Abstract
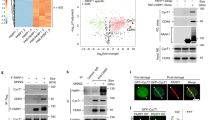
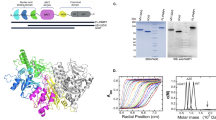
It has been suggested that DNA-dependent protein kinase (DNA-PK) is a central component of DNA double-strand-break repair. The mechanism of DNA-PK action, however, has not been fully understood. Poly(ADP-ribose) polymerase (PARP) is another nuclear enzyme which has high affinity to DNA ends. In this study, we analysed the interaction between these two enzymes. First, DNA-PK was found to suppress the PARP activity and alters the pattern of poly(ADP-ribosyl)ation. Although DNA-PK phosphorylates PARP in a DNA-dependent manner, this modification is unlikely to be responsible for the suppression of PARP activity, since this suppression occurs even in the absence of ATP. Conversely, PARP was found to ADP-ribosylate DNA-PK in vitro. However, the auto-phosphorylation activity of DNA-PK was not influenced by this modification. In a competitive electrophoretic mobility shift assay, Ku 70/80 complex, the DNA binding component of DNA-PK, was found to have higher affinity to a short fragment of DNA than does PARP. Furthermore, co-immunoprecipitation analysis suggested direct or close association between Ku and PARP. Thus, DNA-PK suppresses PARP activity, probably through direct binding and/or sequestration of DNA-ends which serve as an important stimulator for both enzymes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Agarwal ML, Agarwal A, Taylor WR, Wang Z-Q, Wagner EF and Stark GR. . 1997 Oncogene 15: 1035–1041.
Ariumi Y, Copeland TD, Nosaka T and Hatanaka M. . 1997 Leukemia 11: Supplement 3 29–31.
Ariumi Y, Shimotohno K, Noda M and Hatanaka M. . 1998 FEBS Lett. 423: 25–30.
Baskaran R, Wood LD, Whitaker LL, Canman CE, Morgan SE, Xu Y, Barlow C, Baltimore D, Wynshaw-Boris A, Kastan MB and Wang JYJ. . 1997 Nature 387: 516–519.
Blunt T, Finnie NJ, Taccioli GE, Smith GCM, Demengeot J, Gottlieb TM, Mizuta R, Varghese AJ, Alt FW, Jeggo PA and Jackson SP. . 1995 Cell 80: 813–823.
Carter T, Vancurová I, Sun I, Lou W and Deleon S. . 1990 Mol. Cell. Biol. 10: 6460–6471.
Chan DW and Lees-Miller SP. . 1996 J. Biol. Chem. 271: 8936–8941.
Darby MK, Schmitt B, Jongstra-Bilen J and Vosberg H-P. . 1985 EMBO J. 4: 2129–2134.
de Murcia G and Ménissier de Murcia J. . 1994 Trends Biochem. Sci. 19: 172–176.
Dignam JD, Lebowitz RM and Roeder RG. . 1983 Nucleic. Acids Res. 11: 1475–1489.
Durkacz BW, Omidiji O, Gray DA and Shall S. . 1980 Nature 283: 593–596.
Ferro AM and Olivera BM. . 1982 J. Biol. Chem. 257: 7808–7813.
Gottlieb TM and Jackson SP. . 1993 Cell 72: 131–142.
Ikejima M, Noguchi S, Yamashita R, Susuki H, Sugimura T and Miwa M. . 1989 Biochem. Biophys. Res. Commun. 163: 739–745.
Ikejima M, Noguchi S, Yamashita R, Ogura T, Sugimura T, Gill DM and Miwa M. . 1990 J. Biol. Chem. 265: 21907–21913.
Jackson SP, MacDonald JJ, Lees-Miller S and Tijian R. . 1990 Cell 63: 155–165.
Jackson SP and Jeggo PA. . 1995 Trends Biochem. Sci. 20: 412–415.
Kamata T, Subleski M, Hara Y, Yuhki N, Kung H-F, Copeland NG, Jenkins NA and Copeland TD. . 1998 Mol. Brain Res. 54: 219–236.
Kawaguchi H, Asai A, Ohtsuka Y, Watanabe H, Wada H and Handa H. . 1989 Nucleic. Acids Res. 17: 6229–6240.
Kharbanda S, Pandey P, Jin S, Inoue S, Bharti A, Yuan Z-M, Weichselbaum R, Weaver D and Kufe D. . 1997 Nature 386: 732–735.
Kirchgessner CU, Patil CK, Evans JW, Cuomo CA, Fried LM, Carter T, Oettinger MA and Brown JM. . 1995 Science 267: 1178–1183.
Lees-Miller SP and Anderson CW. . 1989 J. Biol. Chem. 264: 17275–17280.
Lees-Miller SP, Chen Y-R and Anderson CW. . 1990 Mol. Cell. Biol. 10: 6472–6481.
Lees-Miller SP, Sakaguchi K, Ullrich SP, Appella E and Anderson CW. . 1992 Mol. Cell. Biol. 12: 5041–5049.
Lees-Miller SP, Godbout R, Chan DW, Weinfeld M, Day III RS, Barron GM and Allalunis-Turner J. . 1995 Science 267: 1183–1185.
Lindahl T, Satoh MS, Poirier GG and Klungland A. . 1995 Trends Biochem. Sci. 20: 405–411.
