Abstract
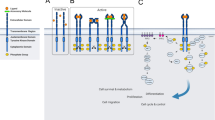
Many human cancers have been associated with the deregulation of receptor tyrosine kinases (RTK). However, the individual contribution of receptor-associated signaling proteins in cellular transformation and metastasis is poorly understood. To examine the role of RTK activated signal transduction pathways to processes involved in cell transformation, we have exploited the oncogenic derivative of the Met RTK (Tpr–Met). Unlike other RTKs, twin tyrosine residues in the carboxy-terminal tail of the Met oncoprotein and receptor are required for all biological and transforming activities, and a mutant lacking these tyrosines is catalytically active but non transforming. Using this mutant we have inserted oligonucleotide cassettes, each encoding a binding site for a specific signaling protein derived from other RTKs. We have generated variant forms of the Tpr–Met oncoprotein with the ability to bind individually to the p85 subunit of PI3′K, PLCγ, or to the Grb2 or Shc adaptor proteins. Variants that recruit the Shc or Grb2 adaptor proteins generated foci of morphologically transformed fibroblast cells and induced anchorage-independent growth, scattering of epithelial cells and experimental metastasis. In contrast, variants that bind and activate PI3′K or PLCγ failed to generate readily detectable foci. Although cell lines expressing the PI3′K variant grew in soft-agar, these cells were non metastatic. Using this unique RTK oncoprotein model, we have established that Grb2 or Shc dependent signaling pathways are sufficient for cell transformation and metastatic spread.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- RTK:
-
receptor tyrosine kinase
- SH2:
-
Src homology domain 2
- PTB:
-
phosphotyrosine binding domain
- PI3′K:
-
phosphatidylinositol 3′-kinase
- GST:
-
glutathione S-transferase
- PLCγ:
-
phospholipase Cγ
- IP:
-
immunoprecipitation
- FBS:
-
fetal bovine serum
References
Bardelli A, Basile ML, Audero E, Giordano S, Wennström S, Ménard S, Comoglio PM, Ponzetto C . 1999 Oncogene 18: 1139–1146
Bardelli A, Longati P, Gramaglia D, Stella MC, Comoglio PM . 1997 Oncogene 15: 3103–3111
Blume-Jensen P, Hunter T . 2001 Nature 411: 355–365
Chang J-S, Noh DY, Park IA, Kim MJ, Song H, Ryu SH, Suh P-G . 1997 Cancer Res. 57: 5465–5468
Cooper CS, Park M, Blair DG, Tainsky MA, Huebner K, Croce CM, Vande Woude GF . 1984 Nature 311: 29–33
Dankort D, Maslikowski B, Warner N, Kanno N, Kim H, Wang Z, Moran MF, Oshima RO, Cardiff RD, Muller WJ . 2001 Mol. Cell. Biol. 21: 1540–1551
Dankort DL, Wang Z, Blackmore V, Moran MF, Muller WJ . 1997 Mol. Cell. Biol. 17: 5410–5425
DeMali KA, Whiteford CC, Ulug ET, Kazlauskas A . 1997 J. Biol. Chem. 272: 9011–9018
Dikic I, Batzer AG, Blaikie P, Obermeier A, Ullrich A, Schlessinger J, Margolis B . 1995 J. Biol. Chem. 270: 15125–15129
Fixman ED, Fournier TM, Kamikura DM, Naujokas MA, Park M . 1996 J. Biol. Chem. 271: 13116–13122
Fixman ED, Holgado-Madruga M, Nguyen L, Kamikura DM, Fournier TM, Wong AJ, Park M . 1997 J. Biol. Chem. 272: 20167–20172
Fixman ED, Naujokas MA, Rodrigues GA, Moran MF, Park M . 1995 Oncogene 10: 237–249
Fournier TM, Kamikura D, Teng K, Park M . 1996 J. Biol. Chem. 271: 22211–22217
Fukazawa T, Miyake S, Band V, Band H . 1996 J. Biol. Chem. 271: 14554–14559
Gu H, Maeda H, Moon JJ, Lord JD, Yoakim M, Nelson BH, Neel BG . 2000 Mol. Cell. Biol. 20: 7109–7120
Jeffers M, Koochekpour S, Fiscella M, Sathyanarayana BK, Vande Woude GF . 1998 Oncogene 17: 2691–2700
Jeffers M, Rong S, Vande Woude GF . 1996 J. Mol. Med. 74: 505–513
Jimenez C, Jones DR, Rodriguez-Vaciana P, Gonzalez-Garcia A, Leonardo E, Wennström S, von Kobbe C, Toran JL, R.-Borlado L, Calvo V, Copin SG, Albar JP, Gaspar ML, Diez E, Marcos MAR, Downward J, Martinez-A C, Merida I, Carrera AC . 1998 EMBO J. 17: 743–753
Kamikura DM, Khoury H, Maroun C, Naujokas MA, Park M . 2000 Mol. Cell. Biol. 20: 3482–3496
Kamikura DM, Naujokas MA, Park M . 1996 Biochemistry 35: 1010–1017
Kashishian A, Cooper JA . 1993 Mol. Cell. Biol. 4: 49–57
Kazlauskas A, Durden DL, Cooper JA . 1991 Cell Regul. 2: 413–425
Kazlauskas A, Kashishian A, Cooper JA, Valius M . 1992 Mol. Cell. Biol. 12: 2534–2544
Kim HK, Kim JW, Zilberstein A, Margolis B, Kim JG, Schlessinger J, Rhee SG . 1991 Cell 65: 435–441
