Abstract
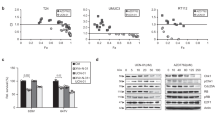
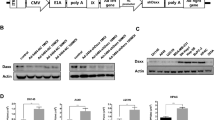
An adenovirus mutant lacking the expression of the large E1B protein (ΔE1B) has been reported to replicate selectively in cells lacking the expression of functionally wild-type (wt) p53. Based on these results the ΔE1B or ONYX-015 virus has been proposed to be an oncolytic virus which might be useful to treat p53-deficient tumors. Recently however, contradictory results have been published indicating that p53-dependent cell death is required for productive adenovirus infection. Since there is an urgent need for new methods to treat aggressive, mutant p53-expressing primary tumors and their metastases we carefully examined adenovirus replication in human cells to determine whether or not the ΔE1B virus can be used for tumor therapy. The results we present here show that not all human tumor cell lines take up adenovirus efficiently. In addition, we observed inhibition of the expression of adenovirus early proteins in tumor cells. We present evidence that these two factors rather than the p53 status of the cell determine whether adenovirus infection results in lytic cell death. Furthermore, the results we obtained by infecting a panel of different tumor cell lines show that viral spread of the ΔE1B is strongly inhibited in almost all p53-proficient and -deficient cell lines compared to the wt virus. We conclude that the efficiency of the ΔE1B virus to replicate efficiently in tumor cells is determined by the ability to infect cells and to express the early adenovirus proteins rather than the status of p53.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Babiss LE and Ginsberg HS. . 1984 J. Virol. 50: 202–212.
Bergelson JM, Cunningham JA, Droguett G, Kurt-Jones EA, Krithivas A, Hong JS, Horwitz MS, Crowell RL and Finberg RW. . 1997 Science 275: 1320–1323.
Bischoff JR, Kirn DH, Williams A, Heise C, Horn S, Muna M, Ng L, Nye JA, Sampson-Johannes A, Fattaey A and McCormick F. . 1996 Science 274: 373–376.
Chandar N, Billig B, McMaster J and Novak J. . 1992 Br. J. Cancer 65: 208–214.
Diller L, Kassel J, Nelson CE, Gryka MA, Litwak G, Gebhardt M, Bressac B, Ozturk M, Baker SJ, Vogelstein B and Friend SH. . 1990 Mol. Cell Biol. 10: 5772–5781.
Dobner T, Horikoshi N, Rubenwolf S and Shenk T. . 1996 Science 272: 1470–1473.
Fallaux FJ, Kranenburg O, Cramer SJ, Houweling A, Van Ormondt H, Hoeben RC and Van Der Eb AJ. . 1996 Hum. Gene Ther. 7: 215–222.
Furuwatari C, Yagi A, Yamagami O, Ishikawa M, Hidaka E, Ueno I, Furihata K, Ogiso Y and Katsuyama T. . 1998 Am. J. Clin. Pathol. 110: 368–373.
Goodrum FD and Ornelles DA . 1997 J. Virol. 71: 548–561.
Hall AR, Dix BR, O'Carroll SJ and Braithwaite AW. . 1998 Nat. Med. 4: 1068–1072.
Heise C, Sampson-Johannes A, Williams A, McCormick F, Von Hoff DD and Kirn DH. . 1997 Nat. Med. 3: 639–645.
Hollstein M, Sidransky D, Vogelstein B and Harris CC. . 1991 Science 253: 49–53.
Jost CA, Marin MC and Kaelin Jr WG. . 1997 Nature 389: 191–194.
Kaghad M, Bonnet H, Yang A, Creancier L, Biscan JC, Valent A, Minty A, Chalon P, Lelias JM, Dumont X, Ferrara P, McKeon F and Caput D. . 1997 Cell 90: 809–819.
Kolls J, Peppel K, Silva M and Beutler B. . 1994 Proc. Natl. Acad. Sci. USA 91: 215–219.
Lehman TA, Modali R, Boukamp P, Stanek J, Bennett WP, Welsh JA, Metcalf RA, Stampfer MR, Fusenig N, Rogan EM and Harris CC. . 1993 Carcinogenesis 14: 833–839.
Nigro JM, Baker SJ, Preisinger AC, Jessup JM, Hostetter R, Cleary K, Bigner SH, Davidson N, Baylin S, Devilee P, Glover T, Collins FS, Weston A, Modali R, Harris CC and Vogelstein B. . 1989a Nature 342: 705–708.
