Abstract
Objective:
To examine the contribution of depressed mood in obese subjects on the prediction of a future coronary heart disease event (CHD).
Design:
A prospective population-based cohort study of three independent cross-sectional surveys with 6239 subjects, 45–74 years of age and free of diagnosed CHD, stroke and cancer. During a mean follow-up of 7 years, 179 CHD events occurred among men and 50 events among women.
Subjects:
A total of 737 (23%) male and 773 (26%) female subjects suffering from obesity (BMI ⩾30 kg/m2).
Measurements:
Body weight determined by trained medical staff following a standardized protocol; standardized questionnaires to assess subsyndromal depressive mood and other psychosocial features.
Results:
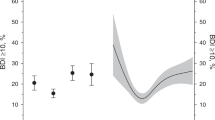
The main effect of obesity to predict a future CHD (hazard ratio, HR=1.38, 95% CI 1.03–1.84; P=0.031) and the interaction term of obesity by depression (HR=1.73, 95% CI 0.98–3.05; P=0.060) were borderline significant, both covariate adjusted for multiple risk factors. Relative to the male subgroup with normal body weight and no depression, the male obese group with no depression was not at significantly increased risk for CHD events (HR=1.17, 95% CI 0.76–1.80; P=0.473) whereas CHD risk in males with both obesity and depressed mood was substantially increased (HR=2.32, 95% CI 1.45–3.72, P>0.0001). The findings for women were similar, however, not significant probably owing to lack of power associated with low event rates. Combining obesity and depressed mood resulted in a relative risk to suffer from a future CHD event of HR 1.84 (95% CI 0.79–4.26; P=0.158).
Conclusions:
Depressed mood substantially amplifies the CHD risk of middle-aged obese, but otherwise apparently healthy men. The impact of depression on the obesity risk in women is less pronounced.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Wilson PW, D'Agostino RB, Sullivan L, Parise H, Kannel WB . Overweight and obesity as determinants of cardiovascular risk: the Framingham experience. Arch Intern Med 2002; 162: 1867–1872.
Festa A, D'Agostino Jr R, Williams K, Karter AJ, Mayer-Davies EJ, Tracy RP et al. The relation of body fat mass and distribution to markers of chronic inflammation. Int J Obes 2001; 25: 1407–1415.
Dyer AR, Stamler J, Garside DB, Greenland P . Long-term consequences of body mass index for cardiovascular mortality: the Chicago Heart Association Detection Project in Industry study. Ann Epidemiol 2004; 14: 101–108.
Manson JE, Colditz GA, Stampfer MJ, Willett WC, Rosner B, Monson RR et al. A prospective study of obesity and risk of coronary heart disease in women. N Engl J Med 1990; 29: 882–889.
Lee IM, Manson JE, Hennekens CH, Paffenbarger Jr RS . Body weight and mortality. A 27-year follow-up of middle-aged men. JAMA 1993; 270: 2823–2828.
Rugulies R . Depression as a predictor for coronary heart disease. A review and meta-analysis. Am J Prev Med 2002; 23: 51–61.
Wulsin LR, Singal BM . Do depressive symptoms increase the risk for the onset of coronary disease. A systematic quantitative review. Psychosom Med 2003; 65: 259–267.
Goodman E, Whitaker RC . A prospective study of the role of depression in the development and persistence of adolescent obesity. Pediatrics 2002; 110: 497–504.
Faith MS, Matz PE, Jorge MA . Obesity–depression associations in the population. J Psychosom Res 2002; 53: 935–942.
Roberts RE, Deleger S, Strawbridge WJ, Kaplan GA . Prospective association between obesity and depression: evidence from the Alameda County Study. Int J Obes 2003; 28: 5143–5521.
Dong C, Sanchez LE, Price RA . Relationship of obesity to depression: a family-based study. Int J Obes 2004; 28: 790–795.
WHO MONICA Project Principal Investigators (prepared by H Tunstall-Pedoe). Myocardial infarction and coronary deaths in the World Health Organization MONICA Project. Registration procedures, event rates, and case-fatality rates in 38 populations from 21 countries in four continents. Circulation 1994; 90: 583–612.
WHO MONICA Project Principal Investigators,MONICA Psychosocial Optional study. Suggested Measurement Instruments. Regional Office for Europe, 1989.
Zerssen Dv . Die Beschwerden-Liste Klinische Selbstbeurteilungsfragebögen aus dem Münchner Psychiatrischen Informationssystem (Psychis). Weinheim (Beltz), 1976.