Klippel A, Escobedo M-A, Wachowicz MS, Apell G, Brown TW, Giedlin MA, Kavanaugh WM, Williams LT . 1998 Mol. Cell. Biol. 18: 5699–5711
Lock LS, Royal I, Naujokas MA, Park M . 2000 J. Biol. Chem. 275: 31536–31545
Lowenstein EJ, Daly RJ, Batzer AG, Li W, Margolis B, Lammers R, Ullrich A, Skolnik EY, Bar-Sagi D, Schlessinger J . 1992 Cell 70: 431–442
Maroun C, Holgado-Madruga M, Royal I, Naujokas MA, Fournier TM, Wong AJ, Park M . 1999 Mol. Cell. Biol. 19: 1784–1799
Meakin SO, MacDonald JIS, Gryz EA, Kubu CJ, Verdi JM . 1999 J. Biol. Chem. 274: 9861–9870
Meisner H, Czech MP . 1995 J. Biol. Chem. 270: 25332–25335
Nguyen L, Holgado-Madruga M, Maroun C, Fixman ED, Kamikura D, Fournier T, Charest A, Tremblay ML, Wong AJ, Park M . 1997 J. Biol. Chem. 272: 20811–20819
Noh D-Y, Shin SH, Rhee SG . 1995 Biochim. Biophys. Acta 1242: 99–114
Park M, Dean M, Cooper CS, Schmidt M, O'Brien SJ, Blair DG, Vande Woude GF . 1986 Cell 45: 895–904
Pawson T . 1995 Nature 373: 573–580
Pelicci G, Giordano S, Zhen Z, Salcini AE, Lanfrancone L, Bardelli A, Panayotou G, Waterfield MD, Ponzetto C, Pelicci PG, Comoglio PM . 1995 Oncogene 10: 1631–1638
Pelicci G, Lanfrancone L, Grignani F, McGlade J, Cavallo F, Forni G, Nicoletti L, Pawson T, Pelicci PG . 1992 Cell 70: 93–104
Ponzetto C, Bardelli A, Zhen Z, Maina F, dalla Zonca P, Giordano S, Graziani A, Panayotou G, Comoglio PM . 1994 Cell 77: 261–271
Ponzetto C, Zhen Z, Audero E, Maina F, Bardelli A, Basile ML, Giordano S, Narsimhan R, Comoglio P . 1996 J. Biol. Chem. 271: 14119–14123
Rodrigues GA, Naujokas MA, Park M . 1991 Mol. Cell. Biol. 11: 2962–2970
Rodrigues GA, Park M . 1993 Mol. Cell. Biol. 13: 6711–6722
Rodrigues GA, Park M . 1994 Curr. Opin. Genet. Dev. 4: 15–24
Royal I, Park M . 1995 J. Biol. Chem. 270: 27780–27787
Rozakis-Adcock M, McGlade J, Mbamalu G, Pelicci G, Daly R, Li W, Batzer A, Thomas S, Brugge J, Pelicci PG, Schlessinger J, Pawson T . 1992 Nature 360: 689–692
Sakaguchi K, Okabayashi Y, Kido Y, Kimura S, Matsumura Y, Inushima K, Kasuga M . 1998 Mol. Endocrinol. 12: 536–543
Salcini AE, McGlade J, Pelicci G, Nicoletti I, Pawson T, Pelicci PG . 1994 Oncogene 9: 2827–2836
Smith MR, Court DW, Kim H-K, Park JB, Rhee SG, Rhim JS, Kung H-F . 1998 Carcinogenesis 19: 177–185
Songyang Z, Shoelson SE, Chaudhuri M, Gish G, Pawson T, Haser WG, King F, Roberts T, Ratnofsky S, Lechleider RJ, Neel BG, Birge RB, Fajardo JE, Chou MM, Hanafusa H, Schaffhausen B, Cantley LC . 1993 Cell 72: 767–778
Webster MA, Hutchinson JN, Rauh MJ, Muthuswamy SK, Anton M, Tortorice CG, Cardiff RD, Graham FL, Hassell JA, Muller WJ . 1998 Mol. Cell. Biol. 18: 2344–2359
Weidner KM, Di Cesare S, Sachs M, Brinkmann V, Behrens J, Birchmeier W . 1996 Nature 384: 173–176
Weidner KM, Sachs M, Riethmacher D, Birchmeier W . 1995 Proc. Natl. Acad. Sci. USA 92: 2597–2601
Zhu H, Naujokas MA, Fixman ED, Torossian K, Park M . 1994 J. Biol. Chem. 269: 29943–29948
Acknowledgements
The authors are grateful to members of the Park laboratory for critical reading of this manuscript. This work was supported by an operating grant from the Canadian Institutes of Health Research awarded to M Park, and C Saucier was supported by post-doctoral fellowships from the Royal Victoria Hospital Research Institute and Fonds de la Recherche en Sante du Quebec.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saucier, C., Papavasiliou, V., Palazzo, A. et al. Use of signal specific receptor tyrosine kinase oncoproteins reveals that pathways downstream from Grb2 or Shc are sufficient for cell transformation and metastasis. Oncogene 21, 1800–1811 (2002). https://doi.org/10.1038/sj.onc.1205261
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205261
Keywords
This article is cited by
-
Met receptor-induced Grb2 or Shc signals both promote transformation of intestinal epithelial cells, albeit they are required for distinct oncogenic functions
BMC Cancer (2014)
-
The interactome of the amyloid β precursor protein family members is shaped by phosphorylation of their intracellular domains
Molecular Neurodegeneration (2009)
-
Overexpression of Grb2/HER2 signaling in Chinese gastric cancer: their relationship with clinicopathological parameters and prognostic significance
Journal of Cancer Research and Clinical Oncology (2009)
-
KIT oncoprotein interactions in gastrointestinal stromal tumors: therapeutic relevance
Oncogene (2007)
-
From Tpr-Met to Met, tumorigenesis and tubes
Oncogene (2007)