Abstract
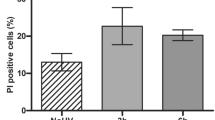
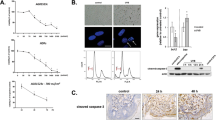
Nuclear DNA damage and death receptor (CD95) activation by ultraviolet-B radiation (UVB) play a major role in UVB-induced apoptosis. Removal of DNA damage combined with inhibition of death receptor activation resulted in pronounced but not complete suppression of apoptosis, indicating that a third independent pathway is involved. Since reactive oxygen species (ROS) cause apoptosis and are induced by UVB, the radical scavenger pyrrolidene-dithiocarbamate (PDTC) was used. PDTC prevented UVB-induced apoptosis partially, H2O2-induced cell death largely, but not CD95-mediated apoptosis. The same was observed for cytochrome c release from mitochondria, another important event during apoptosis. The proapoptotic protein Bid was cleaved upon exposure to UVB or to agonistic anti-CD95-antibodies, but not to H2O2, indicating that H2O2 uses a different pathway. The fact that PDTC neither inhibited CD95-mediated apoptosis nor affected UV-induced DNA damage indicated that ROS generated during UVB irradiation may directly trigger mitochondrial cytochrome c release, thereby contributing to apoptosis. Accordingly, complete inhibition of apoptosis was observed when in addition to DNA damage removal via photoreactivation and blockade of CD95 signaling by caspase-8 inhibitor zIETD, PDTC was added before UVB exposure. This indicates that DNA damage, death receptor activation and ROS formation contribute to UVB-induced apoptosis in an essential and independent way.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- BHA:
-
butylated hydroxyanisole
- CPD:
-
cyclobutane pyrimidine dimers
- NAC:
-
N-acetyl-cysteine
- PDTC:
-
pyrrolidene-dithiocarbamate
- ROS:
-
reactive oxygen species
- UV:
-
ultraviolet radiation
- UVB:
-
ultraviolet-B radiation
References
Aragane Y, Kulms D, Metze D, Kothny G, Pöppelmann B, Luger TA, Schwarz T . 1998 J. Cell Biol. 140: 171–182
Black HS . 1987 Photochem. Photobiol. 46: 213–221
Black HS, Lambert CR . 2001 Curr. Probl. Dermatol. 29: 140–156
Boldin MP, Varfolomeev EE, Pancer Z, Mett IL, Camonis JH, Wallach D . 1995 J. Biol. Chem. 270: 7795–7798
Bongarzone ER, Pasquini JM, Soto EF . 1995 J. Neuroscience Res. 41: 213–221
Buttke TM, Sandstrom PA . 1995 Free Radic. Res. 5: 389–397
Cadenas E, Davies KJ . 2000 Free Radic. Biol. Med. 29: 222–230
Chinnaiyan AM, Tepper CG, Sedlin MF, O'Rourke K, Kischkel FC, Hellbardt S, Krammer PH, Peter ME, Dixit VM . 1996 J. Biol. Chem. 271: 4961–4965
de Kok TMCM, ten Vaarwerk F, Zwingmann I, van Maanen JMS, Kleinjans JCS . 1994 Carcinogenesis 15: 1399–1404
de Laat WL, Jaspers NG, Hoeijmakers JH . 1999 Genes Dev. 13: 768–785
Desagher S, Osen-Sand A, Nichols A, Eskes R, Montessuit S, Lauper S, Maundrell K, Antonsson B, Martinou JC . 1999 J. Cell. Biol. 144: 891–901
Dreher F, Maibach H . 2001 Curr. Probl. Dermatol. 29: 157–164
Eker AP, Kooiman P, Hessels JKC, Yasui A . 1990 J. Biol. Chem. 265: 8009–8015
Farber JL . 1994 Environ. Health Perspect. 102: 10 17–24
Floyd RA . 1990 FASEB J. 4: 2587–2597
Gloster HM, Brodland DG . 1996 Dermatol. Surg. 22: 217–226
Hu Y, Benedict MA, Ding L, Nunez G . 1999 EMBO J. 18: 3586–3589
Kraemer KH, Lee MM, Andrews AD, Lambert WC . 1994 Arch. Dermatol. 130: 1018–1021
Kraemer KH . 1997 Proc. Natl. Acad. Sci., USA 94: 1–14
Kroemer G, Reed JC . 2000 Nat. Med. 6: 513–519
Kulms D, Pöppelmann B, Yarosh D, Luger TA, Krutmann J, Schwarz T . 1999 Proc. Natl. Acad. Sci. USA 96: 7974–7979
Kulms D, Schwarz T . 2000 Photodermatol. Photoimmunol. Photomed. 16: 195–201
