Abstract
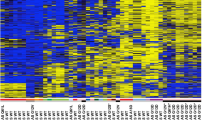
Loss of heterozygosity at 3p21.3 occurs in more than 90% of small cell lung carcinomas (SCLCs). The Ras association domain family 1 (RASSF1) gene cloned from the lung tumor suppressor locus 3p21.3 consists of two major alternative transcripts, RASSF1A and RASSF1C. Epigenetic inactivation of isoform A (RASSF1A) was observed in 40% of primary non-small cell lung carcinomas and in several tumor cell lines. Transfection of RASSF1A suppressed the growth of lung cancer cells in vitro and in nude mice. Here we have analysed the methylation status of the CpG island promoters of RASSF1A and RASSF1C in primary SCLCs. In 22 of 28 SCLCs (=79%) the promoter of RASSF1A was highly methylated at all CpG sites analysed. None of the SCLCs showed evidence for methylation of the CpG island of RASSF1C. The results suggest that hypermethylation of the CpG island promoter of the RASSF1A gene is associated with SCLC pathogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Antequera F, Boyes J, Bird A . 1990 Cell 62: 503–514
Bar-Sagi D, Feramisco JR . 1985 Cell 42: 841–848
Baylin SB, Herman JG . 2000 Trends Genet. 16: 168–174
Baylin SB, Herman JG, Graff JR, Vertino PM., Issa JP . 1998 Adv. Cancer Res. 72: 141–196
Belinsky SA, Nikula KJ, Palmisano WA, Michels R, Saccomanno G, Gabrielson E, Baylin SB, Herman JG . 1998 Proc. Natl. Acad. Sci. USA 95: 11891–11896
Chen CY, Liou J, Forman LW, Faller DV . 1998 J. Biol. Chem. 273: 16700–16709
Clark SJ, Harrison J, Paul CL, Frommer M . 1994 Nucleic Acids Res. 22: 2990–2997
Dammann R, Li C, Yoon JH, Chin PL, Bates S, Pfeifer GP . 2000 Nature Genet. 25: 315–319
Downward J . 1998 Curr. Opin. Genet. Dev. 8: 49–54
Girard L, Zochbauer-Muller S, Virmani AK, Gazdar AF, Minna JD . 2000 Cancer Res. 60: 4894–4906
Hibi K, Takahashi T, Yamakawa K, Ueda R, Sekido Y, Ariyoshi Y, Suyama M, Takagi H, Nakamura Y . 1992 Oncogene 7: 445–449
Hung J, Kishimoto Y, Sugio K., Virmani A, McIntire DD, Minna JD, Gazdar AF . 1995 JAMA 273: 558–563
Jones PA, Wolkowicz MJ, Rideout WM, Gonzales FA, Marziasz CM, Coetzee GA, Tapscott SJ . 1990 Proc. Natl. Acad. Sci. USA 87: 6117–6121
Jones PA, Laird PW . 1999 Nature Genet. 21: 163–167
Killary AM, Wolf ME, Giambernardi TA, Naylor SL . 1992 Proc. Natl. Acad. Sci. USA 89: 10877–10881
Kohno T, Yokota J . 1999 Carcinogenesis 20: 1403–1410
Kok K, Naylor SL, Buys CHCM . 1997 Adv. Cancer Res. 71: 27–92
Lerman MI, Minna JD . 2000 Cancer Res. 60: 6116–6133
Lindblad-Toh K, Tanenbaum DM, Daly MJ, Winchester E, Lui WO, Villapakkam A, Stanton SE, Larsson C, Hudson TJ, Johnson BE, Lander ES, Meyerson M . 2000 Nature Biotechnol. 18: 1001–1005
Mayo MW, Wang CY, Cogswell PC, Rogers-Graham KS, Lowe SW, Der CJ, Baldwin Jr. AS . 1997 Science 278: 1812–1815
Merlo A, Herman JG, Mao L, Lee DJ, Gabrielson E, Burger PC, Baylin SB, Sidransky D . 1995 Nature Med. 1: 686–692
Mitsudomi T, Viallet J, Mulshine JL, Linnoila RI, Minna JD., Gazdar AF . 1991 Oncogene 6: 1353–1362
Naylor SL, Johnson BE, Minna JD, Sakaguchi AY . 1987 Nature 329: 451–454
Otterson GA, Khleif SN, Chen W, Coxon AB, Kaye FJ . 1995 Oncogene 11: 1211–1216
Palmisano WA, Divine KK, Saccomanno G, Gilliland FD, Baylin SB, Herman JG, Belinsky SA . 2000 Cancer Res. 60: 5954–5958
Sekido Y, Ahmadian M, Wistuba II, Latif F, Bader S, Wei M-H, Duh F-M, Gazdar AF, Lerman MI, Minna JD . 1998 Oncogene 16: 3151–3157
