Abstract
Background:
Contradicting results have been published regarding the effect of conjugated linoleic acid (CLA) on insulin resistance. However, only a few studies have used the euglycemic hyperinsulinemic clamp method, which is considered the standard for measuring insulin resistance.
Objective:
To evaluate if CLA as a mixture of the main isomers trans-10 cis-12 and cis-9 trans-11 affects the insulin resistance in healthy overweight and obese male and female adults.
Design:
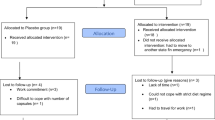
The main study was a randomized, double-blind, placebo-controlled trial with change in body composition as primary end point comprising 118 subjects receiving supplementation with either placebo (olive oil) or CLA (Clarinol) for 6 months. A sub-population of 49 subjects agreed additionally to participate in an euglycemic hyperinsulinemic clamp study at baseline and after 6 months of supplementation with study drug. The primary outcome was the change in glucose uptake (M) as measured by the hyperinsulinemic euglycemic glucose clamp method. Secondary outcomes were the correlates between insulin resistance and changes in body composition or blood chemistry parameters. Forty-one subjects completed the clamp test at both time points.
Results:
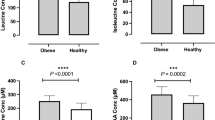
The median M of the CLA group was 11.0 mg min−1 lean body mass (lbm)−1 (n=24) at baseline, 10.3 mg min−1 lbm−1 (n=24) after 6 months, and the median difference was +0.21 mg min−1 lbm−1 (n=24). The median M of placebo group was 8.4 mg min−1 lbm−1 at baseline and 9.3 mg min−1 lbm−1 after 6 months and the median difference was −0.22 mg min−1 lbm−1 (n=17). No significant (P<0.05) differences were found within groups or between groups. Likewise, the glucose uptake insulin concentration ratio during clamp (M/I) was independent of treatment and time. Homeostasis model assessment (HOMA) and quantitative insulin sensitivity check index derived from fasting glucose and insulin were also independent of treatment and time, and HOMA for the clamp population (n=49) corresponded well with HOMA for the per protocol population (n=83). Correlation analysis showed that changes in M were inversely correlated to changes in glucohemoglobin (P=0.002), but did not correlate with changes in either glucose, insulin, insulin c-peptide, leptin, adiponectin or percent body fat.
Conclusions:
CLA does not affect glucose metabolism or insulin sensitivity in a population of overweight or obese volunteers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Atkinson RL . Conjugated linoleic acid for altering body composition and treating obesity. In: Yurawecz MP, Mossoba MM, Kramer JKG, Pariza MW, Nelson GJ (eds). Advances in Conjugated Linoleic Acid Research vol. 1 AOCS Champaign: Illinois, 1999. pp 348–353.
Berven G, Bye A, Hals O, Blankson H, Fagertun H, Thom E et al. Safety of conjugated linoleic acid (CLA) in overweight or obese human volunteers. Eur J Lipid Sci Technol 2000; 102: 455–462.
Blankson H, Stakkestad JA, Fagertun H, Thom E, Wadstein J, Gudmundsen O . Conjugated linoleic acid reduces body fat mass in overweight and obese humans. J Nutr 2000; 130: 2943–2948.
Gaullier JM, Halse J, Høye K, Kristiansen K, Fagertun H, Vik H et al. Conjugated linoleic acid (CLA) supplementation for one year reduces body fat mass in healthy, overweight humans. Am J Clin Nutr 2004; 79: 1118–1125.
Gaullier J, Halse J, Høye K, Kristiansen K, Fagertun H, Vik H et al. Supplementation with conjugated linoleic acid for 24 months is well tolerated by and reduces body fat mass in healthy, overweight humans. J Nutr 2005; 135: 778–784.
Kreider RB, Ferreira MP, Greenwood M, Wilson M, Almada AL . Effects of conjugated linoleic acid supplementation during resistance-training on body composition, bone density, strength, and selected hematological markers. J Strength Cond Res 2002; 3: 325–334.
