our custom-made CGH-array. Infrequent homozygous deletion of ADAM23 was also seen in primary gastric tumors (1/39, 2.6%). ADAM23 mRNA was expressed in normal stomach tissue, but not in the majority of GC cell lines without homozygous deletion of this gene. Expression of ADAM23 mRNA was restored to gene-silenced GC cells after treatment with 5-aza 2′-deoxycytidine. The methylation status of the ADAM23 CpG island, which showed promoter activity, correlated inversely with its expression. Methylation of this CpG island was observed both in GC cell lines and in primary GC tissues; in primary tumors with a hypermethylated CpG island, expression of ADAM23 was lower than in adjacent noncancerous tissues. Moreover, restoration of ADAM23 in GC cells reduced their numbers in colony-formation assays. These results suggest that genetic or epigenetic silencing by hypermethylation of the ADAM23 CpG-rich promoter region leads to loss of ADAM23 function, which may be a factor in gastric carcinogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Akama Y, Yasui W, Kuniyasu H, Yokozaki H, Akagi M, Tahara H, Ishikawa T and Tahara E . (1996). Jpn J Cancer Res., 87, 824–830.
Aqeilan RI, Kuroki T, Pekarsky Y, Albagha O, Trapasso F, Baffa R, Huebner K, Edmonds P and Croce CM . (2004). Clin Cancer Res., 10, 3053–3058.
Baylin SB, Herman JG, Graff JR, Vertino PM and Issa JP . (1998). Adv Cancer Res., 72, 141–196.
Cal S, Freije JM, Lopez JM, Takada Y and Lopez-Otin C . (2000). Mol Biol Cell, 11, 1457–1469.
Carl-McGrath S, Lendeckel U, Ebert M, Roessner A and Rocken C . (2005). Int J Oncol., 26, 17–24.
Costa FF, Verbisck NV, Salim AC, Ierardi DF, Pires LC, Sasahara RM, Sogayar MC, Zanata SM, Mackay A, O'Hare M, Soares F, Simpson AJ and Camargo AA . (2004). Oncogene, 23, 1481–1488.
Feakins RM, Nickols CD, Bidd H and Walton SJ . (2003). Hum Pathol., 34, 1276–1282.
Fukuda Y, Kurihara N, Imoto I, Yasui K, Yoshida M, Yanagihara K, Park JG, Nakamura Y and Inazawa J . (2000). Genes Chromosomes Cancer, 29, 315–324.
Gorringe KL, Boussioutas A, Bowtell DD and Melbourne Gastric Cancer Group. Peter Mac Micro Array Facility. (2005). Genes Chromosomes Cancer, 42, 247–259.
Huiping C, Kristjansdottir S, Bergthorsson JT, Jonasson JG, Magnusson J, Egilsson V and Ingvarsson S . (2002). Eur J Cancer, 38, 728–735.
Imoto I, Yuki Y, Sonoda I, Ito T, Shimada Y, Imamura M and Inazawa J . (2003). Cancer Res., 63, 5691–5696.
Inazawa J, Inoue J and Imoto I . (2004). Cancer Sci., 95, 559–563.
Kang GH, Lee HJ, Hwang KS, Lee S, Kim JH and Kim JS . (2003). Am J Pathol., 163, 1551–1556.
Kimura Y, Noguchi T, Kawahara K, Kashima K, Daa T and Yokoyama S . (2004). Mod Pathol., 17, 1328–1337.
Kodama T, Ikeda E, Okada A, Ohtsuka T, Shimoda M, Shiomi T, Yoshida K, Nakada M, Ohuchi E and Okada Y . (2004). Am J Pathol., 165, 1743–1753.
Koo SH, Kwon KC, Shin SY, Jeon YM, Park JW, Kim SH and Noh SM . (2000). Cancer Genet Cytogenet., 117, 97–103.
McCulloch DR, Akl P, Samaratunga H, Herington AC and Odorico DM . (2004). Clin Cancer Res., 10, 314–323.
Noguchi M, Furuya S, Takeuchi T and Hirohashi S . (1997). Pathol Int, 47, 685–691.
O'Shea C, McKie N, Buggy Y, Duggan C, Hill AD, McDermott E, O'Higgins N and Duffy MJ . (2003). Int J Cancer, 105, 754–761.
Peng DF, Sugihara H, Mukaisho K, Tsubosa Y and Hattori T . (2003). J Pathol., 201, 439–450.