Ladwig KH, Marten-Mittag B, Baumert J, Doering A, Loewel H . Case-finding for vital exhaustion and depressive mood in the general population: reliability and validity of a symptom driven diagnostic scale. Results from the MONICA Augsburg Study. Ann Epidemiol 2004; 14: 332–338.
Seeman TE, Kaplan GA, Knudsen L, Cohen R, Guralnik J . Social network ties and mortality among the elderly in the Alameda County. Am J Epidemiol 1987; 126: 714–723.
Mallon L, Broman JE, Hetta J . Sleep complaints predict coronary artery disease mortality in males: a 12-year follow-up study of a middle-aged Swedish population. J Int Med 2002; 251: 207–216.
Loewel H, Lewis M, Hoermann A, Keil U . Case finding, data quality aspects and comparability of myocardial infarction registers: results of a south German register study. J Clin Epidemiol 1991; 44: 249–260.
Cox DR, Oakes D . Analysis of Survival Data. Chapman & Hall: London, 1984.
Peeters A, Barendregt JJ, Willekens F, Mackenbach JP, Al Mamun A, Bonneux L . Obesity in adulthood and its consequences for life expectancy: a life-table analysis. Ann Intern Med 2003; 138: 24–32.
Klein S, Burke LE, Bray GA, Blair S, Allison DB, Pi-Sunyer X et al. Clinical implications of obesity with specific focus on cardiovascular disease: a statement for professionals from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism: endorsed by the American College of Cardiology Foundation. Circulation 2004; 110: 2952–2967.
McGee DL, Reed DM, Yano K, Kagan A, Tillotson J . Ten-year incidence of coronary heart disease in the Honolulu Heart Program. Relationship to nutrient intake. Am J Epidemiol 1984; 119: 667–676.
Wilcosky T, Hyde J, Anderson JJ, Bangdiwala S, Duncan B . Obesity and mortality in the lipid research clinics program follow-up study. J Clin Epidemiol 1990; 43: 743–752.
Yao CH, Slattery ML, Jacobs Jr DR, Folsom AR, Nelson ET . Anthropometric predictors of coronary heart disease and total mortality: findings from the US Railroad Study. Am J Epidemiol 1991; 134: 1278–1289.
Jousilahti P, Tuomilehto J, Vartiainen E, Pekkanen J, Puska P . Body weight, cardiovascular risk factors, and coronary mortality. 15-year follow-up of middle-aged men and women in eastern Finland. Circulation 1996; 93: 1372–1379.
Dorn JM, Schisterman EF, Winkelstein Jr W, Trevisan M . Body mass index and mortality in a general population sample of men and women. The Buffalo Health Study. Am J Epidemiol 1997; 146: 919–931.
Yao CH, Slattery ML, Jacobs Jr DR, Folsom AR, Nelson ET . Anthropometric predictors of coronary heart disease and total mortality: findings from the US Railroad Study. Am J Epidemiol 1991; 134: 1278–1289.
Gray RS, Fabsitz RR, Cowan LD, Lee ET, Welty TK, Jablonski KA et al. Relation of generalized and central obesity to cardiovascular risk factors and heart disease in a sample of American Indians: the Strong Heart Study. Int J Obes 2000; 24: 849–860.
Hopkinson G, Bland RC . Depressive syndromes in grossly obese women. Can J Psychiatry 1982; 18: 24–28.
Istvan J, Zavela K, Weidner G . Body weight and psychological distress in NHANES I. Int J Obes 1992; 16: 999–1003.
Hassan MK, Joshi AV, Amonkar MM . Obesity and health related quality of life: a cross-sectional analysis of the US population. Int J Obes 2003; 27: 1227–1232.
Siegel JM, Yancey AK, McCarthy WJ . Overweight and depressive symptoms among African-American women. Prev Med 2000; 31: 232–240.
Carpenter KM, Hasin DS, Allison DB, Faith MS . Relationships between obesity and DSM-IV major depressive disorder, suicide ideation, and suicide attempts: results from a general population study. Am J Public Health 2000; 90: 251–257.
Roberts RE, Kaplan GA, Shema SJ, Strawbridge WJ . Are the obese at greater risk for depression? Am J Epidemiol 2000; 152: 935–942.
Roberts RE, Strawbridge WJ, Deleger S, Kaplan GA . Are the fat more jolly? Ann Behav Med 2002; 53: 935–942.