Larsen TM, Toubro S, Gudmundsen O, Astrup A . Conjugated linoleic acid supplementation for 1 year does not prevent weight or body fat regain. Am J Clin Nutr 2006; 83: 606–612.
Lowery LM, Appicelli PA, Lemon PWR . Conjugated linoleic acid enhances muscle size and strength gains in novice bodybuilders [Abstract]. Med Sci Sport Exercise 1998; 30: 182.
Riserus U, Berglund L, Vessby B . Conjugated linoleic acid (CLA) reduced abdominal adipose tissue in obese middle-aged men with signs of the metabolic syndrome: a randomised controlled trial. Int J Obes Relat Metab Disord 2001; 25: 1129–1135.
Smedman A, Vessby B . Conjugated linoleic acid supplementation in humans-metabolic effects. Lipids 2001; 36: 773–781.
Terpstra AHM . Effect of conjugated linoleic acid on body composition and plasma lipids in humans: an overview of the literature. Am J Clin Nutr 2004; 79: 352–361.
Thom E, Wadstein J, Gudmundsen O . Conjugated linoleic acid reduces body fat in healthy exercising humans. J Int Med Res 2001; 29: 392–396.
Albers R, van der Wielen RP, Brink EJ, Hendriks HF, Dorovska-Taran VN, Mohede IC . Effects of cis-9, trans-11 and trans-10, cis-12 conjugated linoleic acid (CLA) isomers on immune function in healthy men. Eur J Clin Nutr 2003; 57: 595–603.
Noone EJ, Roche HM, Nugent AP, Gibney MJ . The effect of dietary supplementation using isomeric blends of conjugated linoleic acid on lipid metabolism in healthy human subjects. Br J Nutr 2002; 88: 243–251.
Aminot-Gilchrist DV, Anderson HDI . Insulin resistance-associated cardiovascular disease: potential benefits of conjugated linoleic acid. Am J Clin Nutr 2004; 79: 1159S–1163S.
Eyjolfson V, Spriet L, Dyck D . Conjugated linoleic acid improves insulin sensitivity in young, sedentary humans. Med Sci Sport Exercise 2004; 36: 814–820.
Moloney F, Yeow T, Mullen A, Nolan J, Roche H . Conjugated linoleic acid supplementation, insulin sensitivity, and lipoprotein metabolism in patients with type 2 diabetes mellitus. Am J Clin Nutr 2004; 80: 887–895.
Riserus U, Arner P, Brismar K, Vessby B . Treatment with dietary trans10cis12 conjugated linoleic acid causes isomer-specific insulin resistance in obese men with the metabolic syndrome. Diabetes Care 2002; 25: 1516–1521.
Whigham LD, O'Shea M, Mohede ICM, Walaski HP, Atkinson RL . Safety profile of conjugated linoleic acid in a 12-month trial in obese humans. Food Chem Toxicol 2004; 42: 1701–1709.
Riserus U, Vessby B, Arnlov J, Basu S . Effects of cis-9, trans-11 conjugated linoleic acid supplementation on insulin sensitivity, lipid peroxidation, and proinflammatory markers in obese men. Am J Clin Nutr 2004; 80: 279–283.
Riserus U, Vessby B, Arner P, Zethelius B . Supplementation with trans10cis12-conjugated linoleic acid induces hyperproinsulinaemia in obese men: close association with impaired insulin sensitivity. Diabetologia 2004; 47: 1016–1019.
Riserus U, Basu S, Jovinge S, Fredrikson GN, Arnlov J, Vessby B . Supplementation with conjugated linoleic acid causes isomer-dependent oxidative stress and elevated C-reactive protein: a potential link to fatty acid-induced insulin resistance. Circulation 2002; 106: 1925–1929.
DeFronzo RA, Tobin JD, Andres R . Glucose clamp technique: a method for quantifying insulin resistance. Am J Physiol 1979; 237: E214–E223.
