Abstract
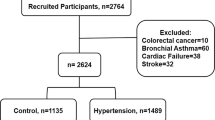
The objective of the present study was to analyze the impact of metabolic syndrome (MS) and its individual components on oxidative stress (OX) and on the activity of antioxidant enzymes of patients with essential hypertension. One hundred and eighty-seven hypertensives, 127 (61.9%) of them having criteria for MS according to the International Diabetes Federation criteria and 30 healthy normotensive subjects were included. OX status was assessed by measuring glutathione oxidized/glutathione reduced and reactive oxygen species-induced byproducts of lipid peroxidation, malondialdehide, and DNA damage, 8-oxo-dG genomic and mitochondrial. Antioxidant enzymatic activity of Cu/Zn extracellular-superoxide dismutase (SOD) and catalase (CAT) was measured in plasma and glutathione peroxidase 1 in hemolysad erythrocytes. In mononuclear cells, total-SOD activity, CAT and glutathione peroxidase 1, were assessed as well. The OX state in both blood and peripheral mononuclear cells observed in hypertensives were not enhanced by the addition of components of the so-called MS. Likewise, the reduction in the activity of antioxidant enzymes, both extracellular and cytoplasmic, was not affected by the presence of additional components of the MS. Neither the number of components nor the individual addition of each of them, low high-density lipoprotein, triglycerides, abdominal obesity or fasting glucose, further impact in the OX abnormalities observed in those with only hypertension in absence of other components. In conclusion, the present data indicates that contribution of MS components to the OX burden generated by high blood pressure is minimal.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kahn R, Buse J, Ferrannini E, Stern M, American Diabetes Association; European Association for the Study of Diabetes. The metabolic syndrome: time for a critical appraisal: joint statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diab Care 2005; 28: 2289–2304.
Romero JC, Reckelhoff JF . Role of angiotensin and oxidative stress in essential hypertension. Hypertension 1999; 34: 943–949.
Raij L . Nitric oxide in hypertension: relationship with renal injury and left ventricular hypertrophy. Hypertension 1998; 31: 189–193.
McIntyre M, Bohr DF, Dominiczak AF . Endothelial function in hypertension. The role of superoxide anion. Hypertension 1999; 34: 539–545.
Orie NN, Zidek W, Tepel M . Reactive oxygen species in essential hypertension and non-insulin-dependent diabetes mellitus. Am J Hypertens 1999; 12: 1169–1174.
Yasunari K, Maeda K, Nakamura M, Yoshikawa J . Oxidative stress in leukocytes is a possible link between blood pressure, blood glucose, and C-reactive protein. Hypertension 2002; 39: 777–780.
Redon J, Oliva MR, Tormos C, Giner V, Chaves FJ, Iradi A et al. Antioxidant activities and oxidative stress byproducts in human hypertension. Hypertension 2003; 41: 1096–1101.
Saez GT, Tormos C, Giner V, Chaves FJ, Lozano JV, Iradi A et al. Factors related to the impact of antihypertensive treatment in antioxidant activities and oxidative stress byproducts in human hypertension. Am J Hypertens 2004; 17: 809–816.
Blakenberg S, Rupprecht HJ, Bickel CH, Torzewki M, Hafner G, Tiret L et al. for the AtheroGene Investigators: Glutathione peroxidase 1 activity and cardiovascular events in patients with coronary artery disease. N Engl J Med 2003; 349: 1605–1613.
Joint National Committee. The sixth report of the joint national committee on prevention, detection, evaluation and treatment of high blood pressure. Arch Intern Med 1997; 157: 2413–2447.
International Diabetes Federation. The IDF Consensus worldwide definition of the metabolic syndrome (article online). (Available from) www.idf.org(Accessed 2 June, 2005).
Muntner P, He J, Chen J, Fonseca V, Whelton PK . Prevalence of non-traditional cardiovascular disease risk factors among persons with impaired fasting glucose, impaired glucose tolerance, diabetes, and the metabolic syndrome: analysis of the Third National Health and Nutrition Examination Survey (NHANES III). Ann Epidemiol 2004; 14: 686–695.
Brigelius R, Muckel C, Akerboom TPM, Sies H . Identification and quantitation of glutathione in hepatic protein mixed disulfides and its relation to glutathione disulfides. Biochem Pharmacol 1983; 32: 2529–2534.
Navarro J, Obrador E, Pellicer JA, Asensi M, Viña J, Estrela JM . Blood glutathione as an index of radiation-induced oxidative stress in mice and humans. Free Radic Biol Med 1997; 22: 1203–1209.