Onyike CU, Crum RM, Lee HB, Lyketsos CG, Eaton WW . Is obesity associated with major depression? Results from the Third National Health and Nutrition Examination Survey. Am J Epidemiol 2003; 158: 1139–1147.
Crisp AH, McGuiness B . Jolly fat: relation between obesity and psychoneurosis in the general population. BMJ 1976; 1: 7–9.
Hallstrom T, Noppa H . Obesity in women in relation to mental illness, social factors and personality traits. J Psychosom Res 1981; 25: 75–82.
Palinkas LA, Wingard DL, Barrett-Connor E . Depressive symptoms in overweight and obese older adults: a test of the ‘jolly fat’ hypothesis. J Psychosom Res 1996; 40: 59–66.
Katz JR, Taylor NF, Goodrick S, Perry L, Yudkin JS, Coppack SW . Central obesity, depression and the hypothalamo-pituitary-adrenal axis in men and postmenopausal women. Int J Obes 2000; 25: 246–251.
Weber-Hamann B, Hentschel F, Kniest A, Deuschle M, Colla M, Lederbogen F et al. Hypercortisolemic depression is associated with increased intra-abdominal fat. Psychosom Med 2002; 64: 274–277.
Miller GE, Stetler CA, Carney RM, Freedland KE, Banks WA . Clinical depression and inflammatory risk markers for coronary heart disease. Am J Cardiol 2002; 90: 1279–1283.
Ladwig KH, Marten-Mittag B, Löwel H, Döring A, Koenig W . Influence of depressive mood on the association of CRP and obesity in 3205 middle aged healthy men. Results from the MONICA Augsburg Study. Brain Behav Immun 2003; 17: 268–275.
Appels A, Mulder P . Excess fatigue as a precursor of myocardial infarction. Eur Heart J 1988; 9: 758–764.
Wassertheil-Smoller S, Applegate WB, Berge K, Chang CJ, Davis BR, Grimm Jr R et al. Change in depression as a precursor of cardiovascular events. Arch Internal Med 1996; 156: 553–561.
Schulz R, Beach SR, Ives DG, Martire LM, Ariyo AA, Kop WJ . Association between depression and mortality in older adults: the Cardiovascular Health Study. Arch Internal Med 2000; 160: 1761–1768.
Kop WJ, Gottdiener JS, Tangen CM, Fried LP, McBurnie MA, Walston J et al. Inflammation and coagulation factors in persons >65 years of age with symptoms of depression but without evidence of myocardial ischemia. Am J Cardiol 2002; 89: 419–424.
Smalley KJ, Knerr AN, Kendrick ZV, Colliver JA, Owen OE . Reassessment of body mass indices. Am J Clin Nutr 1990; 52: 405–408.
Gallagher D, Visser M, Sepulveda D, Pierson RN, Harris T, Heymsfield SB . How useful is body mass index for comparison of body fatness across age, sex, and ethnic groups? Am J Epidemiol 1996; 143: 228–239.
Acknowledgements
The MONICA–KORA Augsburg study is financed by the GSF-National Research Center for Environment and Health. We are grateful to the MONICA Augsburg teams, which have conducted the studies and identified the clinical outcomes of the survey participants. We also thank Andrea Schneider for data handling and quality control. We thank Ulrich Keil, MD, who initiated the MONICA Study Augsburg as first principal investigator.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ladwig, KH., Marten-Mittag, B., Löwel, H. et al. Synergistic effects of depressed mood and obesity on long-term cardiovascular risks in 1510 obese men and women: results from the MONICA–KORA Augsburg Cohort Study 1984–1998. Int J Obes 30, 1408–1414 (2006). https://doi.org/10.1038/sj.ijo.0803285
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0803285
Keywords
This article is cited by
-
Comorbid depression and obesity, and its transition on the risk of functional disability among middle-aged and older Chinese: a cohort study
BMC Geriatrics (2022)
-
The combined role of obesity and depressive symptoms in the association with ischaemic heart disease and its subtypes
Scientific Reports (2022)
-
Food and Mood: the Corresponsive Effect
Current Nutrition Reports (2020)
-
The Effect of a Behavioral Weight-Loss Intervention on Depressive Symptoms Among Latino Immigrants in a Randomized Controlled Trial
Journal of Immigrant and Minority Health (2018)
-
Epidemiology in Germany—general development and personal experience
European Journal of Epidemiology (2017)