Abstract
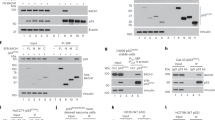
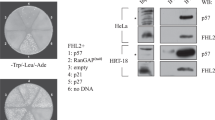
A recently discovered potential tumor suppressor protein, Zac1, was previously shown to promote cell cycle arrest and apoptosis, and to act as a positive or negative transcriptional cofactor for nuclear receptors. Since these activities are common to Zac1 and p53, we tested for a functional interaction between these two proteins by investigating possible effects of Zac1 on the transcriptional activator function of p53. Zac1 specifically enhanced the activity of p53-responsive promoters in cells expressing wild type p53. The same promoters were not activated by Zac1 in cells lacking functional p53, but the Zac1 effect was restored by co-expression of p53. Zac1 bound to p53 and enhanced the activity of p53 or its N-terminal transcriptional activation domain fused to the DNA binding domain of Gal4. These results indicate that Zac1 served as a transcriptional coactivator for p53. The enhancement of p53 activity by Zac1 was much more dramatic in HeLa cells than in other cell lines tested. HeLa cells express human papillomavirus type 18 E6 protein which inactivates and causes the degradation of p53. Physical and functional interactions observed between Zac1 and E6 protein indicated that the dramatic activity of Zac1 in HeLa cells was due not only to Zac1's coactivator effect on p53, but also to the ability of Zac1 to reverse E6 inhibition of p53.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Bartek J, Iggo R, Gannon J, Lane DP . 1990 Oncogene 5: 893–899
Bhat MK, Yu C, Yap N, Zhan Q, Hayashi Y, Seth P, Cheng S . 1997 J. Biol. Chem. 272: 28989–28993
Bunz F, Dutriaux A, Lengauer C, Waldman T, Zhou S, Brown JP, Sedivy JM, Kinzler KW, Vogelstein B . 1998 Science 282: 1497–1501
Casey G, Lo-Hsueh M, Lopez ME, Vogelstein B, Stanbridge EJ . 1991 Oncogene 6: 1791–1797
Chen D, Ma H, Hong H, Koh SS, Huang S-M, Schurter BT, Aswad DW, Stallcup MR . 1999 Science 284: 2174–2177
Desaintes C, Demeret C, Goyat S, Yaniv M, Thierry F . 1997 EMBO J. 16: 504–514
El-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW, Vogelstein B . 1993 Cell 75: 817–825
Harvey M, Sands AT, Weiss RS, Hegi ME, Wiseman RW, Pantazis P, Giovanella BC, Tainsky MA, Bradley A, Donehower LA . 1993 Oncogene 8: 2457–2467
Hoffmann A, Ciani E, Houssami S, Brabet P, Journot L, Spengler D . 1998 Ann. N. Y. Acad. Sci. 865: 49–58
Hong H, Kohli K, Trivedi A, Johnson DL, Stallcup MR . 1996 Proc. Natl. Acad. Sci. USA 93: 4948–4952
Huang S-M, Stallcup MR . 2000 Mol. Cell Biol. 20: 1855–1867
Kas K, Voz ML, Hensen K, Meyen E, Van de Ven WJ . 1998 J. Biol. Chem. 273: 23026–23032
Kern SE, Pietenpol JA, Thiagalingam S, Seymour A, Kinzler KW, Vogelstein B . 1992 Science 256: 827–830
Levine AJ . 1997 Cell 88: 323–331
Little JB, Nove J, Dahlberg WK, Troilo P, Nichols WW, Strong LC . 1987 Cancer Res. 47: 4229–4234
Matlashewski G, Banks L, Pim D, Crawford L . 1986 Eur. J. Biochem. 154: 665–672
May P, May E . 1999 Oncogene 18: 7621–7636
Nigro JM, Baker SJ, Preisinger AC, Jessup JM, Hostetter R, Cleary K, Bigner SH, Davidson N, Baylin S, Devilee P, Glover T, Collins FS, Weston A, Modali R, Harris CC, Vogelstein B . 1989 Nature 342: 705–708
Paech K, Webb P, Kuiper GGJM, Nilsson S, Gustafsson J-A, Kushner PJ, Scanlan TS . 1997 Science 277: 1508–1510
Patel D, Huang S-M, Baglia LA, McCance DJ . 1999 EMBO J. 18: 5061–5072
Runnebaum IB, Nagarajan M, Bowman M, Soto D, Sukumar S . 1991 Proc. Natl. Acad. Sci. USA 88: 10657–10661
Scheffner M, Werness BA, Huibregtse JM, Levine AJ, Howley PM . 1990 Cell 63: 1129–1136
Schwarz E, Freese UK, Gissmann L, Mayer W, Roggenbuck B, Stremlau A, Zur HH . 1985 Nature 314: 111–114
Spengler D, Villalba M, Hoffmann A, Pantaloni C, Houssami S, Bockaert J, Journot L . 1997 EMBO J. 16: 2814–2825
Tam SW, Theodoras AM, Shay JW, Draetta GF, Pagano M . 1994 Oncogene 9: 2663–2674
Thomas M, Pim D, Banks L . 1999 Oncogene 18: 7690–7700
Varrault A, Ciani E, Apiou F, Bilanges B, Hoffmann A, Pantaloni C, Bockaert J, Spengler D, Journot L . 1998 Proc. Natl. Acad. Sci. USA 95: 8835–8840
Werness BA, Levine AJ, Howley PM . 1990 Science 248: 76–79
Wu RC, Schonthal AH . 1997 J. Biol. Chem. 272: 29091–29098
Yu CL, Driggers P, Barrera-Hernandez G, Nunez SB, Segars JH, Cheng S . 1997 Biochem. Biophys. Res. Commun. 239: 617–620
Zimmermann H, Degenkolbe R, Bernard HU, O'Connor MJ . 1999 J. Virol. 73: 6209–6219
Acknowledgements
We thank Dr W El-Deiry (University of Pennsylvania) for providing plasmids used in this study. This work was supported by US Public Health Service grants DK55274 to MR Stallcup and CA74278 to AH Schönthal from the National Institutes of Health. S-M Huang was supported by a predoctoral fellowship from the Defense Department, Taiwan, Republic of China.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Huang, SM., Schönthal, A. & Stallcup, M. Enhancement of p53-dependent gene activation by the transcriptional coactivator Zac1. Oncogene 20, 2134–2143 (2001). https://doi.org/10.1038/sj.onc.1204298
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204298
Keywords
This article is cited by
-
PLAGL1 is associated with prognosis and cell proliferation in pancreatic adenocarcinoma
BMC Gastroenterology (2023)
-
Gene expression profiling identifies the role of Zac1 in cervical cancer metastasis
Scientific Reports (2020)
-
Metformin causes cancer cell death through downregulation of p53-dependent differentiated embryo chondrocyte 1
Journal of Biomedical Science (2018)
-
The cytotoxic mechanism of epigallocatechin gallate on proliferative HaCaT keratinocytes
Journal of Biomedical Science (2017)
-
PLAGL1: an important player in diverse pathological processes
Journal of Applied Genetics (2017)