Abstract
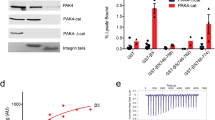
The serine threonine kinase protein kinase B regulates cellular activities as diverse as glycogen metabolism and apoptosis. Full activation of protein kinase B requires 3-phosphoinositides and dual phosphorylation on threonine-308 and serine-473. CaM-K kinase and 3-phosphoinositide dependent-kinase-1 phosphorylate threonine-308. Integrin-linked kinase reportedly phophorylates serine-473. Consistent with this, in a model COS cell system we show that expression of wild-type integrin-linked kinase promotes the wortmannin sensitive phosphorylation of serine-473 of protein kinase B and its downstream substrates, and inhibits C2-ceramide induced apoptosis. In contrast, integrin-linked kinase mutated in a lysine residue critical for function in protein kinases is inactive in these experiments, and furthermore, acts dominantly to block serine-473 phosphorylation induced by ErbB4. However, alignment of analogous sequences from different species demonstrates that integrin-linked kinase is not a typical protein kinase and identifies a conserved serine residue which potentially regulates kinase activity in a phosphorylation dependent manner. Mutation of this serine to aspartate or glutamate, but not alanine, in combination with the inactivating lysine mutation restores integrin-linked kinase dependent phosphorylation of serine-473 of protein kinase B. These data strongly suggest that integrin-linked kinase does not possess serine-473 kinase activity but functions as an adaptor to recruit a serine-473 kinase or phosphatase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Andjelkovic M, Jakubowicz T, Cron P, Ming XF, Han JW and Hemmings BA. . 1996 Proc. Natl. Acad. Sci. 93: 5699–5704.
Andjelkovic M, Alessi DR, Meier R, Fernandez A, Lamb NJC, Frech M, Cron P, Cohen P, Lucocq JM and Hemmings BA. . 1997 J. Biol. Chem. 272: 31515–31524.
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P and Hemmings BA. . 1996 EMBO J. 15: 6541–6551.
Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB and Cohen P. . 1997a Curr. Biol. 7: 261–269.
Alessi DR, Deak M, Casamayoy A, Caudwell FB, Morrice N, Norman DG, Gaffney P, Reece CB, MacDougal CN, Harbison D, Ashworth A and Bownes M. . 1997b Curr. Biol. 7: 776–789.
Balendran A, Casamayor C, Deak M, Paterson A, Gaffney P, Currie R, Downes CP and Alessi D. . 1999 Curr. Biol. 9: 393–404.
Bellacosa A, Testa JR, Staal SP and Tsichlis PN. . 1991 Science 254: 274–277.
Bellacosa A, Chan TO, Ahmed NN, Datta K, Malstrom S, Stokoe D, McCormick F, Feng J and Tsichlis P. . 1998 Oncogene 17: 313–325.
Blume-Jensen P, Janknecht R and Hunter T. . 1998 Curr. Biol. 8: 779–782.
Brunet A, Bonni A, Zigmond M, Lin M, Juo P, Hu L, Anderson M, Arden K, Blenis J and Greenberg M. . 1999 Cell 96: 857–868.
Cardone MH, Roy N, Stennicke HR, Salvesen GS, Franke TF, Stanbridge E, Frisch S and Reed JC. . 1998 Science 282: 1318–1321.
Carrera AC, Alexandrov K and Roberts TM. . 1993 Proc. Natl. Acad. Sci. 90: 442–446.
Chao DT and Korsmeyer SJ. . 1998 Annu. Rev. Immunol. 16: 395–419.
Coffer PJ and Woodgett JR. . 1991 Eur. J. Biochem. 201: 475–481.
Coffer PJ, Jin J and Woodgett JR. . 1998 Biochem. J. 335: 1–13.
Cohen P, Alessi DR and Cross DAE. . 1997 FEBS Lett. 410: 3–10.
Cook D, Fry MJ, Hughes K, Sumathipala R, Woodgett JR and Dale TC. . 1996 EMBO J. 15: 4526–4536.
Cross DAE, Alessi DR, Cohen P, Andjelkovich M and Hemmings BA. . 1995 Nature 378: 785–789.
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y and Greenberg ME. . 1997 Cell 91: 231–241.
Delcommenne M, Tan C, Gray V, Rue L, Woodgett J and Dedhar S. . 1998 Proc. Natl. Acad. Sci. 95: 11211–11216.
Dimmeler S, Assmus B, Hermann C, Haedeler J and Zeier AM. . 1998 Circ. Res. 83: 334–341.
Dudek H, Datta SR, Franke TF, Birnbaum MJ, Yao R, Cooper GM, Segal RA, Kaplan DR and Greenberg ME. . 1997 Science 275: 661–665.
Elenius K, Choi CJ, Paul S, Santiestevan EE and Klagsbrun M. . 1999 Oncogene 18: 2607–2615.
Foster DC and Garbers DL. . 1998 J. Biol. Chem. 273: 16311–16318.
Franke TF, Kaplan DR, Cantley LC and Toker A. . 1997 Science 275: 665–668.
Frisch SM and Francis H. . 1994 J. Cell. Biol. 124: 619–626.
Frisch SM and Ruoslahti E. . 1997 Curr. Opin. Cell Biol. 9: 701–706.
Gibbs CS and Zoller MJ. . 1991 J. Biol. Chem. 266: 8923–8931.
Gingras AC, Kennedy SG, Oleary MA, Sonenberg N and Hay N. . 1998 Genes. Dev. 12: 502–513.
