Abstract
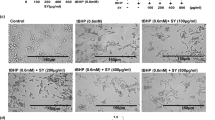
Chemoprevention has become an effective cancer control modality; however, the search for novel agent(s) for the armamentarium of cancer chemoprevention continues. We argue that agents capable for inhibition of promotion stage of tumorigenesis with the ability to intervene at several critical pathways in the tumorigenesis process will have greater advantage over other single-target agents. Lupeol, a triterpene, is the principal constituent of common fruit plants such as olive, mango, fig and medicinal herbs that have been used to treat skin aliments. Lupeol has been reported to possess a wide range of medicinal properties that include strong antioxidant, antimutagenic, anti-inflammatory and antiarthritic effects. In the present study, we show that Lupeol possesses antitumor-promoting effects in a mouse skin tumorigenesis model. We first determined the effect of topical application of Lupeol to CD-1 mouse against 12-O-tetradecanoyl-phorbol-13-acetate (TPA)-induced conventional markers and other novel markers of skin tumor promotion. We found that topical application of Lupeol (1–2 mg/mouse) 30 min prior to TPA (3.2 nmol/mouse) application onto the skin of CD-1 mice afforded significant inhibition, in a time- and dose-dependent manner, against TPA-mediated increase in (i) skin edema and hyperplasia, (ii) epidermal ornithine decarboxylase (ODC) activity, and (iii) protein expression of ODC, cyclo-oxygenase-2 and nitric oxide synthase. As of the role of nuclear factor kappa B (NF-κB) and phosphatidyl inositol 3-kinase (PI3K)/Akt signaling in tumor promotion, we next determined the effect of topical application of Lupeol to mouse skin against these signaling pathways. We found that Lupeol treatment to mouse skin resulted in the inhibition of TPA-induced (i) activation of PI3K, (ii) phosphorylation of Akt at Thr308, (iii) activation of NF-κB and IKKα, and (iv) degradation and phosphorylation of IκBα. The animals pretreated with Lupeol showed significantly reduced tumor incidence, lower tumor body burden and a significant delay in the latency period for tumor appearance. At the termination of the experiment at 28 weeks, 100% of the animals in TPA-treated group exhibited seven to eight tumors/mouse, whereas only 53% of the mice receiving Lupeol prior to TPA treatment exhibited one to three tumors/mouse. These results for the first time provide evidence that Lupeol possesses antiskin tumor-promoting effects in CD-1 mouse and inhibits conventional as well as novel biomarkers of tumor promotion. We suggest that Lupeol is an attractive antitumor-promoting agent that must be evaluated in tumor models other than skin carcinogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Afaq F, Adhami VM, Ahmad N and Mukhtar H . (2003). Oncogene, 22, 1035–1044.
Ahmad N, Gilliam AC, Katiyar SK, O’Brien TG and Mukhtar H . (2001). Am. J. Pathol., 159, 885–892.
Ahmad N, Gupta S, Husain MM, Heiskanen KM and Mukhtar H . (2000). Clin. Cancer Res., 6, 1524–1528.
Anjaneyulu V, Prasad KH and Rao GS . (1982). Indian J. Pharm. Sci., 44, 58–59.
Auvinen M . (1997). J. Natl. Cancer Inst., 89, 533–537.
Badria FA, Mikhaeil BR, Maatooq GT and Amer MM . (2003). Z. Naturforsch[C]., 58, 505–516.
Baeuerle PA and Baltimore D . (1996). Cell, 87, 13–20.
Baldwin Jr AS . (1996). Annu. Rev. Immunol., 14, 649–683.
Beveridge TH, Li TS and Drover JC . (2002). J. Agric. Food Chem., 50, 744–750.
Bhimani RS, Troll W, Grunberger D and Frenkel K . (1993). Inhibition of oxidative stress in HeLa cells by chemopreventive agents. Cancer Res., 53, 4528–4533.
Bickers DR and Athar M . (2000). J. Dermatol., 27, 691–695.
Bours V, Bentires-Alj M, Hellin AC, Viatour P, Robe P, Delhalle S, Benoit V and Merville MP . (2000). Biochem. Pharmacol., 60, 1085–1089.
Callejas NA, Casado M, Bosca L and Martin-Sanz P . (1999). J. Cell Sci., 18, 3147–3155.
Carpenter CL and Cantley LC . (1996). Curr. Opin. Cell Biol., 8, 153–158.
