Abstract
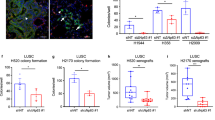
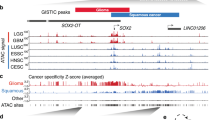
Sil (SCL interrupting locus) was cloned from the most common chromosomal rearrangement in T-cell acute lymphoblastic leukemia. It is an immediate early gene whose expression is associated with cell proliferation. Sil protein levels are tightly regulated during the cell cycle, reaching peak levels in mitosis and disappearing on transition to G1. A recent study found Sil to be one of 17 genes whose overexpression in primary adenocarcinomas predicts metastatic spread. We hypothesized that Sil might have a role in carcinogenesis. To address this question, we utilized several approaches. Using a multitumor tissue array, we found that Sil protein expression was increased mostly in lung cancer, but also at lower levels, in a subset of other tumors. Microarray gene expression analysis and immunohistochemistry of lung cancer samples verified these observations. Sil gene expression in lung cancer correlated with the expression of several kinetochore check-point genes and with the histopathologic mitotic index. These observations suggest that overexpression of the Sil gene characterizes tumors with increased mitotic activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Aplan PD, Lombardi DP, Ginsberg AM, Cossman J, Bertness VL and Kirsch IR . (1990). Science, 250, 1426–1429.
Aplan PD, Lombardi DP and Kirsch IR . (1991). Mol. Cell. Biol., 11, 5462–5469.
Bhattacharjee A, Richards WG, Staunton J, Li C, Monti S, Vasa P, Ladd C, Beheshti J, Bueno R, Gillette M, Loda M, Weber G, Mark EJ, Lander ES, Wong W, Johnson BE, Golub TR, Sugarbaker DJ and Meyerson M . (2001). Proc. Natl. Acad. Sci. USA, 98, 13790–13795.
Collazo-Garcia N, Scherer P and Aplan PD . (1995). Genomics, 30, 506–513.
Golling G, Amsterdam A, Sun Z, Antonelli M, Maldonado E, Chen W, Burgess S, Haldi M, Artzt K, Farrington S, Lin SY, Nissen RM and Hopkins N . (2002). Nat. Genet., 31, 135–140.
Izraeli S, Colaizzo-Anas T, Bertness VL, Mani K, Aplan PD and Kirsch IR . (1997). Cell Growth Differ., 8, 1171–1179.
Izraeli S, Lowe LA, Bertness VL, Campaner S, Hahn H, Kirsch IR and Kuehn MR . (2001). Genesis, 31, 72–77.
Izraeli S, Lowe LA, Bertness VL, Good DJ, Dorward DW, Kirsch IR and Kuehn MR . (1999). Nature, 399, 691–694.
Kaminski N and Friedman N . (2002). Am. J. Respir. Cell Mol. Biol., 27, 125–132.
Liu ST, Chan GK, Hittle JC, Fujii G, Lees E and Yen TJ . (2003). Mol. Biol. Cell., 14, 1638–1651.
Lynch J, Pattekar R, Barnes DM, Hanby AM, Camplejohn RS, Ryder K and Gillett CE . (2002). J. Pathol., 196, 275–279.
Michel LS, Liberal V, Chatterjee A, Kirchwegger R, Pasche B, Gerald W, Dobles M, Sorger PK, Murty VV and Benezra R . (2001). Nature, 409, 355–359.
Musacchio A and Hardwick KG . (2002). Nat. Rev. Mol. Cell. Biol., 3, 731–741.
Ortiz LA, Gambelli F, McBride C, Gaupp D, Baddoo M, Kaminski N and Phinney DG . (2003). Proc. Natl. Acad. Sci. USA, 100, 8407–8411.
Ramaswamy S, Ross KN, Lander ES and Golub TR . (2003). Nat. Genet., 33, 49–54.
Rattner JB, Hendzel MJ, Furbee CS, Muller MT and Bazett-Jones DP . (1996). J. Cell. Biol., 134, 1097–1107.
Starr DA, Saffery R, Li Z, Simpson AE, Choo KH, Yen TJ and Goldberg ML . (2000). J Cell Sci, 113, 1939–1950.
Takahashi T, Haruki N, Nomoto S, Masuda A, Saji S, Osada H and Takahashi T . (1999). Oncogene, 18, 4295–4300.
Travis WD, Colby TV, Corrin B, Shimosato Y and Brambilla E . (1999). Histological Typing of Lung and Pleural Tumours, 3rd edn Springer: Berlin.
Watkins DN, Berman DM, Burkholder SG, Wang B, Beachy PA and Baylin SB . (2003). Nature, 422, 313–317.
Whitfield ML, Sherlock G, Saldanha AJ, Murray JI, Ball CA, Alexander KE, Matese JC, Perou CM, Hurt MM, Brown PO and Botstein D . (2002). Mol. Biol. Cell, 13, 1977–2000.
Zhou J, Yao J and Joshi HC . (2002). J. Cell Sci., 115, 3547–3555.
Acknowledgements
We thank Sergey Nemez for the statistical analysis. This study was supported by research grants from the Israel Science Foundation, the Israel Cancer Research foundation and from Tel-Aviv University Recanati foundation to S Izraeli. N Kaminski is the Dorothy P and Richard P Simmons chair of Interstitial Lung Diseases at the University of Pittsburgh Medical School. G Rechavi holds the Gregorio and Dora Shapiro Chair for Hematology Malignancies, Sackler School of Medicine, Tel-Aviv University. This work was preformed in partial fulfillment of the requirements for the PhD degree of Ayelet Erez, Sackler Faculty of Medicine, Tel-Aviv University.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Erez, A., Perelman, M., Hewitt, S. et al. Sil overexpression in lung cancer characterizes tumors with increased mitotic activity. Oncogene 23, 5371–5377 (2004). https://doi.org/10.1038/sj.onc.1207685
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207685
Keywords
This article is cited by
-
ncRNAs-mediated overexpression of STIL predict unfavorable prognosis and correlated with the efficacy of immunotherapy of hepatocellular carcinoma
Cancer Cell International (2023)
-
STIL/AURKA axis promotes cell proliferation by influencing primary cilia formation in bladder cancer
Journal of Translational Medicine (2023)
-
A novel HIF1α-STIL-FOXM1 axis regulates tumor metastasis
Journal of Biomedical Science (2022)
-
The biological function and clinical significance of STIL in osteosarcoma
Cancer Cell International (2021)
-
Centrosome: A Microtubule Nucleating Cellular Machinery
Journal of the Indian Institute of Science (2021)