Abstract
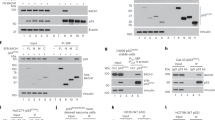
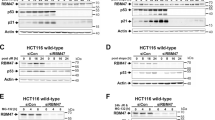
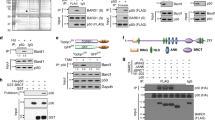
Inherited mutations in the breast and ovarian cancer susceptibility gene BRCA1 are associated with high risk for developing breast and ovarian cancers. Several studies link BRCA1 to transcriptional regulation, DNA repair, apoptosis and growth/tumor suppression. BRCA1 associates with p53 and stimulates transcription in both p53 dependent and p53-independent manners. BRCA1 splice variants BRCA1a (p110) and BRCA1b (p100) associates with CBP/p300 co-activators. Here we show that BRCA1a and BRCA1b proteins stimulate p53-dependent transcription from the p21WAF1/CIP1 promoter. In addition, the C-terminal second BRCA1 (BRCT) domain is sufficient for p53 mediated transactivation of the p21 promoter. Previous studies emphasized the importance of the BRCT domain, which shows homology with p53 binding protein (53BP1), in transcriptional activation, growth inhibition and tumor suppression. Our findings demonstrate an additional function for this domain in protein – protein interaction and co-activation of p53. We also found that BRCA1a and BRCA1b proteins interact with p53 in vitro and in vivo. The p53 interaction domain of BRCA1a/1b maps, in vitro, to the second BRCT domain (aa 1760 – 1863). The BRCT domain binds to the central domain of p53 which is required for sequence specific DNA binding. These results demonstrate for the first time the presence of a second p53 interaction domain in BRCA1 proteins and suggests that BRCA1a and BRCA1b proteins, like BRCA1, function as p53 co-activators. This BRCT domain also binds in vitro to CBP. These results suggest that one of the mechanisms by which BRCA1 proteins function is through recruitment of CBP/p300 associated HAT/FAT activity for acetylation of p53 to specific promoters resulting in transcriptional activation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bork P, Hofmann K, Bucher P, Neuwald AF, Altschul SF and Koonin EV. . 1997 FASEB J. 11: 68–76.
Brugarolas J and Jacks T. . 1997 Nature Med. 7: 721–722.
Callebaut I and Mornon J-P. . 1997 FEBS Lett. 400: 25–30.
Chapman MS and Verma IM. . 1996 Nature 382: 678–679.
Couch FJ and Weber BL. . 1996 Hum. Mutat. 8: 8–18.
Cui JQ, Wang H, Reddy ESP and Rao VN. . 1998a Oncol. Reports 5: 585–589.
Cui JQ, Shao N, Chai YL, Wang H, Reddy ESP and Rao VN. . 1998b Oncol. Reports 5: 591–595.
Easton DF, Ford D, Bishop DT and Breast cancer linkage Consortium. . 1995 Am. J. Hum. Genet. 56: 265–271.
El-Deiry WS, Tokino T, Waldman T, Oliner JD, Velculescu VE, Burrell M, Hill DE, Healy E, Rees JL, Hamilton SR, Kinzler KW and Vogelstein B. . 1995 Cancer Res. 55: 2910–2919.
Ford D, Easton DF and Peto J. . 1995 Am. J. Hum. Genet. 57: 1457–1462.
Hollstein M, Sidransky D, Vogelstein B and Harris CC. . 1991 Science 53: 49–53.
Holt JT, Thompson ME, Szabo C, Robinson-Benion C, Arteaga CL, King MC and Jensen RA. . 1996 Nature Genet. 12: 298–302.
Humphrey JS, Salin A, Erodos MR, Collins FS, Brody LC and Klausner RD. . 1997 Proc. Natl. Acad. Sci. USA 94: 5820–5825.
Iwabuchi K, Bartel PL, Li B, Marraccino R and Fields S. . 1994 Proc. Natl. Acad. Sci. USA 91: 6098–6102.
Ko LJ and Prives C. . 1996 Genes Dev. 10: 1054–1072.
Koonin EV, Altschul SF and Bork P. . 1996 Nature Genet. 13: 266–268.
Lane TF, Deng C, Elson A, Lyu MS, Kozak CA and Leder P. (1995). . Genes and Dev. 7: 2712–2722.
Marquis ST, Rajan JV, Wynshaw-Borsi A, Xu J, Yin G-Y, Abel KJ, Weber BL and Chodosh LA. . 1995 Nature Genet. 11: 17–26.
Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, Tavtigian S, Liu Q, Cochran C, Bennett LM, Ding W, Bell R, Rosenthal J, Hussey C, Tran T, McClure M, Frye C, Hattier T, Phelps R, Haugen-Strano A, Katcher H, Yakumo K, Gholami Z, Shaffer D, Stone S, Bayer S, Wray C, Bogden R, Dayanath P, Ward J, Tonin P, Narod S, Bristow PK, Norris FH, Helvering L, Morrison P, Rosteck P, Lai M, Barrett JC, Lewis C, Neuhausen S, Cannon-Albright L, Goldgar D, Wiseman R, Kamb A, Skolnick MH. . 1994 Science 266: 66–71.
Monteiro AN, August A and Hanafusa H. . 1996 Proc. Natl. Acad. Sci. USA 93: 13595–13599.
Nakajima T, Uchida C, Anderson SF, Pavin JD and Montiminy M. . 1997 Genes Dev. 11: 738–747.
Ouchi T, Monteiro ANA, August A and Aaronson SA. . 1998 Proc. Natl. Acad. Sci. USA 95: 2302–2306.
Rao VN, Shao N, Ahmad M and Reddy ESP. . 1996 Oncogene 12: 523–528.
Scully R, Anderson SF, Chao DM, Wei W, Ye RA, Livingston DM and Parvin JD. . 1997a Proc. Natl. Acad. Sci. USA 94: 5605–5610.
Scully R, Chen J, Plug A, Xiao Y, Weaver D, Feunteun J, Ashley T and Livingston DM. . 1997b Cell 88: 265–275.
Shao N, Chai YL, Reddy ESP and Rao VN. . 1996 Oncogene 13: 1–7.
Somasundaram K, Zhang H, Zeng YX, Houvras H, Peng Y, Zhang H, Wu GS, Licht JD, Weber BL and El-Deiry WS. . 1997 Nature 389,: 187–190.
Stürzbecher HW, Brain R, Addison C, Rudge K, Remm M, Grimaldi M, Keenan E and Jenkins JR. . 1992 Oncogene 7: 1513–1523.
Szabo CI, Wagner LA, Francisco LV, Roach JC, Argonza R, King MC and Ostrander EA. . 1996 Human Mol. Genet. 5: 1289–1298.
Thompson ME, Jensen RA, Obermiller PS, Page DL and Holt JT. . 1995 Nature Genet. 9: 444–450.
Wang H, Shao N, Ding QM, Cui JQ, Reddy ESP and Rao VN. . 1997 Oncogene 15: 143–157.
Wu CJ, Wang ZW, Tsan JT, Spillman MA, Phung A, Xu XL, Yang MC, Hwang LY, Bowcock AM and Baer R. . 1996 Nature Genet. 14: 430–447.
Zhang H, Somasundaram K, Peng Y, Tian H, Zhang H, Bi D, Weber BL and El-Deiryh WS. . 1998 Oncogene 16: 1713–1721.
Acknowledgements
We thank B Vogelstein, T Shenk and Klas Wiman for their generous gift of p53 reporter and expression plasmids, T Kouzarides for CBP plasmids and J Gioanni for CAL51 cell line. We thank G Chipitsyna for excellent technical help, Dr RE Pyeritz for critical comments on the manuscript, support and encouragement and L Boyer for secretarial assistance. We thank the other members of Rao and Reddy labs for their help. This work was supported by NIH grants CA57322 and CA50507 and US Army Medical Research Grant DAMD17-4-J-4280 to VNR and NIH grant CA58642 to ESPR.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chai, Y., Cui, Jq., Shao, N. et al. The second BRCT domain of BRCA1 proteins interacts with p53 and stimulates transcription from the p21WAF1/CIP1 promoter. Oncogene 18, 263–268 (1999). https://doi.org/10.1038/sj.onc.1202323
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202323
Keywords
This article is cited by
-
RBBP8/CtIP suppresses P21 expression by interacting with CtBP and BRCA1 in gastric cancer
Oncogene (2020)
-
Distinct associations of the Saccharomyces cerevisiae Rad9 protein link Mac1-regulated transcription to DNA repair
Current Genetics (2020)
-
Impact of Etoposide on BRCA1 Expression in Various Breast Cancer Cell Lines
Drugs in R&D (2017)
-
Role of multifunctional transcription factor TFII-I and putative tumour suppressor DBC1 in cell cycle and DNA double strand damage repair
British Journal of Cancer (2013)
-
Stat6 cooperates with Sp1 in controlling breast cancer cell proliferation by modulating the expression of p21Cip1/WAF1 and p27Kip1
Cellular Oncology (2013)