Chun KS, Kang JY, Kim OH, Kang H and Surh YJ . (2002). J. Environ. Pathol. Toxicol. Oncol., 21, 131–139.
Conney AH, Lou YR, Xie JG, Osawa T, Newmark HL, Liu Y, Chang RL and Huang MT . (1997). Proc. Soc. Exp. Biol. Med., 216, 234–245.
DiGiovanni J . (1991). Modification of Tumor Development in Rodents: Progress in Experimental Tumor Research, Ito N and Sugano H (ed) Karger: Basel, 33, pp. 192–229.
DiGiovanni J . (1992). Pharmacol. Ther., 54, 63–128.
Dos Santos Pereira A and De Aquino Neto FR . (2003). Z. Naturforsch [C], 58, 201–206.
Downward J . (1998). Curr. Opin. Cell Biol., 10, 262–267.
Einspahr JG, Bowden GT and Alberts DS . (2003). Recent Results Cancer Res., 163, 151–164.
Epinat JC and Gilmore TD . (1999). Oncogene, 18, 6896–6909.
Fernandez MA, de las Heras B, Garcia MD, Saenz MT and Villar A . (2001). J. Pharm. Pharmacol., 53, 1533–1539.
Fournet A, Angelo A, Munoz V, Roblot F, Hocquemiller R and Cave A . (1992). J. Ethnopharmacol., 37, 159–164.
Furstenberger G and Marks F . (1985). Arachidonic Acid Metabolism and Tumor Promotion, Fischer SM and Slaga TJ (ed) Martinus Nijhoff Publishing: Boston, pp. 49–72.
Geetha T and Varalakshmi P . (1999). Mol. Cell. Biochem., 201, 83–87.
Geetha T and Varalakshmi P . (2001). J. Ethnopharmacol., 76, 77–80.
Guevara AP, Amor E and Russell G . (1996). Mutat. Res., 361, 67–72.
Gupta S and Mukhtar H . (2001). Skin Pharmacol. Appl. Skin Physiol., 14, 373–385.
Gupta S and Mukhtar H . (2002). Cancer Metast. Rev., 21, 363–380.
Gupta S, Ahmad N, Mohan RR, Husain MM and Mukhtar H . (1999). Cancer Res., 59, 2115–2120.
Hasmeda M, Kweifio-Okai G, Macrides T and Polya GM . (1999). Planta Med., 65, 14–18.
Hata K, Hori K and Takahashi S . (2002). J. Nat. Prod., 65, 645–648.
Hata K, Hori K and Takahashi S . (2003). J. Biochem. (Tokyo), 134, 441–445.
Herschman HR . (1994). Cancer Metast. Rev., 13, 241–256.
Hodges LD, Kweifio-Okai G and Macrides TA . (2003). Mol. Cell. Biochem., 252, 97–101.
Israel A . (1995). Trends Genet., 11, 203–205.
Kakuda R, Iijima T, Yaoita Y, Machida K and Kikuchi M . (2002). Phytochemistry, 59, 791–794.
Katiyar SK and Mukhtar H . (1997). Carcinogenesis, 18, 1911–1916.
Katiyar SK, Challa A, McCormick TS, Cooper KD and Mukhtar H . (1999). Carcinogenesis, 20, 2117–2124.
Katiyar SK, Agarwal R and Mukhtar H . (1996). Cancer Res., 56, 1023–1030.
Katiyar SK, Mohan RR, Agarwal R and Mukhtar H . (1997). Carcinogenesis, 18, 497–502.
Kim DJ, Shin DH, Ahn B, Kang JS, Nam KT, Park CB, Kim CK, Hong JT, Kim YB, Yun YW, Jang DD and Yang KH . (2003). Mutat. Res., 523–524, 99–107.
Liang YC, Tsai DC, Lin-Shiau SY, Chen CF, Ho CT and Lin JK . (2002). Nutr. Cancer, 42, 217–223.
Lin LC, Chou CJ and Kuo YC . (2001). J. Nat. Prod., 64, 674–676.
Luo J, Manning BD and Cantley LC . (2003). Cancer Cell, 4, 257–262.
Maniatis T . (1997). Science, 278, 818–819.
Mills GB, Kohn E, Lu Y, Eder A, Fang X, Wang H, Bast RC, Gray J, Jaffe R and Hortobagyi G . (2003). Semin. Oncol., 30, 93–104.