Sakakura C, Mori T, Sakabe T, Ariyama Y, Shinomiya T, Date K, Hagiwara A, Yamaguchi T, Takahashi T, Nakamura Y, Abe T and Inazawa J . (1999). Genes Chromosomes Cancer, 24, 299–305.
Seals DF and Courtneidge SA . (2003). Genes Dev., 17, 7–30.
Shintani Y, Higashiyama S, Ohta M, Hirabayashi H, Yamamoto S, Yoshimasu T, Matsuda H and Matsuura N . (2004). Cancer Res., 64, 4190–4196.
Snijders AM, Nowak N, Segraves R, Blackwood S, Brown N, Conroy J, Hamilton G, Hindle AK, Huey B, Kimura K, Law S, Myambo K, Palmer J, Ylstra B, Yue JP, Gray JW, Jain AN, Pinkel D and Albertson DG . (2001). Nat Genet., 29, 263–264.
Sonoda I, Imoto I, Inoue J, Shibata T, Shimada Y, Chin K, Imamura M, Amagasa T, Gray JW, Hirohashi S and Inazawa J . (2004). Cancer Res, 64, 3741–3747.
Tahara E . (1995). World J Surg., 19, 484–490.
Takada H, Imoto I, Tsuda H, Sonoda I, Ichikura T, Mochizuki H, Okanoue T and Inazawa J . (2005). Cancer Sci., 96, 100–110.
Tanabe C, Aoyagi K, Sakiyama T, Kohno T, Yanagitani N, Akimoto S, Sakamoto M, Sakamoto H, Yokota J, Ohki M, Terada M, Yoshida T and Sasaki H . (2003). Genes Chromosomes Cancer, 38, 168–176.
Tay ST, Leong SH, Yu K, Aggarwal A, Tan SY, Lee CH, Wong K, Visvanathan J, Lim Dennis, Wong WK, Soo KC, Kon OL and Tan P . (2003). Cancer Res., 63, 3309–3316.
Weiss MM, Kuipers EJ, Postma C, Snijders AM, Pinkel D, Meuwissen SG, Albertson D and Meijer GA . (2004). Cell Oncol., 26, 307–317.
Wu E, Croucher PI and McKie N . (1997). Biochem Biophys Res Commun., 235, 437–442.
Yuki Y, Imoto I, Imaizumi M, Hibi S, Kaneko Y, Amagasa T and Inazawa J . (2004). Cancer Sci., 95, 503–507.
Acknowledgements
We are grateful to Professor Yusuke Nakamura (Human Genome Center, The Institute of Medical Science, The University of Tokyo) for his continuous encouragement throughout this work. We thank Professor Jae-Gahb Park (Laboratory of Cell Biology, Cancer Research Institute, Seoul National University College of Medicine) and Dr Kazuyoshi Yanagihara (Central Animal Laboratory, National Cancer Center Research Institute) for providing GC cell lines and Ai Watanabe for technical assistance.
This work was supported by Grants-in-Aid for Scientific Research on Priority Areas (C) from the Ministry of Education, Culture, Sports, Science and Technology, Japan; by a Grant-in-Aid from Core Research for Evolutional Science and Technology (CREST) of the Japan Science and Technology Corporation (JST); by a Center of Excellence (COE) program for Frontier Research on Molecular Destruction and Reconstitution of Tooth and Bone; by the program for promotion of Fundamental Studies in Health Sciences of the Pharmaceuticals and Medical Devices Agency (PMDA); and by the Third Term Comprehensive Control Research for Cancer of the Ministry of Health, Labour and Welfare.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takada, H., Imoto, I., Tsuda, H. et al. ADAM23, a possible tumor suppressor gene, is frequently silenced in gastric cancers by homozygous deletion or aberrant promoter hypermethylation. Oncogene 24, 8051–8060 (2005). https://doi.org/10.1038/sj.onc.1208952
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208952
Keywords
This article is cited by
-
Integrated genomic and functional analyses reveal glyoxalase I as a novel metabolic oncogene in human gastric cancer
Oncogene (2015)
-
A novel method, digital genome scanning detects KRAS gene amplification in gastric cancers: involvement of overexpressed wild-type KRAS in downstream signaling and cancer cell growth
BMC Cancer (2009)
-
The ADAMs: signalling scissors in the tumour microenvironment
Nature Reviews Cancer (2008)
-
Array comparative genomic hybridization analysis of olfactory neuroblastoma
Modern Pathology (2008)
-
A novel amplification target, DUSP26, promotes anaplastic thyroid cancer cell growth by inhibiting p38 MAPK activity
Oncogene (2007)