Abstract
Background:
No other studies have compared the relationship between body mass index (BMI) and health-related quality of life (HRQL) on more than one utility measure. Estimating the HRQL effects of obesity on a (common) utility scale enables the relative cost-effectiveness of interventions designed to alleviate obesity to be estimated.
Objective:
To examine the relationship between BMI and HRQL according to the EQ-5D, EuroQol visual analogue scale (EQ-VAS) and SF-6D.
Methods:
Patients aged ⩾45 years at one UK general practice were asked to complete the EQ-5D, EQ-VAS, SF-36 questionnaire (used to derive the SF-6D), and information on their characteristics and co-morbidity. Body mass index was categorized according to the World Health Organization (WHO) recommendations. Regression analysis was used to compare the HRQL of normal BMI patients to the HRQL of patients in other BMI categories, while controlling for patient characteristics and co-morbidity.
Results:
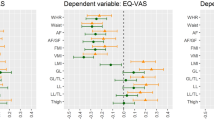
A total of 1865 patients responded (67%), mean BMI 26.0 kg/m2, 16% obese (BMI⩾30). Patients with back pain, hip pain, knee pain, asthma, diabetes or osteoarthritis were also significantly more likely to be obese. After controlling for other factors, compared to normal BMI patients, obese patients had a lower HRQL according to the EQ-5D (P<0.01), EQ-VAS (P<0.001) and SF-6D (P<0.001). Pre-obese patients were not estimated to have a significantly lower HRQL, and underweight patients were only estimated to have a significantly lower HRQL according to the SF-6D. These results arose because, on the EQ-5D, obese patients were found to have significantly more problems with mobility and pain, compared to physical functioning, social functioning and role limitations on the SF-6D. Whereas, according to the SF-6D, underweight patients had significantly more problems on the dimension of role limitation.
Conclusion:
The EQ-5D, EQ-VAS and SF-6D were in agreement that, relative to a normal BMI, obesity is associated with a lower HRQL, even after controlling for patient characteristics and co-morbidity. These three measures are thereby sensitive to the HRQL effects of obesity and can be used to estimate the cost-effectiveness of interventions designed to alleviate obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Barofsky I, Fontaine KR, Cheskin LJ . Pain in the obese: impact on health-related quality-of-life. Ann Behav Med 1997; 19: 408–410.
Brown WJ, Dobson AJ, Mishra G . What is a healthy weight for middle aged women? Int J Obes Relat Metab Disord 1998; 22: 520–528.
Han TS, Tijhuis MA, Lean ME, Seidell JC . Quality of life in relation to overweight and body fat distribution. Am J Public Health 1998; 88: 1814–1820.
Hill AJ, Williams J . Psychological health in a non-clinical sample of obese women. Int J Obes Relat Metab Disord 1998; 22: 578–583.
Lean ME, Han TS, Seidell JC . Impairment of health and quality of life in people with large waist circumference. Lancet 1998; 351: 853–856.
Le Pen C, Levy E, Loos F, Banzet MN, Basdevant A . ‘Specific’ scale compared with ‘generic’ scale: a double measurement of the quality of life in a French community sample of obese subjects. J Epidemiol Community Health 1998; 52: 445–450.
Fine JT, Colditz GA, Coakley EH, Moseley G, Manson JE, Willett WC et al. A propective study of weight change and health-related quality of life in women. JAMA 1999; 282: 2136–2142.
Lean ME, Han TS, Seidell JC . Impairment of health and quality of life using new US federal guidelines for the identification of obesity. Arch Intern Med 1999; 159: 837–843.
Doll HA, Petersen SEK, Stewart-Brown SL . Obesity and physical and emotional well-being: associations between body mass index, chronic illness, and the physical and mental components of the SF-36 questionnaire. Obes Res 2000; 8: 160–170.
Ford ES, Moriarty DG, Zack MM, Mokdad AH, Chapman DP . Self-reported body mass index and health-related quality of life: findings from the Behavioral Risk Factor Surveillance System. Obes Res 2001; 9: 21–31.
Larsson U, Karlsson J, Sullivan M . Impact of overweight and obesity on health-related quality of life – a swedish population study. Int J Obes Relat Metab Disord 2002; 26: 417–424.
Heo M, Allison DB, Faith MS, Zhu S, Fontaine KR . Obesity and quality of life: mediating effects of pain and comorbidities. Obes Res 2003; 11: 209–216.
Groessl EJ, Kaplan RM, Barrett-Connor E, Ganiats TG . Body mass index and quality of well-being in a community of older adults. Am J Prev Med 2004; 26: 126–129.
Yan LL, Daviglus ML, Liu K, Pirzada A, Garside DB, Schiffer L et al. BMI and health-related quality of life in adults 65 years and older. Obes Res 2004; 12: 69–76.
Jia H, Lubetkin EI . The impact of obesity on health-related quality-of-life in the general adult US population. J Public Health (Oxford) 2005; 27: 156–164.
Gold MR, Seigel JE, Russell LB, Weinstein MC . Cost-Effectiveness in Health and Medicine. Oxford University Press: New York, 1996.
National Institute of Health and Clinical Excellence. Guide to the Methods of Technology Appraisal. Abba Litho Sales Limited: London, UK, 2004.
Brooks R . EuroQol: the current state of play. Health Policy 1996; 37: 53–72.
Brazier J, Roberts J, Deverill M . The estimation of a preference-based measure of health from the SF-36. J Health Econ 2002; 21: 271–292.
Ware JE, Sherbourne C . The MOS 36 item short-form health survey: conceptual framework and item selection. Med Care 1992; 30: 473–483.
Longworth L, Bryan S . An empirical comparison of EQ-5D and SF-6D in liver transplant patients. Health Econ 2003; 12: 1061–1067.
