Abstract
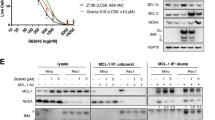
Mantle cell lymphoma (MCL) is a mature B-cell proliferation characterized by the presence of translocation t(11;14)(q13;q32), an aggressive clinical course, and poor response to chemotherapy. The majority of drugs currently used in the treatment of lymphoproliferative disorders induce cell death by triggering apoptosis, but few data concerning drug-induced apoptosis in MCL have been reported. We have analysed the mechanisms of drug-induced cell death in four cell lines with the t(11;14) and in primary cells from 10 patients with MCL. Mitoxantrone, a topoisomerase II inhibitor, induced a strong cytotoxic effect in three cell lines (JVM-2, REC-1, and Granta 519), and in primary MCL cells. This cytotoxic effect due to apoptosis induction was observed despite the presence of either p53 or ATM abnormalities. However, no cytotoxic effect was detected after incubation with DNA-damaging agents in the NCEB-1 cell line, carrying p53 and ATM alterations, despite the presence of functional mitochondrial machinery. These results support that mitoxantrone can be effective in the treatment of MCL but that this activity requires the integrity of functional DNA-damage response genes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Argatoff LH, Connors JM, Klasa RJ, Horsman DE and Gascoyne RD . (1997). Blood, 89, 2067–2078.
Bailly JD, Skladanowski A, Bettaieb A, Mansat V, Larsen AK and Laurent G . (1997). Leukemia, 11, 1523–1532.
Banin S, Moyal L, Shieh S, Taya Y, Anderson CW, Chessa L, Smorodinsky NI, Prives C, Reiss Y, Shiloh Y and Ziv Y . (1998). Science, 281, 1674–1677.
Bellosillo B, Colomer D, Pons G and Gil J . (1998). Br. J. Haematol., 100, 142–146.
Bellosillo B, Villamor N, Colomer D, Pons G, Montserrat E and Gil J . (1999). Blood, 94, 2836–2843.
Bellosillo B, Villamor N, Lopez-Guillermo A, Marce S, Esteve J, Campo E, Colomer D and Montserrat E . (2001). Blood, 98, 2771–2777.
Bellosillo B, Villamor N, Lopez-Guillermo A, Marce S, Bosch F, Campo E, Montserrat E and Colomer D . (2002). Blood, 100, 1810–1816.
Bertoni F, Zucca E and Cotter FE . (2004). Br. J. Haematol., 124, 130–140.
Bogner C, Ringshausen I, Schneller F, Fend F, Quintanilla-Martinez L, Hacker G, Goetze K, Oostendorp R, Peschel C and Decker T . (2003). Br. J. Haematol., 122, 260–268.
Bosch F, Jares P, Campo E, Lopez-Guillermo A, Piris MA, Villamor N, Tassies D, Jaffe ES, Montserrat E and Rozman C . (1994). Blood, 84, 2726–2732.
Bosch F, Ferrer A, Lopez-Guillermo A, Gine E, Bellosillo B, Villamor N, Colomer D, Cobo F, Perales M, Esteve J, Altes A, Besalduch J, Ribera JM and Montserrat E . (2002). Br. J. Haematol., 119, 976–984.
Bosch F, Lopez-Guillermo A, Campo E, Ribera JM, Conde E, Piris MA, Vallespi T, Woessner S and Montserrat E . (1998). Cancer, 82, 567–575.
Camacho E, Hernandez L, Hernandez S, Tort F, Bellosillo B, Bea S, Bosch F, Montserrat E, Cardesa A, Fernandez PL and Campo E . (2002). Blood, 99, 238–244.
Campo E . (2003). Hum. Pathol., 34, 330–335.
Campo E, Raffeld M and Jaffe ES . (1999). Semin. Hematol., 36, 115–127.
Chou TC and Talalay P . (1984). Adv. Enzyme Regul., 22, 27–55.
Cory S and Adams JM . (2002). Nat. Rev. Cancer, 2, 647–656.
Cory S, Huang DC and Adams JM . (2003). Oncogene, 22, 8590–8607.
De Isabella P, Capranico G and Zunino F . (1991). Life Sci., 48, 2195–2205.
Dohner H, Fischer K, Bentz M, Hansen K, Benner A, Cabot G, Diehl D, Schlenk R, Coy J and Stilgenbauer S . (1995). Blood, 85, 1580–1589.
Fang NY, Greiner TC, Weisenburger DD, Chan WC, Vose JM, Smith LM, Armitage JO, Mayer RA, Pike BL, Collins FS and Hacia JG . (2003). Proc. Natl. Acad. Sci. USA, 100, 5372–5377.
Fedier A, Schlamminger M, Schwarz VA, Haller U, Howell SB and Fink D . (2003). Ann. Oncol., 14, 938–945.
Flinn IW, Byrd JC, Morrison C, Jamison J, Diehl LF, Murphy T, Piantadosi S, Seifter E, Ambinder RF, Vogelsang G and Grever MR . (2000). Blood, 96, 71–75.
Hernandez L, Fest T, Cazorla M, Teruya-Feldstein J, Bosch F, Peinado MA, Piris MA, Montserrat E, Cardesa A, Jaffe ES, Campo E and Raffold M . (1996). Blood, 87, 3351–3359.
Hofmann WK, De Vos S, Tsukasaki K, Wachsman W, Pinkus GS, Said JW and Koeffler HP . (2001). Blood, 98, 787–794.
Khoury JD, Medeiros LJ, Rassidakis GZ, McDonnell TJ, Abruzzo LV and Lai R . (2003). J. Pathol., 199, 90–97.
