Abstract
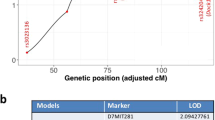
Our previous allelic loss analysis of γ-ray induced thymic lymphomas in F1 hybrid and backcross mice between BALB/c and MSM strains mapped the Tlsr4 region exhibiting a high frequency of allelic loss (62%) to a 2.9 cM interval between the markers D12Mit53 and D12Mit279 on mouse chromosome 12. To narrow further the interval harboring a putative tumor suppressor gene, a high-density scan has been carried out for informative 361 thymic lymphomas. Construction of a physical map of Tlsr4 with 3 YAC and 15 BAC clones and isolation of YAC- and BAC-derived polymorphic probes lead to fine allelic loss mapping. Three successive polymorphic sites within one BAC exhibit the retention of both alleles in seven, one and four lymphomas, suggesting that a common region of allelic loss for Tlsr4 exists within the BAC region. Pulsed-field gel electrophoresis of NotI digests of this and other clones determines that the commonly lost region is a 35 kb interval with a NotI site. NotI sites are frequently associated with coding regions, and our preliminary sequencing has identified ESTs in the region. Thus, the present study facilitates the identification of genes in the Tlsr4 region that would lead to isolation of a novel tumor suppressor gene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- YAC:
-
yeast artificial chromosome
- BAC:
-
bacteria artificial chromosome
References
Antequera F and Bird A. . 1993 Proc. Natl. Acad. Sci. USA 90: 11995–11999.
Bandera CA, Takahashi H, Behbakht K, Liu PC, LiVolsi VA, Benjamin I, Morgan MA, King SA, Rubin SC and Boyd J. . 1997 Cancer Res. 57: 513–515.
Bird A. . 1992 Cell 70: 5-8.
Bonhomme F and Guenet J-L. . 1989 Genetic Variants and Strains of the Laboratory Mouse. Lyon MF and Searle AG (eds).. Oxford University Press: Cambridge p. 658.
Brown MA and Solomon E. . 1997 Trends Genet. 13: 202–206.
Chang WY-H, Cairns P, Schoenberg MP, Polascik TJ and Sidransky D. . 1995 Cancer Res. 55: 3246–3249.
Dietrich WF, Radany EH, Smith JS, Bishop JM, Hanahan DH and Lander ES. . 1994 Proc. Natl. Acad. Sci. USA 91: 9451–9455.
Dietrich WF, Miller J, Steen R, Merchant MA, Damron-Boles D, Husain Z, Dredge R, Daly MJ, Ingalls KA, O'Connor TJ, Evans CA, DeAngelis MM, Levinson DM, Kruglyak L, Goodman N, Copeland NG, Jenkins NA, Hawkins TL, Stein L, Page DC and Lander ES. . 1996 Nature 380: 149–152.
Hayashi T, Ohtsuka H, Kuwabara K, Mafune Y, Miyashita N, Moriwaki K, Takahashi Y and Kominami R. . 1993 Genomics 17: 490–492.
Hegi ME, Devereux TR, Dietrich WF, Cochran CJ, Lander ES, Foley JF, Maronpot RR, Anderson MW and Wiseman RW. . 1994 Cancer Res. 54: 6257–6264.
Jacks T. . 1996 Annu. Rev. Genet. 30: 603–633.
Kemp CJ, Fee F and Balmain A. . 1993 Cancer Res. 53: 6022–6027.
Knudson AG. . 1985 Cancer Res. 45: 1437–1443.
Matsumoto Y, Kosugi S, Shinbo T, Chou D, Ohashi M, Wakabayashi Y, Sakai K, Okumoto M, Mori N, Aizawa S, Niwa O and Kominami R. . 1998 Oncogene 16: 2747–2754.
Okamoto M, Ohtsu H, Kominami R and Yonekawa H. . 1995 Carconigenesis 16: 2659–2666.
Radany EH, Hong K, Kesharvarzi S, Lander ES and Bishop JM. . 1997 Proc. Natl. Acad. Sci. USA 94: 8664–8669.
Riley J, Butler R, Ogilvie D, Finniear R, Jenner D, Powell S, Anand R, Smith JC and Markham AF. . 1990 Nucl. Acids Res. 18: 2887–2890.
Santos J, de Castro IP, Herranz M, Pellicer A and Fernandez-Piqueras J. . 1996 Oncogene 12: 669–676.
Santos J, Herranz M, de Castro IP, Pellicer A and Fernandez-Piqueras J. . 1998 Oncogene 17: 925–929.
Suzuki T, Yokota J, Mugishima H, Okabe I, Ookuni M, Sugimura T and Terada M. . 1989 Cancer Res. 49: 1095–1098.
Vogelstein B and Kinzler KW. . 1993 Trends Genet. 9: 138–141.
Weinberg RA. . 1991 Science 254: 1138–1146.
Young J, Leggett B, Ward M, Thomas L, Buttenshaw R, Searle J and Chenevix-Trench G. . 1993 Oncogene 8: 671–675.
Zhuang S-M, Eklund LK, Cochran C, Rao GN, Wiseman RW and Soderkvist P. . 1996 Cancer Res. 56: 3338–3343.
Acknowledgements
This work was supported by Health Science Research Grants for Human Genome and Gene Therapy from Ministry of Health and Welfare of Japan.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shinbo, T., Matsuki, A., Matsumoto, Y. et al. Allelic loss mapping and physical delineation of a region harboring a putative thymic lymphoma suppressor gene on mouse chromosome 12. Oncogene 18, 4131–4136 (1999). https://doi.org/10.1038/sj.onc.1202767
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202767
Keywords
This article is cited by
-
Multi-step lymphomagenesis deduced from DNA changes in thymic lymphomas and atrophic thymuses at various times after γ-irradiation
Oncogene (2007)
-
Lack of Bcl11b tumor suppressor results in vulnerability to DNA replication stress and damages
Oncogene (2007)
-
Bcl11b is required for differentiation and survival of αβ T lymphocytes
Nature Immunology (2003)
-
Bcl11: sibling rivalry in lymphoid development
Nature Immunology (2003)
-
Genetic loci controlling susceptibility to γ-ray-induced thymic lymphoma
Oncogene (2001)