Conner-Spady B, Suarez-Almazor ME . Variation in the estimation of quality-adjusted life-years by different preference-based instruments. Med Care 2003; 41: 791–801.
Pickard AS, Johnson JA, Feeny DH . Responsiveness of generic health-related quality of life measures in stroke. Qual Life Res 2005; 14: 207–219.
Brazier JE, Deverill M, Green C, Harper R, Booth A . A review of the use of health status measures in economic evaluation. Health Technol Assess 1999; 3: 1–164.
Hakim Z, Wolf A, Garrison LP . Estimating the effect of body mass index on health state preferences. Pharmacoeconomics 2002; 20: 393–404.
Spector TD, Hart DJ, Doyle DV . Incidence and progression of osteoarthritis in women with unilateral knee disease in the general population: the effect of obesity. Ann Rheum Dis 1994; 53: 565–568.
Andersen RE, Crespo CJ, Bartlett SJ, Bathon JM, Fontaine KR . Relationship between body weight gain and significant knee, hip, and back pain in older Americans. Obes Res 2003; 11: 1159–1162.
National Task Force on the Prevention and Treatment of Obesity. Overweight, obesity, and health risk. Arch Intern Med 2000; 160: 898–904.
Ronmark E, Andersson C, Nystrom L, Forsberg B, Jarvholm B, Lundback B . Obesity increases the risk of incident asthma among adults. Eur Respir J 2005; 25: 282–288.
Escalante A, Haas RW, del Rincon I . Measurement of global functional performance in patients with rheumatoid arthritis using rheumatology function tests. Arthritis Res Ther 2004; 6: S315–S325.
WHO. Obesity: Preventing and Managing the Global Epidemic. Report of a WHO consultation on Obesity. WHO: Geneva, 2001.
National Institutes of Health. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults – the evidence report. Obes Res 1998; 6 (Suppl 2): 51S–209S.
Torrance GW . Measurement of health state utilities for economic appraisal. J Health Econ 1986; 5: 1–30.
Dolan P, Gudex C, Kind P, Williams A . A Social Tariff for the EuroQol: Results from a UK General Population Survey (Discussion Paper 138). Centre for Health Economics, University of York, 1995.
Gerard K, Nicholson T, Mullee M, Mehta R, Roderick P . EQ-5D versus SF-6D in an OLDER, CHRONIcally Ill Patient Group. Appl Health Econ Health Policy 2004; 3: 91–102.
Marra CA, Woolcott JC, Kopec JA, Shojania K, Offer R, Brazier JE et al. A comparison of generic, indirect utility measures (the HUI2, HUI3, SF-6D, and the EQ-5D) and disease-specific instruments (the RAQoL and the HAQ) in rheumatoid arthritis. Soc Sci Med 2005; 60: 1571–1582.
Mokdad AH, Bowman BA, Ford ES, Vinicor F, Marks JS, Koplan JP . The continuing epidemics of obesity and diabetes in the United States. JAMA 2001; 286: 1195–1200.
Szende A, Svensson K, Stahl E, Meszaros A, Berta GY . Psychometric and utility-based measures of health status of asthmatic patients with different disease control level. Pharmacoeconomics 2004; 22: 537–547.
Bryan S, Longworth L . Measuring health-related utility: why the disparity between EQ-5D and SF-6D? Eur J Health Econ 2005; 6: 253–260.
Barton GR, Bankart J, Davis AC, Summerfield AQ . Comparing utility scores before and after hearing-aid provision: results according to the EQ-5D, HUI3 and SF-6D. Appl Health Econ Health Policy 2004; 3: 103–105.
Fisk JD, Brown MG, Sketris IS, Metz LM, Murray TJ, Stadnyk KJ . A comparison of health utility measures for the evaluation of multiple sclerosis treatments. J Neurol Neurosurg Psychiatry 2005; 76: 58–63.
Colditz GA . Economic costs of obesity and inactivity. Med Sci Sports Exerc 1999; 31: S663–S667.
Palta M, Prineas RJ, Berman R, Hannan P . Comparison of self-reported and measured height and weight. Am J Epidemiol 1982; 115: 223–230.
Rowland ML . Self-reported weight and height. Am J Clin Nutr 1990; 52: 1125–1133.
Acknowledgements
We thank all patients who completed the Lifestyle Interventions for Knee Pain (LIKP) study questionnaire. The LIKP study was funded by the UK Arthritis Research Campaign (ARC) (Grant number 13550). PhD funding for Garry Barton was provided by the UK Economic & Social Research Council (ESRC) (PTA-037-2004-00051).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sach, T., Barton, G., Doherty, M. et al. The relationship between body mass index and health-related quality of life: comparing the EQ-5D, EuroQol VAS and SF-6D. Int J Obes 31, 189–196 (2007). https://doi.org/10.1038/sj.ijo.0803365
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0803365
Keywords
This article is cited by
-
Mapping IWQOL-Lite onto EQ-5D-5L and SF-6Dv2 among overweight and obese population in China
Quality of Life Research (2024)
-
Differentials in Health and Wellbeing in Older Adults with Obesity in England: A Cross-Sectional Analysis Using the English Longitudinal Study of Ageing
Journal of Population Ageing (2023)
-
Diabesity: a superadded effect contributing to worse total primary hip replacement operative outcomes for patients with diabetes and obesity
Archives of Orthopaedic and Trauma Surgery (2022)
-
The association between obesity and quality of life: a retrospective analysis of a large-scale population-based cohort study
BMC Public Health (2021)
-
Factors associated with health-related quality of life in women using path analyses: mediation effect of the adiposity traits
BMC Women's Health (2021)