Ornelles DA and Shenk T. . 1991 J. Virol. 65: 424–429.
Osada M, Ohba M, Kawahara C, Ishioka C, Kanamaru R, Katoh I, Ikawa Y, Nimura Y, Nakagawara A, Obinata M and Ikawa S. . 1998 Nat. Med. 4: 839–843.
Peltenburg LT and Schrier PI. . 1994 Immunogenetics 40: 54–61.
Puisieux A, Galvin K, Troalen F, Bressac B, Marcais C, Galun E, Ponchel F, Yakicier C, Ji J and Ozturk M. . 1993 Faseb J. 7: 1407–1413.
Querido E, Marcellus RC, Lai A, Charbonneau R, Teodoro JG, Ketner G and Branton PE. . 1997 J. Virol. 71: 3788–3798.
Scheffner M, Munger K, Byrne JC and Howley PM. . 1991 Proc. Natl. Acad. Sci. USA 88: 5523–5527.
Schmale H and Bamberger C. . 1997 Oncogene 15: 1363–1367.
Steegenga WT, Riteco N, Jochemsen AG, Fallaux FJ and Bos JL. . 1998 Oncogene 16: 349–357.
Steegenga WT, van Laar T, Riteco N, Mandarino A, Shvarts A, van der Eb AJ and Jochemsen AG. . 1996 Mol. Cell Biol. 16: 2101–2109.
Steegenga WT, Van Laar T, Shvarts A, Terleth C, Van der Eb AJ and Jochemsen AG. . 1995 Virology 212: 543–554.
Steenbergen RD, Walboomers JM, Meijer CJ, van der Raaij-Helmer EM, Parker JN, Chow LT, Broker TR and Snijders PJ. . 1996 Oncogene 13: 1249–1257.
Stratford-Perricaudet LD, Makeh I, Perricaudet M and Briand P. . 1992 J. Clin. Invest. 90: 626–630.
Van der Eb AJ and Graham FL. . 1980 Methods Enzymol. 65: 826–839.
Weissman BE, Saxon PJ, Pasquale SR, Jones GR, Geiser AG and Stanbridge EJ. . 1987 Science 236: 175–180.
Wickham TJ, Mathias P, Cheresh DA and Nemerow GR. . 1993 Cell 73: 309–319.
Yew PR and Berk AJ. . 1992 Nature 357: 82–85.
Yew PR, Kao CC and Berk AJ. . 1990 Virology 179: 795–805.
Acknowledgements
We thank Mrs E van der Raaij-Helmer and Professor Dr R Willemse for the generous gift of the primary keratinocytes and Dr R Steenbergen and Professor Dr J Walbomers for the FK18B HPV-transformed keratinocytes. We are grateful to Mrs A-M Van den Hoeven-Kaiser and Dr Ph. de Groot for the primary endothelial cells. Dr AG Jochemsen was thanked for the G401-mt stable transfectant and for carefully reading the manuscript. We would like to thank Dr Kolls for the CMVLacZ virus. Furthermore, we are grateful to Drs I Ganly, A Balmain and F McCormick for helpful discussion throughout the project and to Dr F Fallaux for helpful discussion and technical assistance. This project was supported by a grant from the Dutch Cancer Society.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Steegenga, W., Riteco, N. & Bos, J. Infectivity and expression of the early adenovirus proteins are important regulators of wild-type and ΔE1B adenovirus replication in human cells. Oncogene 18, 5032–5043 (1999). https://doi.org/10.1038/sj.onc.1202886
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202886
Keywords
This article is cited by
-
HSF1 overexpression enhances oncolytic effect of replicative adenovirus
Journal of Translational Medicine (2010)
-
Selective replication of E1B55K-deleted adenoviruses depends on enhanced E1A expression in cancer cells
Cancer Gene Therapy (2006)
-
Inactivating intracellular antiviral responses during adenovirus infection
Oncogene (2005)
-
An armed oncolytic adenovirus system, ZD55-gene, demonstrating potent antitumoral efficacy
Cell Research (2003)
-
Loss of p14ARF in tumor cells facilitates replication of the adenovirus mutant dl1520 (ONYX-015)
Nature Medicine (2000)