Wong SHY, Knight JA, Hopfer SM, Zaharia O, Leach Jr CN, Sunderman Jr FW . Lipoperoxides in plasma as measured by liquid-chromatographic separation of malondialdehyde-thiobarbituric acid adduct. Clin Chem 1987; 33: 214–220.
Clairbone A . Catalase activity. In: Green-Wald RA (ed). Handbook of Methods for Oxygen Radical Research. CRC Press Inc.: Boca Raton, FL, 1986, pp 283–284.
McCord JM, Fridowich I . The reduction of cytochrome c by milk xanthine oxidase. J Biol Chem 1968; 243: 5753–5760.
Muñiz P, Valls V, Perez-Broseta C, Iradi A, Climent JV, Oliva MR et al. The role of 8-hydroxy-2′-deoxyguanosine in rifamycin-induced DNA damage. Free Radic Biol Med 1995; 18: 747–755.
Bland JM, Altmann DG . Statistical method for assessing agreement between two methods of clinical measurements. Lancet 1986; 1: 307–310.
Moore K, Roberts II LJ . Measurement of lipid peroxidation. Free Radic Res 1998; 28: 659–671.
Simic DV, Mimic-Oka J, Pljesa-Ercegovac M, Savic-Radojevic A, Opacic M, Matic D et al. Byproducts of oxidative protein damage and antioxidant enzyme activities in plasma of patients with different degrees of essential hypertension. J Hum Hypertens 2006; 20: 149–155.
Wu L, Jourlink BH . Increased methylglyoxal and oxidative stress in hypertensive rat vascular smooth muscle cells. Hypertension 2002; 39: 809–814.
Lerman LO, Nath KA, Rodriguez-Porcel M, Krier JD, Schwartz RS, Napoli C et al. Increased oxidative stress in experimental renovascular hypertension. Hypertension 2001; 37: 541–546.
Trolliet MR, Rudd MA, Loscalzo J . Oxidative stress and renal dysfunction in salt-sensitive hypertension. Kidney Blood Press Res 2001; 24: 116–123.
Dobrian AD, Davies MJ, Schriver SD, Lauterio TJ, Prewitt RL . Oxidative stress in a rat model of obesity-induced hypertension. Hypertension 2001; 37: 554–560.
Nakazono K, Watanabe N, Matsuno K, Sasaki J, Sato T, Inoue M . Does superoxide underlie the pathogenesis of hypertension? Proc Natl Acad Sci USA 1991; 88: 10045–10048.
Lassègue B, Griendling KK . Reactive oxygen species in hypertension: an update. Am J Hypertens 2004; 17: 852–860.
Giner V, Tormos C, Chaves FJ, Sáez G, Redón J . Microalbuminuria and oxidative stress in essential hypertension. J Intern Med 2004; 255: 588–594.
Salo DC, Pacifici RE, Lin SW, Giulivi C, Davies KJ . Superoxide dismutase undergoes proteolysis and fragmentation following oxidative modification and inactivation. J Biol Chem 1990; 265: 11919–11927.
Davies KJ . Protein damage and degradation by oxygen radicals. I. General aspects. J Biol Chem 1987; 262: 9895–9901.
Pigeolet E, Remacle J . Susceptibility of glutathione peroxidase to proteolysis after oxidative alteration by peroxides and hydroxyl radicals. Free Radic Biol Med 1991; 11: 191–195.
Acknowledgements
This study was supported by Grants 01/0069 (1 and 2), 01/3047, PI03/0862 and RCMN C03/01 (RECAVA) of the Fondo de Investigaciones Sanitarias (FIS), of the Spanish Health Ministry and grant GRUPOS 03/101 from the Valencian Government.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdilla, N., Tormo, M., Fabia, M. et al. Impact of the components of metabolic syndrome on oxidative stress and enzymatic antioxidant activity in essential hypertension. J Hum Hypertens 21, 68–75 (2007). https://doi.org/10.1038/sj.jhh.1002105
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jhh.1002105
Keywords
This article is cited by
-
Serum Malondialdehyde Levels in Hypertensive Patients: A Non-invasive Marker of Oxidative Stress. A Systematic Review and Meta-analysis
High Blood Pressure & Cardiovascular Prevention (2022)
-
Microvascular function and oxidative stress in adult individuals with early onset of cardiovascular disease
Scientific Reports (2020)
-
Small dense LDL cholesterol is associated with metabolic syndrome traits independently of obesity and inflammation
Nutrition & Metabolism (2019)
-
Oxidatively modified forms of albumin in patients with risk factors of metabolic syndrome
Journal of Endocrinological Investigation (2014)
-
Oxidative stress is associated with the number of components of metabolic syndrome: LIPGENE study
Experimental & Molecular Medicine (2013)