Guilherme A and Czech MP. . 1998 J. Biol. Chem. 273: 33119–33122.
Hanks SK and Hunter T. . 1995 FASEB J. 9: 576–596.
Hannigan GE, Leung-Hagesteijn C, Fitz-Gibbon L, Coppolino MG, Radeva G, Filmus J, Bell JC and Dedhar S. . 1996 Nature 379: 91–96.
Her JH, Lakhani S, Zu K, Vila J, Dent P, Sturgill TW and Weber MJ. . 1993 Biochem. J. 296: 25–31.
Huang X and Miller W. . 1991 Adv. Appl. Math. 12: 337–357.
Hubbard SR, Wei L, Ellis L and Henderson WA. . 1994 Nature 372: 746–754.
Jones PJ, Jakubowicz T, Pitossi FJ, Maurer F and Hemmings BA. . 1991 Proc. Natl. Acad. Sci. 88: 4171–4175.
Kauffman-Zeh A, Rodriguez-Viciana P, Ulrich E, Gilbert C, Coffer P, Downward J and Evan G. . 1997 Nature 385: 544–548.
Khwaja A, Rodriguez-Viciana P, Wennstrom S, Warne PH and Downward J. . 1997 EMBO J. 16: 2783–2793.
King WG, Mattaliano MD, Chan TO, Tsichlis PN and Brugge JS. . 1997 Mol. Cell Biol. 17: 4406–4418.
Klippel A, Kavanaugh WM, Pot D and Williams LT. . 1997 Mol. Cell Biol. 17: 338–344.
Kitamura T, Ogawa W, Sakaue H, Hino Y, Kuroda S, Takata M, Matsumoto M, Maeda T, Konishi H, Kikkawa U and Kasuga M. . 1998 Mol. Cell Biol. 18: 3708–3717.
Kulik G, Klippel A and Weber MJ. . 1997 Mol. Cell Biol. 17: 1595–1606.
Marte BM and Downward J. . 1997 Trends Biochem. Sci. 22: 355–358.
Meier R, Alessi DR, Cron P, Andjelkovic M and Hemmings BA. . 1997 J. Biol. Chem. 272: 30491–30497.
Millward TA, Zolnierowicz S and Hemmings BA. . 1999 Trends Biochem. Sci. 24: 186–191.
Pearson RB, Dennis PB, Han JW, Williamson NA, Kozma SC, Wettenhall REH and Thomas G. . 1995 EMBO J. 14: 5278–5287.
Potter LR and Hunter T. . 1998 Mol. Cell. Biol. 18: 2164–2172.
Sambrook J, Fritsch E and Maniatis T. . 1987 Molecular Cloning: A laboratory Manual, 2nd edn. Cold Spring Harbor Laboratory Press 16.20–16.22.
Sawyer C, Hiles I, Page M, Crompton M and Dean C. . 1998 Oncogene 17: 919–924.
Scheid MP and Duronio V. . 1998 Proc. Natl. Acad. Sci. 95: 7439–7444.
Stephens L, Anderson K, Stokoe D, Erdjument-Bromage H, Painter GF, Holmes AB, Gaffney PR, Reese CB, McCormick F, Tempst P, Coadwell J and Hawkins PT. . 1998 Science 279: 710–714.
Thompson JD, Higgins DG and Gibson TJ. . 1994 Nucleic Acids Res. 22: 4673–4680.
Ueki K, Yamamoto-honda R, Kaburagi Y, Yamauchi T, Tobe K, Burgering BMT, Coffer PJ, Komuro I, Akanuma Y, Yazaki Y and Kadowaki T. . 1998 J. Biol. Chem. 273: 5315–5322.
Ui M, Okada T and Hazeki O. . 1995 Trends Biochem. Sci. 20: 303–307.
Van Weeren PC, Debruyn KMT, Devriessmits AMM, Vanlint J and Burgering BMT. . 1998 J. Biol. Chem. 273: 13150–13156.
Yano S, Tokumitsu H and Soderling TR. . 1998 Nature 396: 584–587.
Zhou H, Summers SA, Birnbaum MJ and Pittman RN. . 1998 J. Biol. Chem. 273: 16568–16575.
Acknowledgements
We thank Simon Barry, Harren Jhoti, Rob Harris and Peter Soden for helpful discussions and technical advice. This work was supported by a MRC collaborative studentship award between GlaxoWellcome and Cambridge University (DK Lynch) and a grant from the Association for International Cancer Research (PAW Edwards).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lynch, D., Ellis, C., Edwards, P. et al. Integrin-linked kinase regulates phosphorylation of serine 473 of protein kinase B by an indirect mechanism. Oncogene 18, 8024–8032 (1999). https://doi.org/10.1038/sj.onc.1203258
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1203258
Keywords
This article is cited by
-
Integrin-linked kinase (ILK): the known vs. the unknown and perspectives
Cellular and Molecular Life Sciences (2022)
-
Influence of integrins on thrombus formation: a road leading to the unravelling of DVT
Molecular and Cellular Biochemistry (2021)
-
Integrin-Linked Kinase Activation Prevents Ventricular Arrhythmias Induced by Ischemia/Reperfusion Via Inhibition of Connexin 43 Remodeling
Journal of Cardiovascular Translational Research (2021)
-
Distinct functions of AKT isoforms in breast cancer: a comprehensive review
Cell Communication and Signaling (2019)
-
ILK–PI3K/AKT pathway participates in cutaneous wound contraction by regulating fibroblast migration and differentiation to myofibroblast
Laboratory Investigation (2016)