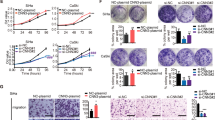
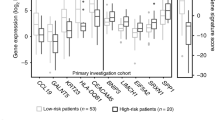
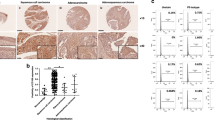
Abstract
Basic studies of oncogenesis have demonstrated that either the elevated production of particular oncogene proteins or the occurrence of qualitative abnormalities in oncogenes can contribute to neoplastic cellular transformation. The purpose of this study was to identify unique oncogenes that are differentially expressed in human cancers and characterize their functions in tumorigenesis. To discover new putative oncogenes, the differential display RT–PCR method was applied using normal cervical tissues, cervical cancer cell lines, cervical cancer tissues, and metastatic tissues. We identified a new human cervical cancer oncogene HCCR-2 that was overexpressed in various human tumors including leukemia, lymphoma, and carcinomas of the breast, kidney, ovary, stomach, colon, and uterine cervix. Ectopic expression of HCCR-2 resulted in direct tumorigenic conversions of NIH/3T3 and Rat1 fibroblasts. Nude mice injected with NIH/3T3 cells stably transfected with HCCR-2 formed tumors in 4 weeks. The resultant tumors display characteristics of an epithelial carcinoma. In HCCR-2 transfected NCI-H460 cells and RKO cells, stabilization of the p53 tumor suppressor occurred without genetic mutation and correlated with functional impairment, as indicated by the defective induction of p53-induced p21WAF1, MDM2, and bax. These results indicate that HCCR-2 probably represents a new oncogene that is related to tumorigenesis, functioning as a negative regulator of the p53 tumor suppressor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Aaronson SA, Jainchill JL and Todaro GJ . (1970). Proc. Natl. Acad. Sci. USA, 66, 1236–1243.
Ahn KY, Madsen KM, Tisher CC and Kone BC . (1991). Am. J. Physiol., 265, F792–F801.
Bishop JM . (1991). Cell, 64, 235–248.
Buick RN, Fry SE and Salmon SE . (1980). Cancer, 45, 1238–1242.
Busby-Earle RMC, Steel CM, Williams ARW, Cohen B and Bird CC . (1994). Br. J. Cancer, 69, 732–737.
Cherif D, Julier C, Delattre O, Derre J, Lathrop GM and Berger R . (1990). Proc. Natl. Acad. Sci. USA, 87, 6639–6643.
Cooper GM (ed) (1995). Oncogenes. Jones and Bartlett Publishers: Boston.
Crook T, Wrede D, Tidy JA, Mason WP, Evans DJ and Vousden KH . (1992). Lancet, 339, 1070–1073.
El-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW and Vogelstein B . (1993). Cell, 75, 817–825.
Farmer G, Bargonetti J, Zhu H, Friedman P, Prywes R and Prives C . (1992). Nature, 358, 83–86.
Hubert WG and Lambert PF . (1993). J. Virol., 67, 2932–2937.
Huibregtse JM, Scheffner M and Howley PM . (1994). Cold Spring Harber Symp. Quant. Biol., LIX, 237–245.
Kim JW, Lee CG, Cho YH, Kim JH, Kim SJ and Kim HK . (1996a). Gynecol. Oncol., 62, 230–240.
Kim J, Cha JH, Tisher CC and Madsen KM . (1996b). Am. J. Physiol. Renal Physiol., 270, F575–F592.
Leonard CJ, Canman CE and Kastan MB . (1995). Imp. Adv. Oncol., 33–42.
McLean IW and Nakane PK . (1974). J. Histochem. Cytochem., 22, 1077–1083.
Momand J, Zambetti GP, Olson DC, George D and Levine AJ . (1992). Cell, 69, 1237–1245.
Oltval ZN, Milliman CL and Korsmeyer SJ . (1993). Cell, 74, 609–619.
Puchtler H and Waldrop FW . (1978). Histochemistry, 57, 177–187.
Sambrook J, Fritsch EF and Maniatis T (eds) (1989). Cold Spring Harbor Laboratory: New York.
Sarnow P, Ho YS, Williams J and Levine AJ . (1982). Cell, 28, 387–394.
Weinberg RA . (1991). Science, 254, 1138–1146.
Weinberg RA . (1994). CA Cancer J. Clin., 44, 160–170.
Wernes BA, Levine AJ and Howley PM . (1990). Science, 248, 76–79.
Xiong Y, Hannon GJ, Zhang H, Casso D, Kobayashi R and Beach D . (1993). Nature, 366c 701–704.
Acknowledgements
Supported by a Grant (HMP-99-B-02-0002) of the 1999, Good Health R&D Project, Ministry of Health & Welfare, ROK. We thank JY Jung and W Kim for critically reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ko, J., Lee, Y., Hwang, S. et al. Identification and differential expression of novel human cervical cancer oncogene HCCR-2 in human cancers and its involvement in p53 stabilization. Oncogene 22, 4679–4689 (2003). https://doi.org/10.1038/sj.onc.1206624
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206624
Keywords
This article is cited by
-
Mitochondrial matrix protein LETMD1 maintains thermogenic capacity of brown adipose tissue in male mice
Nature Communications (2023)
-
Germline whole exome sequencing of a family with appendiceal mucinous tumours presenting with pseudomyxoma peritonei
BMC Cancer (2020)
-
Targeting HCCR expression resensitizes gastric cancer cells to chemotherapy via down-regulating the activation of STAT3
Scientific Reports (2016)
-
A panel of autoantibodies against multiple tumor-associated antigens in the immunodiagnosis of esophageal squamous cell cancer
Cancer Immunology, Immunotherapy (2016)
-
Hox B4 as potential marker of non-differentiated cells in human cervical cancer cells
Journal of Cancer Research and Clinical Oncology (2012)