Serrano, M, Lin AW, McCurrach ME, Beach D, Lowe SW . 1997 Cell 88: 593–602
Shao J, Sheng H, DuBois RN, Beauchamp RD . 2000 J. Biol. Chem. 275: 22916–22924
Shapiro GI, Park JE, Edwards CD, Mao L, Merlo A, Sidransky D, Ewen ME, Rollins BJ . 1995 Cancer Res. 55: 6200–6209
Sundaresan V, Ganly P, Hasleton P, Rudd R, Sinha G, Bleehen N, Rabbitts P . 1992 Oncogene 7: 1989–1997
Tang X, Khuri FR, Lee JJ, Kemp BL, Liu D, Hong WK, Mao L . 2000 J. Natl. Cancer Inst. 92: 1511–1516
Thiberville, L, Payne P, Vielkinds J, LeRiche J, Horsman D, Nouvet G, Palcic B, Lam S . 1995 Cancer Res. 55: 5133–5139
Todd S, Franklin WA, Varella-Garcia M, Kennedy T, Hilliker Jr CE, Hahner L, Anderson M, Wiest JS, Drabkin HA, Gemmill RM . 1997 Cancer Res. 57: 1344–1352
Vavvas D, Li X, Avruch J, Zhang X-F . 1998 J. Biol. Chem. 273: 5439–5442
Vos MD, Ellis CA, Bell A, Birrer MJ, Clark GJ . (2000). J. Biol. Chem. 275: 35669–35672
Wagner SN, Muller R, Boehm J, Putz B, Wunsch PH, Hofler H . 1993 Virchows Arch. B 63 325–329
Wei M-H, Latif F, Bader S, Kashuba V, Chen J-Y, Duh F-M, Sekido Y, Lee C-C, Geil L, Kuzmin I, Zabarovsky E, Klein G, Zbar B, Minna JD, Lerman MI . 1996 Cancer Res. 56: 1487–1492
Whang-Peng J, Kao-Shan CS, Lee EC, Bunn PA, Carney DN, Gazdar AF, Minna JD . 1982 Science 215: 181–182
Wingo PA, Ries LA, Giovino GA, Miller DS, Rosenberg HM, Shopland DR, Thun MJ, Edwards BK . 1999 J. Natl. Cancer Inst., 91: 675–690
Wistuba II., Behrens C, Milchgrub S, Bryant D, Hung J, Minna JD, Gazdar AF . 1999 Oncogene 18: 643–650
Wistuba II, Behrens C, Virmani AK, Mele G, Milchgrub S, Girard L, Fondon III, JW, Garner HR, McKay B, Latif F, Lerman MI, Lam S, Gazdar AF, Minna JD . 2000 Cancer Res. 60: 1949–1960
Wu W, Kemp BL, Proctor ML, Gazdar AF, Minna JD, Hong WK, Mao L . 1999 Cancer Res. 59: 1846–1851
Xiong Z, Laird PW . 1997 Nucleic Acids Res. 25: 2532–2534
Yamakawa K, Takahashi T, Horio Y, Murata Y, Takahashi E, Hibi K, Yokoyama S, Ueda R, Nakamura Y . 1993 Oncogene 8: 327–330
Yatabe Y, Konishi H, Mitsudomi T, Nakamura S, Takahashi T . 2000 Am. J. Pathol. 157: 985–993
Yokota J, Wada M, Shimosato Y, Terada M, Sugimura T . 1987 Proc. Natl. Acad. Sci. USA 84: 9252–9256
Acknowledgements
This work was supported by a grant from the University of California Tobacco Related Disease Research Program (9RT-0175) to GP Pfeifer.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dammann, R., Takahashi, T. & Pfeifer, G. The CpG island of the novel tumor suppressor gene RASSF1A is intensely methylated in primary small cell lung carcinomas. Oncogene 20, 3563–3567 (2001). https://doi.org/10.1038/sj.onc.1204469
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204469
Keywords
This article is cited by
-
The epigenetic regulatory mechanism of PIWI/piRNAs in human cancers
Molecular Cancer (2023)
-
Diagnostic value of SHOX2, RASSF1A gene methylation combined with CEA level detection in malignant pleural effusion
BMC Pulmonary Medicine (2023)
-
MFSD4A inhibits the malignant progression of nasopharyngeal carcinoma by targeting EPHA2
Cell Death & Disease (2022)
-
ZAR1 is a novel epigenetically inactivated tumour suppressor in lung cancer
Clinical Epigenetics (2017)
-
Epigenetic silencing of downstream genes mediated by tandem orientation in lung cancer
Scientific Reports (2017)