M'kacher R, Bennaceur A, Farace F, Lauge A, Plassa LF, Wittmer E, Dossou J, Violot D, Deutsch E, Bourhis J, Stoppa-Lyonnet D, Ribrag V, Carde P, Parmentier C, Bernheim A and Turhan AG . (2003). Oncogene, 22, 5961–5968.
Martinez N, Camacho FI, Algara P, Rodriguez A, Dopazo A, Ruiz-Ballesteros E, Martin P, Martinez-Climent JA, Garcia-Conde J, Menarguez J, Solano F, Mollejo M and Piris MA . (2003). Cancer Res., 63, 8226–8232.
Molina-Arcas M, Bellosillo B, Casado FJ, Montserrat E, Gil J, Colomer D and Pastor-Anglada M . (2003). Blood, 101, 2328–2334.
Nicoletti I, Migliorati G, Pagliacci MC, Grignani F and Riccardi C . (1991). J. Immunol. Methods, 139, 271–279.
Pham LV, Tamayo AT, Yoshimura LC, Lo P and Ford RJ . (2003). J. Immunol., 171, 88–95.
Pinyol M, Hernandez L, Cazorla M, Balbin M, Jares P, Fernandez PL, Montserrat E, Cardesa A, Lopez-Otin C and Campo E . (1997). Blood, 89, 272–280.
Pinyol M, Hernandez L, Martinez A, Cobo F, Hernandez S, Bea S, Lopez-Guillermo A, Nayach I, Palacin A, Nadal A, Fernandez PL, Montserrat E, Cardesa A and Campo E . (2000). Am. J. Pathol., 156, 1987–1996.
Rasheed ZA and Rubin EH . (2003). Oncogene, 22, 7296–7304.
Reiman T, Graham KA, Wong J, Belch AR, Coupland R, Young J, Cass CE and Mackey JR . (2002). Leukemia, 16, 1886–1887.
Rosenwald A, Wright G, Wiestner A, Chan WC, Connors JM, Campo E, Gascoyne RD, Grogan TM, Muller-Hermelink HK, Smeland EB, Chiorazzi M, Giltnane JM, Hurt EM, Zhao H, Averett L, Henrickson S, Yang L, Powell J, Wilson WH, Jaffe ES, Simon R, Klausner RD, Montserrat E, Bosch F, Greiner TC, Weisenburger DD, Sanger WG, Dave BJ, Lynch JC, Vose J, Armitage JO, Fisher RI, Miller TP, LeBlanc M, Ott G, Kvaloy S, Holte H, Delabie J and Staudt LM . (2003). Cancer Cell, 3, 185–197.
Schlegelberger B, Zwingers T, Harder L, Nowotny H, Siebert R, Vesely M, Bartels H, Sonnen R, Hopfinger G, Nader A, Ott G, Muller-Hermelink K, Feller A and Heinz R . (1999). Blood, 94, 3114–3120.
Schrader C, Meusers P, Brittinger G, Teymoortash A, Siebmann JU, Janssen D, Parwaresch R and Tiemann M . (2004). Leukemia, 18, 1200–1206.
Slee EA, Adrain C and Martin SJ . (1999). Cell Death. Differ., 6, 1067–1074.
Solal-Celigny P, Brice P, Brousse N, Caspard H, Bastion Y, Haioun C, Bosly A, Tilly H, Bordessoule D, Sebban C, Harousseau JL, Morel P, Dupas B, Plassart F, Vasile N, Fort N and Leporrier M . (1996). J. Clin. Oncol., 14, 514–519.
Stankovic T, Hubank M, Cronin D, Stewart GS, Fletcher D, Bignell CR, Alvi AJ, Austen B, Weston VJ, Fegan C, Byrd PJ, Moss PA and Taylor AM . (2004). Blood, 103, 291–300.
Swerdlow SH, Berger F, Isaacson P, Muller-Hermelink HK, Nathwani BN, Piris MA and Harris NL . (2001). Pathology & Genetics. Tumours of haematopoietic and lymphoid tissues Jaffe ES, Harris NL, Stein H and Vardiman JW (ed). IARC press. Lyon, 168–170.
Vorechovsky I, Luo L, Dyer MJ, Catovsky D, Amlot PL, Yaxley JC, Foroni L, Hammarstrom L, Webster AD and Yuille MA . (1997). Nat. Genet., 17, 96–99.
Wattel E, Preudhomme C, Hecquet B, Vanrumbeke M, Quesnel B, Dervite I, Morel P and Fenaux P . (1994). Blood, 84, 3148–3157.
Xue LY, Chiu SM and Oleinick NL . (2003). Exp. Cell Res., 283, 135–145.
Zhao WL, Daneshpouy ME, Mounier N, Briere J, Leboeuf C, Plassa LF, Turpin E, Cayuela JM, Ameisen JC, Gisselbrecht C and Janin A . (2004). Blood, 103, 695–697.
Acknowledgements
This work was supported in part by Grants FIS 02/250, 03/0398, CICYT SAF 02/3261, and Red Estudio neoplasias Linfoides G03/179. AF had a fellowship from Hospital Clínic.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferrer, A., Marcé, S., Bellosillo, B. et al. Activation of mitochondrial apoptotic pathway in mantle cell lymphoma: high sensitivity to mitoxantrone in cases with functional DNA-damage response genes. Oncogene 23, 8941–8949 (2004). https://doi.org/10.1038/sj.onc.1208084
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208084
Keywords
This article is cited by
-
Deciphering molecular mechanisms underlying chemoresistance in relapsed AML patients: towards precision medicine overcoming drug resistance
Cancer Cell International (2021)
-
Multifunctional mitoxantrone-conjugated magnetic nanosystem for targeted therapy of folate receptor-overexpressing malignant cells
Journal of Nanobiotechnology (2015)