Ménissier de Murcia J, Niedergang C, Trucco C, Ricoul M, Dutrillaux B, Mark M, Oliver FJ, Masson M, Dierich A, LeMeur M, Walztinger C, Chambon P and de Murcia G. . 1997 Proc. Natl. Acad. Sci. USA 94: 7303–7307.
Mimori T, Hardin JA and Steitz JA. . 1986 J. Biol. Chem. 261: 2274–2278.
Mimori T, Ohosone Y, Hama N, Suwa A, Akizuki M, Homma M, Griffith AJ and Hardin JA. . 1990 Proc. Natl. Acad. Sci. USA 87: 1777–1781.
Molinete M, Vermeulen W, Bürkle A, Ménissier de Murcia J, Küpper JH, Hoeijmarkers JH and de Murcia G. . 1993 EMBO J. 12: 2109–2117.
Morrison C, Smith GCM, Stingl L, Jackson SP, Wagner EF and Wang Z-Q. . 1997 Nature Genetics 17: 479–482.
Nosaka T, Ariumi Y, Sakurai M, Takeuchi R and Hatanaka M. . 1993 Nucl. Acids Res. 21: 5124–5129.
Ohtsuki M, Sekimizu K, Agemori M, Shizuta Y and Natori S. . 1984 FEBS Lett. 168: 275–277.
Reeves WH and Sthoeger ZM. . 1989 J. Biol. Chem. 264: 5047–5052.
Rice WG, Schaeffer CA, Villinger F, South TL, Summers MF, Henderson LE, Bess Jr JW, Arthur LO, McDougal JS, Orloff SL, Mendeleyev J and Kun E. . 1993a Nature 361: 473–475.
Rice WG, Schaeffer CA, Graham L, Bu M, McDougal JS, Orloff SL, Villinger F, Young M, Oroszlan S, Fesen MR, Pommier Y, Mendeleyev J and Kun E. . 1993b Proc. Natl. Acad. Sci. USA 90: 9721–9724.
Ruscetti T, Lehnert BE, Halbrook J, Trong HL, Hoekstra MF, Chen DJ and Peterson SR. . 1998 J. Biol. Chem. 273: 14461–14467.
Sato MS and Lindahl T. . 1992 Nature 356: 356–358.
Shafman T, Khanna KK, Kedar P, Spring K, Kozlov S, Yen T, Hobson K, Gatei M, Zhang N, Watters D, Egerton M, Shiloh Y, Kharbanda S, Kufe D and Lavin MF. . 1997 Nature 387: 520–523.
Shieh S-Y, Ikeda M, Taya Y and Prives C. . 1997 Cell 91: 325–334.
Taniguchi T, Morisawa K, Ogawa M, Yamamoto H and Fujimoto S. . 1988 Biochem. Biophys. Res. Commun. 154: 1034–1040.
Ueda K, Omachi A, Kawaichi M and Hayaishi O. . 1975 Proc. Natl. Acad. Sci. USA 72: 205–209.
Ueda K and Hayaishi O. . 1985 Ann. Rev. Biochem. 54: 73–100.
Vaziri H, West MD, Allsopp RC, Davison TS, Wu Y-S, Arrowsmith CH, Poirier GG and Benchimol S. . 1997 EMBO J. 16: 6018–6033.
Wang Z-Q, Auer B, Stingl L, Berghammer H, Haidacher D, Schweiger M and Wagner EF. . 1995 Genes Dev. 9: 509–520.
Wang Z-Q, Stingl L, Morrison C, Jantsch M, Los M, Schulze-Osthoff K and Wagner EF. . 1997 Genes Dev. 11: 2347–2358.
Yamagoe S, Kohda T and Oishi M. . 1991 Mol. Cell. Biol. 11: 3522–3527.
Yuan Z-M, Huang Y, Whang Y, Sawyers C, Weichselbaum R, Kharbanda S and Kufe D. . 1996 Nature 382: 272–274.
Zhang J, Dawson VL, Dawson TM and Snyder SH. . 1994 Science 263: 687–689.
Acknowledgements
We thank Dr J Turner for M059K and M059J cells, Drs T Wada and H Handa for the supply of affinity latex particles, Dr T Taniguchi for anti-PARP antibody, Dr T Hamakubo for PARP protein as well as helpful discussion. We also thank Drs M Hijikata, M Takata, C Morrison and S Takeda for advice and discussion, and Ms K Yamagami, K Yamazaki and M Shirai for technical assistance. This work was supported in part by Grants-in-Aid for Scientific Research from Ministry of Education, Science, Sports and Culture of Japan (08280102). This work was also sponsored in part by the National Cancer Institute, DHHS, under contract with ABL.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ariumi, Y., Masutani, M., Copeland, T. et al. Suppression of the poly(ADP-ribose) polymerase activity by DNA-dependent protein kinase in vitro. Oncogene 18, 4616–4625 (1999). https://doi.org/10.1038/sj.onc.1202823
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202823
Keywords
This article is cited by
-
DNA-PKcs post-translational modifications and associated diseases
Genome Instability & Disease (2022)
-
The regulatory landscape of the human HPF1- and ARH3-dependent ADP-ribosylome
Nature Communications (2021)
-
NAD+ metabolism: pathophysiologic mechanisms and therapeutic potential
Signal Transduction and Targeted Therapy (2020)
-
PARP1 and DNA-PKcs synergize to suppress p53 mutation and telomere fusions during T-lineage lymphomagenesis
Oncogene (2013)