Miura K, Kikuzaki H and Nakatani N . (2001). Phytochemistry, 58, 1171–1175.
Mohan RR, Challa A, Gupta S, Bostwick DG, Ahmad N, Agarwal R, Marengo SR, Amini SB, Paras F, MacLennan GT, Resnick MI and Mukhtar H . (1999). Clin. Cancer Res., 5, 143–147.
Moriarity DM, Huang J, Yancey CA, Zhang P, Setzer WN, Lawton RO, Bates RB and Caldera S . (1998). Planta Med., 64, 370–372.
Nagaraj M, Sunitha S and Varalakshmi P . (2000). J. Appl. Toxicol., 20, 413–417.
Nakadate T, Aizu E, Yamamoto S, Fujiki H, Sugimura T and Kato R . (1985). Jpn. J. Pharmacol., 37, 253–258.
Nikiema JB, Vanhaelen-Fastre R, Vanhaelen M, Fontaine J, De Graef C and Heenen M . (2001). Phytother. Res., 15, 131–134.
Osaki M, Kase S, Adachi K, Takeda A, Hashimoto K and Ito H . (2004). J. Cancer Res. Clin. Oncol., 130, 8–14.
Rajic A, Kweifio-Okai G, Macrides T, Sandeman RM, Chandler DS and Polya GM . (2000). Planta Med., 66, 206–210.
Romashkova JA and Makarov SS . (1999). Nature, 401, 86–90.
Saeed MA and Sabir AW . (2002). Fitoterapia, 73, 417–420.
Saleem M, Alam A, Arifin S, Shah MS, Ahmed B and Sultana S . (2001). Pharmacol. Res., 43, 127–134.
Saleem R, Ahmad SI, Ahmed M, Faizi Z, Zikr-ur-Rehman S, Ali M and Faizi S . (2003). Biol. Pharm. Bull., 26, 41–46.
Seo HJ, Park KK, Han SS, Chung WY, Son MW, Kim WB and Surh YJ . (2002). Int. J. Cancer, 100, 456–462.
Smith WL, Garavito RM and DeWitt DL . (1996). J. Biol. Chem., 271, 33157–33160.
Sosa A . (1963). Bull. Soc. Chim. Bio. (Paris), 45, 117–126.
Stambolic V, Mak TW and Woodgett JR . (1999). Oncogene, 18, 6094–6103.
Sunitha S, Nagaraj M and Varalakshmi P . (2001). Fitoterapia, 72, 516–523.
Surh YJ . (2003). Nat. Rev. Cancer, 3, 768–780.
Thomas T and Thomas TJ . (2003). J. Cell. Mol. Med., 7, 113–126.
Ulubelen A, Topcu G and Johansson CB . (1997). J. Nat. Prod., 60, 1275–1280.
Verma AK, Shapas BG, Rice HM and Boutwell RK . (1979). Cancer Res., 39, 419–425.
Vidya L, Lenin M and Varalakshmi P . (2002). Phytother. Res., 16, 514–518.
Wada S, Iida A and Tanaka R . (2001). J. Nat. Prod., 64, 1545–1547.
Ziegler HL, Staerk D, Christensen J, Hviid L, Hagerstrand H and Jaroszewski JW . (2002). Antimicrob. Agents Chemother., 46, 1441–1446.
Acknowledgements
This work was supported by United States Public Health Service Grants RO1 CA 78809 and RO3 CA 99909.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saleem, M., Afaq, F., Adhami, V. et al. Lupeol modulates NF-κB and PI3K/Akt pathways and inhibits skin cancer in CD-1 mice. Oncogene 23, 5203–5214 (2004). https://doi.org/10.1038/sj.onc.1207641
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207641
Keywords
This article is cited by
-
Anti-inflammatory effects of immature Citrus unshiu fruit extracts via suppression of NF-κB and MAPK signal pathways in LPS-induced RAW264.7 macrophage cells
Food Science and Biotechnology (2024)
-
Lupeol synergizes with doxorubicin to induce anti-proliferative and apoptotic effects on breast cancer cells
DARU Journal of Pharmaceutical Sciences (2022)
-
RETRACTED ARTICLE: Lupeol inhibits migration and invasion of colorectal cancer cells by suppressing RhoA-ROCK1 signaling pathway
Naunyn-Schmiedeberg's Archives of Pharmacology (2020)