Abstract
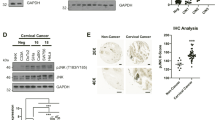
Cdc25A is a tyrosine phosphatase that is involved in the regulation of the G1/S phase transition by activating cyclin E/Cdk2 and cyclin A/Cdk2 complexes through removal of inhibitory phosphorylations. The E6 and E7 oncoproteins of the high-risk human papillomaviruses (HPV) interact with and functionally abrogate the p53 and pRB proteins, respectively. In the present study we have investigated the regulation of the Cdc25A promoter during G1 and S-phases of the cell cycle and by the HPV-16 E7 oncoprotein. Serum induction leads to a derepression of the Cdc25A promoter and can be mediated through two E2F binding sites, E2F-A and E2F-C. While E2F-A is by both E2F1 and E2F4, E2F-C is regulated only by E2F1. The Cdc25A promoter is transactivated by the E7 oncogene of HPV-16. Furthermore, Cdc25A levels are highly increased in E7-expressing cell lines. Inducible expression of E7 leads to an immediate increase in Cdc25A protein levels. These data suggest that Cdc25A may be a critical target of HPV-16 E7 in the disruption of the G1/S phase transition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Arata Y, Fujita M, Ohtani K, Kijima S and Kato JY. . 2000 J. Biol. Chem. 275: 6337–6345.
Blomberg I and Hoffmann I. . 1999 Mol. Cell. Biol. 19: 6183–6194.
Botz J, Zerfass-Thome K, Spitkovsky D, Delius H, Vogt B, Eilers M, Hatzigeorgiou A and Jansen-Durr P. . 1996 Mol. Cell. Biol. 16: 3401–3409.
Boyer SN, Wazer DE and Band V. . 1996 Cancer Res. 56: 4620–4624.
Chen X and Prywes R. . 1999 Mol. Cell. Biol. 19: 4695–4702.
Crook T, Morgenstern JP, Crawford L and Banks L. . 1989 EMBO J. 8: 513–519.
Dalton S. . 1992 EMBO J. 11: 1797–1804.
DeGregori J, Leone G, Miron A, Jakoi L and Nevins JR. . 1997 Proc. Natl. Acad. Sci. USA 94: 7245–7250.
Funk JO, Waga S, Harry JB, Espling E, Stillman B and Galloway DA. . 1997 Genes Dev. 11: 2090–2100.
Galaktionov K and Beach D. . 1991 Cell 67: 1181–1194.
Galaktionov K, Lee AK, Eckstein J, Draetta G, Meckler J, Loda M and Beach D. . 1995 Science 269: 1575–1577.
Gasparotto D, Maestro R, Piccinin S, Vukosavljevic T, Barzan L, Sulfaro S and Boiocchi M. . 1997 Cancer Res. 57: 2366–2368.
Hoffmann I, Clarke PR, Marcote MJ, Karsenti E and Draetta G. . 1993 EMBO J. 12: 53–63.
Hoffmann I, Draetta G and Karsenti E. . 1994 EMBO J. 13: 4302–4310.
Hoppe-Seyler F and Butz K. . 1992 Nucl. Acids Res. 20: 6701–6706.
Iavarone A and Massague J. . 1999 Mol. Cell. Biol. 19: 916–922.
Ioannou PA, Amemiya CT, Garnes J, Kroisel PM, Shizuya H, Chen C, Batzer MA and de Jong PJ. . 1994 Natl. Genet. 6: 84–89.
Karlsson C, Katich S, Hagting A, Hoffmann I and Pines J. . 1999 J. Cell. Biol. 146: 573–584.
Lam EW, Morris JD, Davies R, Crook T, Watson RJ and Vousden KH. . 1994 EMBO J. 13: 871–878.
Lammer C, Wagerer S, Saffrich R, Mertens D, Ansorge W and Hoffmann I. . 1998 J. Cell. Sci. 111: 2445–2453.
Lee JO, Russo AA and Pavletich NP. . 1998 Nature 391: 859–865.
Lukas J, Petersen BO, Holm K, Bartek J and Helin K. . 1996 Mol. Cell. Biol. 16: 1047–1057.
Nilsson I and Hoffmann I. . 2000 Prog. Cell Cycle Res., Meijer L (ed.). 4: 107–115.
Ohtani K, DeGregori J and Nevins JR. . 1995 Proc. Natl. Acad. Sci. USA 92: 12146–12150.
Pagano M, Pepperkok R, Verde F, Ansorge W and Draetta G. . 1992 EMBO J. 11: 961–971.
Peng CY, Graves PR, Thoma RS, Wu Z, Shaw AS and Piwnica-Worms H. . 1997 Science 277: 1501–1505.
Sadhu K, Reed SI, Richardson H and Russell P. . 1990 Proc. Natl. Acad. Sci. USA 87: 5139–5143.
Sanchez Y, Wong C, Thoma RS, Richman R, Wu Z, Piwnica-Worms H and Elledge SJ. . 1997 Science 277: 1497–1501.
Schulze A, Zerfass K, Spitkovsky D, Middendorp S, Berges J, Helin K, Jansen-Durr P and Henglein B. . 1995 Proc. Natl. Acad. Sci. USA 92: 11264–11268.
Spitkovsky D, Jansen-Dürr P, Karsenti E and Hoffmann I. . 1996 Oncogene 12: 2549–2554.
Vigo E, Muller H, Prosperini E, Hateboer G, Cartwright P, Moroni MC and Helin K. . 1999 Mol. Cell. Biol. 19: 6379–6395.
zur Hausen H. . 1999 Semin. Cancer Biol. 9: 405–411.
Acknowledgements
We are grateful to H zur Hausen for support and helpful comments on the manuscript. L Banks, N Dyson, P Jansen-Dürr, G Superti-Furga and K Vousden are thanked for providing reagents. We thank F Rösl and C O'Loughlin for critically reading the manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Katich, S., Zerfass-Thome, K. & Hoffmann, I. Regulation of the Cdc25A gene by the human papillomavirus Type 16 E7 oncogene. Oncogene 20, 543–550 (2001). https://doi.org/10.1038/sj.onc.1204130
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204130
Keywords
This article is cited by
-
Cyclin-dependent kinases and CDK inhibitors in virus-associated cancers
Infectious Agents and Cancer (2020)
-
Transcriptional regulation of genes involved in keratinocyte differentiation by human papillomavirus 16 oncoproteins
Archives of Virology (2015)
-
Increased expression of RRM2 by human papillomavirus E7 oncoprotein promotes angiogenesis in cervical cancer
British Journal of Cancer (2014)
-
Human papillomavirus oncoproteins: pathways to transformation
Nature Reviews Cancer (2010)
-
High-risk human papillomavirus type 16 E7 oncogene associates with Cdc25A over-expression in oral squamous cell carcinoma
Virchows Archiv (2007)