Stern SE, Williams K, Ferrannini E, DeFronzo RA, Bogardus C, Stern MP . Identification of individuals with insulin resistance using routine clinical measurements. Diabetes 2005; 54: 333–339.
McAuley KA, Williams SM, Mann JI, Walker RJ, Lewis-Barned NJ, Temple LA et al. Diagnosing insulin resistance in the general population. Diabetes Care 2001; 24: 460–464.
Katz A, Nambi SS, Mather K, Baron AD, Follmann DA, Sullivan G et al. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab 2000; 85: 2402–2410.
Stumvoll M, Mitrakou A, Pimenta W, Jenssen T, Yki-Järvinen H, Haeften TV et al. Use of oral glucose tolerance test to assess insulin release and insulin sensitivity. Diabetes Care 2000; 23: 295–301.
Virtanen KA, Iozzo P, Hällsten K, Huupponen R, Parkkola R, Janatuinen T et al. Increased fat mass compensates for insulin resistance in abdominal obesity and type 2 diabetes. Diabetes 2005; 54: 2720–2726.
Birkeland KI, Kilhovd B, Thorsby P, Torjesen PA, Ganss R, Vaaler S et al. Heterogeneity of non-insulin-dependent diabetes expressed as variability in insulin sensitivity, β-cell function and cardiovascular risk profile. Diabetic Med 2003; 20: 37–45.
Kamphuis MM, Lejeune MP, Saris WH, Westerterp-Plantenga MS . The effect of conjugated linoleic acid supplementation after weight loss on body weight regain, body composition, and resting metabolic rate in overweight subjects. Int J Obes Relat Metab Disord 2003; 27: 840–847.
Song H, Grant I, Rotondo D, Mohede I, Sattar N, Heys S et al. Effect of CLA supplementation on immune function in young healthy volunteers. Eur J Clin Nutr 2005; 59: 508–517.
Tricon S, Burdge G, Kew S, Banerjee T, Russell J, Grimble R et al. Effects of cis-9,trans-11 and trans-10,cis-12 conjugated linoleic acid on immune cell function in healthy humans. Am J Clin Nutr 2004; 80: 1626–1633.
Malpuech-Brugere C, Verboeket-van de Venne WP, Mensink RP, Arnal MA, Morio B, Brandolini M et al. Effects of two conjugated linoleic acids isomers on body fat mass in overweight humans. Obes Res 2004; 12: 591–598.
McAuley KA, Williams SM, Mann JI, Goulding A, Chisholm A, Wilson N et al. Intensive lifestyle changes are necessary to improve insulin sensitivity: a randomized controlled trial. Diabetes Care 2002; 25: 445–452.
Acknowledgements
We are very thankful to the clinical nurses Bente Ryen, Lill Johannessen and Sissel Paulsen Sundeng for their active contribution in the success of this study. Lipid Nutrition, a division of Loders Croklaan has supported this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Syvertsen, C., Halse, J., Høivik, H. et al. The effect of 6 months supplementation with conjugated linoleic acid on insulin resistance in overweight and obese. Int J Obes 31, 1148–1154 (2007). https://doi.org/10.1038/sj.ijo.0803482
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0803482
Keywords
This article is cited by
-
A review on effects of conjugated linoleic fatty acid (CLA) upon body composition and energetic metabolism
Journal of the International Society of Sports Nutrition (2015)
-
The relationship between high-fat dairy consumption and obesity, cardiovascular, and metabolic disease
European Journal of Nutrition (2013)
-
Les acides linoléiques conjugués (CLA) permettent-ils de lutter contre l’obésité sans risque ?
Obésité (2013)
-
The efficacy of long-term conjugated linoleic acid (CLA) supplementation on body composition in overweight and obese individuals: a systematic review and meta-analysis of randomized clinical trials
European Journal of Nutrition (2012)
-
Conjugated Linoleic Acid Isomers, t10c12 and c9t11, are Differentially Incorporated into Adipose Tissue and Skeletal Muscle in Humans
Lipids (2009)