Abstract
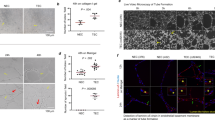
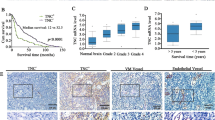
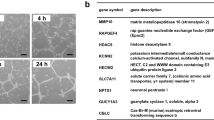
Angiomotin was identified by its ability to bind angiostatin and has been shown to mediate its activity in vitro. The family of angiomotin-like protein consists of three members that have coiled-coil domains and conserved c-terminal PDZ-binding motifs. We show here that expression of angiomotin in mouse aortic endothelial (MAE) cells results in stabilization of tubes in the Matrigel assay. Control tubes start to regress after 72 h, whereas MAE-angiomotin (MAE Amot) tubes were stable for over 30 days. In contrast, cells expressing a functional mutant lacking the PDZ protein interaction motif did not migrate and form tubes. Cells from the established tubes invaded into the solidified matrigel. We therefore tested whether angiomotin promotes endothelial invasion. In microcarrier-based invasion in vitro assay, angiomotin-expressing cells invaded collagen matrix and formed tube-like branches. This was confirmed in vivo as injection of MAE-Amot cells promoted tumor growth and invasion into surrounding muscle tissue. Injection of cells transfected with the functional mutant resulted in establishment of noninvasive tumors surrounded by a capsule of fibrous tissue. These tumors remained in constant size or dormant over 3 weeks. Zymogel analysis of the transfected cells did not reveal any differences in proteolytic activity. However, time-lapse photography showed a significant increase in random motility in MAE-Amot cells. We conclude that angiomotin may promote angiogenesis by both stimulating invasion as well as stabilizing established tubes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Auerbach R, Lewis R, Shinners B, Kubai L and Akhtar N . (2003). Clin. Chem., 49, 32–40.
Ballestrem C, Wehrle-Haller B, Hinz B and Imhof BA . (2000). Mol. Biol. Cell., 11, 2999–3012.
Bastaki M, Nelli EE, Dell'Era P, Rusnati M, Molinari-Tosatti MP, Parolini S, Auerbach R, Ruco LP, Possati L and Presta M . (1997). Arterioscler. Thromb. Vasc. Biol., 17, 454–464.
Bratt A, Wilson WJ, Troyanovsky B, Aase K, Kessler R, Meir EG and Holmgren L . (2002). Gene, 298, 69–77.
Edwards DR and Murphy G . (1998). Nature, 394, 527–528.
Eriksson K, Magnusson P, Dixelius J, Claesson-Welsh L and Cross MJ . (2003). FEBS Lett., 536, 19–24.
Hanahan D and Folkman J . (1996). Cell, 86, 353–364.
Holmgren L . (1996). Cancer Metast. Rev., 15, 241–245.
Kassis J, Lauffenburger DA, Turner T and Wells A . (2001). Semin. Cancer Biol., 11, 105–117.
Lawler J . (2002). J. Cell. Mol. Med., 6, 1–12.
Levchenko T, Aase K, Troyanovsky B, Bratt A and Holmgren L . (2003). J. Cell Sci., 116, 3803–3810.
Liotta LA . (1984). Am. J. Pathol., 117, 339–348.
Murphy G and Gavrilovic J . (1999). Curr. Opin. Cell. Biol., 11, 614–621.
Nehls V and Drenckhahn D . (1995). Microvasc. Res., 50, 311–322.
Nyberg P, Heikkila P, Sorsa T, Luostarinen J, Heljasvaara R, Stenman UH, Pihlajaniemi T and Salo T . (2003). J. Biol. Chem., Jun 20; 278 (25), 22404–22411.
Parker KK, Brock AL, Brangwynne C, Mannix RJ, Wang N, Ostuni E, Geisse NA, Adams JC, Whitesides GM and Ingber DE . (2002). FASEB J., 16, 1195–1204.
Pepper MS, Montesano R, Mandriota SJ, Orci L and Vassalli JD . (1996). Enzyme Protein, 49, 138–162.
Pintucci G, Bikfalvi A, Klein S and Rifkin DB . (1996). Semin. Thromb. Hemost., 22, 517–524.
Serini G, Ambrosi D, Giraudo E, Gamba A, Preziosi L and Bussolino F . (2003). EMBO J., 22, 1771–1779.
Staff AC . (2001). Scand. J. Clin. Lab. Invest., 61, 257–268.
Troyanovsky B, Levchenko T, Mansson G, Matvijenko O and Holmgren L . (2001). J. Cell Biol., 152, 1247–1254.
Wang N, Ostuni E, Whitesides GM and Ingber DE . (2002). Cell. Motil. Cytoskeleton, 52, 97–106.
Woodhouse EC, Chuaqui RF and Liotta LA . (1997). Cancer, 80, 1529–1537.
Vouret-Craviari V, Boquet P, Pouyssegur J and Van Obberghen-Schilling E . (1998). Mol. Biol. Cell, 9, 2639–2653.
Acknowledgements
This work was supported by American Skin Association, the Atorvastastin Research Award (Pfizer) and NIAMS, National Institutes of Health, Grant AR44947 (to J.L.A.), Emory Skin Disease Research Core Center P30, and National Institutes of Health Grants AR42687 and AR02030 (to J.L.A).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Levchenko, T., Bratt, A., Arbiser, J. et al. Angiomotin expression promotes hemangioendothelioma invasion. Oncogene 23, 1469–1473 (2004). https://doi.org/10.1038/sj.onc.1207264
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207264
Keywords
This article is cited by
-
The physiological role of Motin family and its dysregulation in tumorigenesis
Journal of Translational Medicine (2018)
-
Long non-coding RNA small nucleolar RNA host gene 12 (SNHG12) promotes cell proliferation and migration by upregulating angiomotin gene expression in human osteosarcoma cells
Tumor Biology (2016)
-
A vaccine targeting angiomotin induces an antibody response which alters tumor vessel permeability and hampers the growth of established tumors
Angiogenesis (2012)
-
The Duality of Angiogenesis: Implications for Therapy of Human Disease
Journal of Investigative Dermatology (2006)