Kuluncsics Z, Perdiz D, Brulay E, Muel B, Sage E . 1999 J. Photochem. Photobiol. B. 49: 71–80
Li H, Zhu H, Xu C, Yuan J . 1998 Cell 94: 491–501
Luo X, Budihardjo I, Zou H, Slaughter C, Wang X . 1998 Cell 94: 481–490
Masaki H, Atsumi T, Sakurai H . 1995 Biochem. Biophys. Res. Commun. 17: 474–479
Murphy G, Young AR, Wulf HC, Kulms D, Schwarz T . 2001 Exp. Dermatol. 10: 155–160
Patrick MH . 1977 Photochem. Photobiol. 25: 357–372
Petit-Frere C, Capulas E, Lowe JE, Koulu L, Marttila RJ, Jaspers NG, Clingen PH, Green MH, Arlett CF . 2000 J. Invest. Dermatol. 115: 687–693
Peus D, Vasa RA, Beyerle A, Meves A, Krautmacher C, Pittelkow MR . 1999 J. Invest. Dermatol. 112: 751–756
Rehemtulla A, Hamilton CA, Chinnaiyan AM, Dixit VM . 1997 J. Biol. Chem. 272: 25783–25786
Rosette C, Karin M . 1996 Science 274: 1194–1197
Schwarz A, Ständer S, Berneburg M, Böhm M, Kulms D, van Steeg H, Große-Heitmeyer K, Krutmann J, Schwarz T . 2002 Nat. Cell Biol. 4: 26–31
Sheikh MS, Antinore MJ, Huang Y, Fornace Jr AJ . 1998 Oncogene 17: 2555–2563
Stege H, Roza L, Vink AA, Grewe M, Ruzicka T, Grether-Beck S, Krutmann J . 2000 Proc. Natl. Acad. Sci. USA 97: 1790–1795
Tyrrell RM . 1994 Mol. Aspects Med. 15: 1–77
Tyrrell RM . 1995 Biochem. Soc. Symp. 61: 547–553
Wenk J, Brenneisen P, Meewes C, Wlaschek M, Peters T, Blaudschun R, Ma W, Kuhr L, Schneider L, Scharffetter-Kochanek K . 2001 Curr. Probl. Dermatol. 29: 83–94
Wolf P, Cox P, Yarosh DB, Kripke ML . 1995 J. Invest. Dermatol. 104: 287–292
Woodcock A, Magnus IA . 1976 Br. J. Dermatol. 95: 459–468
Yarosh D, Alas LG, Yee V, Oberyszyn A, Kibitel JT, Mitchell D, Rosenstein R, Spinowitz A, Citron M . 1992 Cancer Res. 52: 4227–4231
Yarosh D, Klein J . 1994 Trends Photochem. Photobiol. 3: 175–181
Zamzami N, Susin SA, Marchetti P, Hirsch T, Gomez-Monterrey I, Castedo M, Kroemer G . 1996 J. Exp. Med. 183: 1533–1544
Ziegler A, Jonason JS, Leffel DW, Simon JA, Sharma AW, Kimmelman J, Remington L, Jacks T, Brash DE . 1994 Nature 372: 773–776
Zou H, Li Y, Liu X, Wang X . 1999 J. Biol. Chem. 274: 11549–11556
Acknowledgements
The authors are grateful to D Yarosh, AGI-Dermatics for generous supply of Photosomes®. This work was supported by grants from the Federal Ministery of Education and Research (07UVB63A/5), the European Community (ENV4-CT97-0556) and the Interdisciplinary Center for Clinical Research (1ZKF, E10).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kulms, D., Zeise, E., Pöppelmann, B. et al. DNA damage, death receptor activation and reactive oxygen species contribute to ultraviolet radiation-induced apoptosis in an essential and independent way. Oncogene 21, 5844–5851 (2002). https://doi.org/10.1038/sj.onc.1205743
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205743
Keywords
This article is cited by
-
A review on cell damage, viability, and functionality during 3D bioprinting
Military Medical Research (2022)
-
Evaluation of Ultraviolet-Light (UV-A) Emitting Diodes Technology on the Reduction of Spiked Aflatoxin B1 and Aflatoxin M1 in Whole Milk
Food and Bioprocess Technology (2022)
-
Rosa davurica inhibits skin photoaging via regulating MAPK/AP-1, NF-κB, and Nrf2/HO-1 signaling in UVB-irradiated HaCaTs
Photochemical & Photobiological Sciences (2022)
-
A natural occurring bifunctional CPD/(6-4)-photolyase from the Antarctic bacterium Sphingomonas sp. UV9
Applied Microbiology and Biotechnology (2020)
-
Preservation effects of geniposidic acid on human keratinocytes (HaCaT) against UVB
Biomedical Dermatology (2018)