Abstract
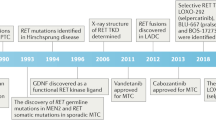
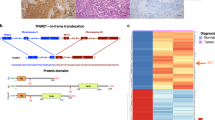
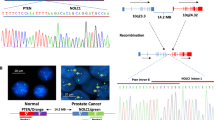
Children exposed to radioactive iodine as a consequence of the Chernobyl reactor accident have an increased risk of papillary thyroid carcinomas (PTC). The predominant molecular lesions in these tumors are rearrangements of the RET receptor tyrosine kinase (tk). Here we report on two novel types of RET rearrangement, PTC6 and 7, and describe the fusion products and the ret fused gene (rfg) proteins. Like the other rfg proteins identified so far they are ubiquitously expressed, not membrane-bound and contain coiled coil domains required for constitutive activation of the ret tk domain. In the PTC6 rearrangement the ret tk domain is fused to the aminoterminal part of the human transcription intermediary factor htif 1. In the PTC7 rearrangement the ret tk domain is fused to a novel protein that is strongly related to htif1. Like htif1 it contains a RBCC motif (ring finger, B boxes, coiled coil domain) located in the aminoterminal part and a phd finger and a bromodomain in the carboxyterminal part. Htif1 and related proteins are transcription coactivators for nuclear receptors, thus participating in controlling cellular development, differentiation and homeostasis. This is the first report on their involvement in human thyroid carcinogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- GDNF:
-
glial cell derived neurotrophic factor
- Htif:
-
human transcription intermediary factor
- PTC:
-
papillary thyroid carcinoma
- rfg:
-
ret fused gene
- tk:
-
tyrosine kinase
References
Alen P, et al. 1999 Mol. Endocrinol. 13: 117–128.
Altshul SF, et al. 1990 J. Mol. Biol. 215: 403–410.
Avantaggiato V, et al. 1994 Cell Growth Diff. 5: 305–311.
Baverstock K, et al. 1992 Nature 359: 21–22.
Bongarzone I, et al. 1993 Mol. Cell. Biol. 13: 358–366.
Bongarzone I, et al. 1994 Cancer Res. 54: 2979–2985.
De Vita G, et al. 1998 Cell Growth Differ. 9: 97–103.
Durbec P, et al. 1996 Nature 381: 789–793.
Feng DF and Doolittle RF. . 1987 J. Mol. Evol. 25: 351–360.
Fugazzola L, et al. 1995 Cancer Res. 55: 5617–5620.
Fugazzola L, et al. 1996 Oncogene 13: 1093–1097.
Grieco M, et al. 1990 Cell 60: 557–663.
Jeanmougin F, et al. 1997 TIBS 22: 151–153.
Jhiang SM, Smanik PA and Mazzaferri EL. . 1994 Cancer Lett. 78: 69–76.
Jing S, et al. 1996 Cell 85: 1113–1124.
Kazakov VS, Demidchik EP and Astakhova LN. . 1992 Nature 359: 21–22.
Klugbauer S, Lengfelder E, Demidchik EP and Rabes HM. . 1995 Oncogene 11: 2459–2467.
Klugbauer S, Lengfelder E, Demidchik EP and Rabes HM. . 1996 Oncogene 13: 1099–1102.
Klugbauer S, Demidchik EP, Lengfelder E and Rabes HM. . 1998a Oncogene 16: 671–675.
Klugbauer S, Demidchik EP, Lengfelder E and Rabes HM. . 1998b Cancer Res. 58: 198–203.
Kunieda T, Matsui M, Nomura N and Ishizaki R. . 1991 Gene 107: 323–328.
Le Douarin B, et al. 1995 EMBO J. 14: 2020–2033.
Le Douarin B, et al. 1996 EMBO J. 15: 6701–6715.
Lehrach H, et al. 1990 In: Genome Analysis Volume 1: Genetic and Physical Mapping. Davies KE and Tilghman SM. (eds).. Cold Spring Harbor Laboratory Press: Cold Spring Harbor pp. 39–81.
Nikiforov Y and Gnepp DR. . 1994 Cancer 74: 748–766.
Nikiforov YE, et al. 1997 Cancer Res. 57: 1690–1694.
Rabes HM and Klugbauer S. . 1997 Verh. Dtsch. Ges. Path. 81: 139–144.
Rabes HM and Klugbauer S. . 1998 Rec. Res. Cancer Res. 154: 149–165.
Sagartz JE, Jhiang SM, Tong Q and Capen CC. . 1997 Lab. Invest. 76: 307–318.
Takahashi M, Inaguma Y, Hiai H and Hirose F. . 1988 Mol. Cell. Biol. 8: 1853–1856.
Thénot S, Henriquet C, Rochefort H and Cavailles V. . 1997 J. Biol. Chem. 272: 12062–12068.
Tong Q, Xing S and Jhiang SJ. . 1997 J. Biol. Chem. 272: 9043–9047.
Trupp M, et al. 1996 Nature 381: 785–789.
Xing S, Furminger TL, Tong Q and Jhiang SM. . 1998 J. Biol. Chem. 273: 4909–4914.
Yeh S and Chang C. . 1996 Proc. Natl. Acad. Sci. USA 93: 5517–5521.
Acknowledgements
We thank Professor Demidchik, Minsk, Belarus, and Professor Lengfelder, Munich, Germany, for generously providing tumor samples and patients data. We are grateful to Michael Ruiter, Dr Sibylle Liebmann, Rita Koch and Sigrid Madsen for excellent technical assistance and to the Otto Hug-Strahleninstitut and Christine Frenzel for support of this work. This work was supported by grants (to HMR) from Dr Mildred Scheel-Stiftung für Krebsforschung, Bonn, Germany and from Wilhelm Sander-Stiftung, Neuburg an der Donau, Germany.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Klugbauer, S., Rabes, H. The transcription coactivator HTIF1 and a related protein are fused to the RET receptor tyrosine kinase in childhood papillary thyroid carcinomas. Oncogene 18, 4388–4393 (1999). https://doi.org/10.1038/sj.onc.1202824
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202824
Keywords
This article is cited by
-
Genetic alterations landscape in paediatric thyroid tumours and/or differentiated thyroid cancer: Systematic review
Reviews in Endocrine and Metabolic Disorders (2024)
-
Kinase gene fusions: roles and therapeutic value in progressive and refractory papillary thyroid cancer
Journal of Cancer Research and Clinical Oncology (2021)
-
Thyroid cancers of follicular origin in a genomic light: in-depth overview of common and unique molecular marker candidates
Molecular Cancer (2018)
-
A comprehensive overview of the role of the RET proto-oncogene in thyroid carcinoma
Nature Reviews Endocrinology (2016)
-
TRIM24 is upregulated in human gastric cancer and promotes gastric cancer cell growth and chemoresistance
Virchows Archiv (2015)